Abstract
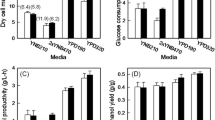
Polylactic acid (PLA) is an important renewable polymer, but current processes for producing its precursor, lactic acid, suffer from process inefficiencies related to the use of bacterial hosts. Therefore, improving the capacity of Saccharomyces cerevisiae to produce lactic acid is a promising approach to improve industrial production of lactic acid. As one such improvement required, the lactic acid tolerance of yeast must be significantly increased. To enable improved tolerance, we employed an RNAi-mediated genome-wide expression knockdown approach as a means to rapidly identify potential genetic targets. In this approach, several gene knockdown targets were identified which confer increased acid tolerance to S. cerevisiae BY4741, of which knockdown of the ribosome-associated chaperone SSB1 conferred the highest increase (52 %). This target was then transferred into a lactic acid-overproducing strain of S. cerevisiae CEN.PK in the form of a knockout and the resulting strain demonstrated up to 33 % increased cell growth, 58 % increased glucose consumption, and 60 % increased l-lactic acid production. As SSB1 contains a close functional homolog SSB2 in yeast, this result was counterintuitive and may point to as-yet-undefined functional differences between SSB1 and SSB2 related to lactic acid production. The final strain produced over 50 g/L of lactic acid in under 60 h of fermentation.





Similar content being viewed by others
Abbreviations
- RNAi:
-
RNA interference
- LAB:
-
Lactic acid bacteria
- PLA:
-
Polylactic acid
- L-LDH:
-
l-Lactate dehydrogenase
- PDC:
-
Pyruvate decarboxylase
- SSB1:
-
Yeast cytoplasmic chaperone
- RPL14B:
-
Yeast ribosomal 60S subunit L41B
- ADH1:
-
Yeast alcohol dehydrogenase
- YSC:
-
Yeast synthetic complete medium
- HPLC:
-
High-performance liquid chromatography
- CYB2:
-
Yeast l-lactate cytochrome-c oxidoreductase
- GPD1:
-
Yeast glycerol-3-phosphate dehydrogenase
- PGK1:
-
Yeast phosphoglycerate kinase
- CCW12:
-
Yeast cell wall mannoprotein
References
Adachi E, Torigoe M, Sugiyama M, Nikawa J-I, Shimizu K (1998) Modification of metabolic pathways of Saccharomyces cerevisiae by the expression of lactate dehydrogenase and deletion of pyruvate decarboxylase genes for the lactic acid fermentation at low pH value. J Ferment Bioeng 86:284–289. doi:10.1016/S0922-338X(98)80131-1
Alani E, Cao L, Kleckner N (1987) A method for gene disruption that allows repeated use of URA3 selection in the construction of multiply disrupted yeast strains. Genetics 116:541–545
Aldiguier AS, Alfenore S, Cameleyre X, Goma G, Uribelarrea JL, Guillouet SE, Molina-Jouve C (2004) Synergistic temperature and ethanol effect on Saccharomyces cerevisiae dynamic behaviour in ethanol bio-fuel production. Bioprocess Biosyst Eng 26:217–222. doi:10.1007/s00449-004-0352-6
Alper H, Moxley J, Nevoigt E, Fink GR, Stephanopoulos G (2006) Engineering yeast transcription machinery for improved ethanol tolerance and production. Science 314:1565–1568. doi:10.1126/science.1131969
Bischoff KM, Liu S, Hughes SR, Rich JO (2010) Fermentation of corn fiber hydrolysate to lactic acid by the moderate thermophile Bacillus coagulans. Biotechnol Lett 32:823–828. doi:10.1007/s10529-010-0222-z
Blomberg A, Adler L (1989) Roles of glycerol and glycerol-3-phosphate dehydrogenase (NAD +) in acquired osmotolerance of Saccharomyces cerevisiae. J Bacteriol 171:1087–1092
Bustos G, Moldes AB, Cruz JM, Dominguez JM (2005) Influence of the metabolism pathway on lactic acid production from hemicellulosic trimming vine shoots hydrolyzates using Lactobacillus pentosus. Biotechnol Prog 21:793–798. doi:10.1021/bp049603v
Chaillou S, Bor YC, Batt CA, Postma PW, Pouwels PH (1998) Molecular cloning and functional expression in lactobacillus plantarum 80 of xylT, encoding the D-xylose-H+ symporter of Lactobacillus brevis. Appl Environ Microbiol 64:4720–4728
Chang AL, Wolf JJ, Smolke CD (2012) Synthetic RNA switches as a tool for temporal and spatial control over gene expression. Curr Opin Biotechnol 23:679–688. doi:10.1016/j.copbio.2012.01.005
Craig EA, Jacobsen K (1985) Mutations in cognate genes of Saccharomyces cerevisiae hsp70 result in reduced growth rates at low temperatures. Mol Cell Biol 5:3517–3524
Crook N, Alper HS (2012) Classical strain improvement. In: Patnaik R (ed) Engineering complex phenotypes in industrial strains. Wiley, Hoboken, NJ, pp 1–33. doi: 10.1002/9781118433034.ch1
Crook N, Alper HS (2013) Model-based design of synthetic, biological systems. Chem Eng Sci 103:2–11. doi:10.1016/j.ces.2012.12.022
Crook NC, Schmitz AC, Alper HS (2013) Optimization of a yeast RNA interference system for controlling gene expression and enabling rapid metabolic engineering. ACS Syn Bio 3:307–313. doi:10.1021/sb4001432
Da Silva NA, Srikrishnan S (2012) Introduction and expression of genes for metabolic engineering applications in Saccharomyces cerevisiae. FEMS Yeast Res 12:197–214. doi:10.1111/j.1567-1364.2011.00769.x
Datta R, Tsai S-P, Bonsignore P, Moon S-H, Frank JR (1995) Technological and economic potential of poly(lactic acid) and lactic acid derivatives. FEMS Microbiol Rev 16:221–231. doi:10.1016/0168-6445(94)00055-4
Dequin S, Barre P (1994) Mixed lactic acid-alcoholic fermentation by Saccharomyces cerevisiae expressing the Lactobacillus casei L(+)-LDH. Bio/technology (Nature Publishing Company) 12:173–177
Gong Y, Kakihara Y, Krogan N, Greenblatt J, Emili A, Zhang Z, Houry WA (2009) An atlas of chaperone-protein interactions in Saccharomyces cerevisiae: implications to protein folding pathways in the cell. Mol Syst Biol 5:275. doi:10.1038/msb.2009.26
Grath SM, van Sinderen D (eds) (2007) Bacteriophage: Genetics and molecular biology. Caister Academic Press, Norfolk
Hegemann JH, Heick SB (2011) Delete and repeat: a comprehensive toolkit for sequential gene knockout in the budding yeast Saccharomyces cerevisiae. In: Williams JA (ed) Methods in Molecular Biology., vol 765. Humana Press, New York, pp 189–206. doi:10.1007/978-1-61779-197-0_12
Hohmann S (2002) Osmotic stress signaling and osmoadaptation in yeasts. Microbiol Mol Biol Rev MMBR 66:300–372
Hu J, Zhang Z, Lin Y, Zhao S, Mei Y, Liang Y, Peng N (2015) High-titer lactic acid production from NaOH-pretreated corn stover by Bacillus coagulans LA204 using fed-batch simultaneous saccharification and fermentation under non-sterile condition. Bioresour Technol 182:251–257. doi:10.1016/j.biortech.2015.02.008
Ilmen M, Koivuranta K, Ruohonen L, Rajgarhia V, Suominen P, Penttila M (2013) Production of l-lactic acid by the yeast Candida sonorensis expressing heterologous bacterial and fungal lactate dehydrogenases. Microb Cell Fact 12:53. doi:10.1186/1475-2859-12-53
Ishida N, Saitoh S, Onishi T, Tokuhiro K, Nagamori E, Kitamoto K, Takahashi H (2006) The effect of pyruvate decarboxylase gene knockout in Saccharomyces cerevisiae on l-lactic acid production. Biosci Biotechnol Biochem 70:1148–1153. doi:10.1271/bbb.70.1148
Kim SY, Craig EA (2005) Broad sensitivity of Saccharomyces cerevisiae lacking ribosome-associated chaperone ssb or zuo1 to cations, including aminoglycosides. Eukaryot Cell 4:82–89. doi:10.1128/ec.4.1.82-89.2005
Kumar A, Snyder M (2001) Genome-wide transposon mutagenesis in yeast. In: Lundblad V, Struhl K (eds) Current protocols in molecular biology, chap 13. Wiley, Hoboken, NJ. doi: 10.1002/0471142727.mb1303s51
Lee JY, Kang CD, Lee SH, Park YK, Cho KM (2015) Engineering cellular redox balance in Saccharomyces cerevisiae for improved production of l-lactic acid. Biotechnol Bioeng 112:751–758. doi:10.1002/bit.25488
Liu L, Redden H, Alper HS (2013) Frontiers of yeast metabolic engineering: diversifying beyond ethanol and Saccharomyces. Curr Opin Biotechnol 24:1023–1030. doi:10.1016/j.copbio.2013.03.005
Luyten K, Albertyn J, Skibbe WF, Prior BA, Ramos J, Thevelein JM, Hohmann S (1995) Fps1, a yeast member of the MIP family of channel proteins, is a facilitator for glycerol uptake and efflux and is inactive under osmotic stress. EMBO J 14:1360–1371
Patel MA, Ou MS, Harbrucker R, Aldrich HC, Buszko ML, Ingram LO, Shanmugam KT (2006) Isolation and characterization of acid-tolerant, thermophilic bacteria for effective fermentation of biomass-derived sugars to lactic acid. Appl Environ Microb 72:3228–3235. doi:10.1128/aem.72.5.3228-3235.2006
Peisker K, Chiabudini M, Rospert S (2010) The ribosome-bound Hsp70 homolog Ssb of Saccharomyces cerevisiae. Biochim Biophys Acta 1803:662–672. doi:10.1016/j.bbamcr.2010.03.005
Pfund C, Lopez-Hoyo N, Ziegelhoffer T, Schilke BA, Lopez-Buesa P, Walter WA, Wiedmann M, Craig EA (1998) The molecular chaperone Ssb from Saccharomyces cerevisiae is a component of the ribosome-nascent chain complex. EMBO J 17:3981–3989. doi:10.1093/emboj/17.14.3981
Porro D, Bianchi MM, Brambilla L, Menghini R, Bolzani D, Carrera V, Lievense J, Liu C-L, Ranzi BM, Frontali L, Alberghina L (1999) Replacement of a metabolic pathway for large-scale production of lactic acid from engineered yeasts. Appl Environ Microb 65:4211–4215
Pronk JT, Yde Steensma H, Van Dijken JP (1996) Pyruvate metabolism in Saccharomyces cerevisiae. Yeast (Chichester, England) 12:1607–1633. doi:10.1002/(SICI)1097-0061(199612)12:16<1607:AID-YEA70>3.0.CO;2-4
Remize F, Cambon B, Barnavon L, Dequin S (2003) Glycerol formation during wine fermentation is mainly linked to Gpd1p and is only partially controlled by the HOG pathway. Yeast (Chichester, England) 20:1243–1253. doi:10.1002/yea.1041
Saitoh S, Ishida N, Onishi T, Tokuhiro K, Nagamori E, Kitamoto K, Takahashi H (2005) Genetically engineered wine yeast produces a high concentration of l-Lactic acid of extremely high optical purity. Appl Environ Microb 71:2789–2792. doi:10.1128/aem.71.5.2789-2792.2005
Si T, Luo Y, Bao Z, Zhao H (2015) RNAi-assisted genome evolution in Saccharomyces cerevisiae for complex phenotype engineering. ACS Syn Bio 4:283–291. doi:10.1021/sb500074a
Sun J, Alper H (2015) Metabolic engineering of strains: from industrial-scale to lab-scale chemical production. J Ind Microbiol Biotechnol 42:423–436. doi:10.1007/s10295-014-1539-8
Troutt AB, McHeyzer-Williams MG, Pulendran B, Nossal GJ (1992) Ligation-anchored PCR: a simple amplification technique with single-sided specificity. P Natl Acad Sci USA 89:9823–9825. doi:10.1073/pnas.89.20.9823
van Maris AJ, Winkler AA, Porro D, van Dijken JP, Pronk JT (2004) Homofermentative lactate production cannot sustain anaerobic growth of engineered Saccharomyces cerevisiae: possible consequence of energy-dependent lactate export. Appl Environ Microbiol 70:2898–2905
Wang M, Weiss M, Simonovic M, Haertinger G, Schrimpf SP, Hengartner MO, von Mering C (2012) PaxDb, a database of protein abundance averages across all three domains of life. Mol Cell Proteom MCP 11:492–500. doi:10.1074/mcp.O111.014704
Winzeler EA, Shoemaker DD, Astromoff A, Liang H, Anderson K, Andre B, Bangham R, Benito R, Boeke JD, Bussey H, Chu AM, Connelly C, Davis K, Dietrich F, Dow SW, El Bakkoury M, Foury F, Friend SH, Gentalen E, Giaever G, Hegemann JH, Jones T, Laub M, Liao H, Liebundguth N, Lockhart DJ, Lucau-Danila A, Lussier M, M’Rabet N, Menard P, Mittmann M, Pai C, Rebischung C, Revuelta JL, Riles L, Roberts CJ, Ross-MacDonald P, Scherens B, Snyder M, Sookhai-Mahadeo S, Storms RK, Veronneau S, Voet M, Volckaert G, Ward TR, Wysocki R, Yen GS, Yu K, Zimmermann K, Philippsen P, Johnston M, Davis RW (1999) Functional characterization of the S. cerevisiae genome by gene deletion and parallel analysis. Science 285:901–906. doi:10.1126/science.285.5429.901
Ye L, Kruckeberg AL, Berden JA, van Dam K (1999) Growth and glucose repression are controlled by glucose transport in Saccharomyces cerevisiae cells containing only one glucose transporter. J Bacteriol 181:4673–4675
Ye L, Zhou X, Hudari MS, Li Z, Wu JC (2013) Highly efficient production of l-lactic acid from xylose by newly isolated Bacillus coagulans C106. Bioresour Technol 132:38–44. doi:10.1016/j.biortech.2013.01.011
Zhao K, Qiao Q, Chu D, Gu H, Dao TH, Zhang J, Bao J (2013) Simultaneous saccharification and high titer lactic acid fermentation of corn stover using a newly isolated lactic acid bacterium Pediococcus acidilactici DQ2. Bioresour Technol 135:481–489. doi:10.1016/j.biortech.2012.09.063
Acknowledgments
This work was funded in part by Samsung Electronics Co., Ltd. under the Global Research Outreach Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical statement/conflict of interest
N. C., J. S., and H. A. do not have any financial or personal relationships that could inappropriately influence or bias the content of the paper. J. L. is an employee of Samsung Corporation.
Additional information
J. J. Lee and N. Crook contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, J.J., Crook, N., Sun, J. et al. Improvement of lactic acid production in Saccharomyces cerevisiae by a deletion of ssb1 . J Ind Microbiol Biotechnol 43, 87–96 (2016). https://doi.org/10.1007/s10295-015-1713-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-015-1713-7