Abstract
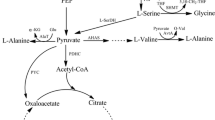
Fructose-1,6-bisphosphatase (FBPase) and fructokinase (ScrK) have important roles in regenerating glucose-6-phosphate in the pentose phosphate pathway (PPP), and thus increasing l-lysine production. This article focuses on the development of l-lysine high-producing strains by heterologous expression of FBPase gene fbp and ScrK gene scrK in C. glutamicum lysC fbr with molasses as the sole carbon source. Heterologous expression of fbp and scrK lead to a decrease of residual sugar in fermentation broth, and heterologous expression of scrK prevents the fructose efflux. Heterologous expression of fbp and scrK not only increases significantly the activity of corresponding enzymes but also improves cell growth during growth on molasses. FBPase activities are increased tenfold by heterologous expression of fbp, whereas the FBPase activity is only increase fourfold during co-expression of scrK and fbp. Compared with glucose, the DCW of heterologous expression strains are higher on molasses except co-expression of fbp and scrK strain. In addition, heterologous expression of fbp and scrK can strongly increase the l-lysine production with molasses as the sole carbon source. The highest increase (88.4 %) was observed for C. glutamicum lysC fbr pDXW-8-fbp-scrK, but the increase was also significant for C. glutamicum lysC fbr pDXW-8-fbp (47.2 %) and C. glutamicum lysC fbr pDXW-8-scrK (36.8 %). By-products, such as glycerol and dihydroxyacetone, are decreased by heterologous expression of fbp and scrK, whereas trehalose is only slightly increased. The strategy for enhancing l-lysine production by regeneration of glucose-6-phosphate in PPP may provide a reference to enhance the production of other amino acids during growth on molasses or starch.






Similar content being viewed by others
References
Jakobsen ØM, Brautaset T, Degnes KF, Heggeset TMB, Balzer S, Flickinger MC et al (2009) Overexpression of wild-type aspartokinase increases l-lysine production in the thermotolerant methylotrophic bacterium Bacillus methanolicus. Appl Environ Microbiol 75:652–661. doi:10.1128/AEM.01176-08
Kaffas M, Stephanopoulos G (2005) Strain improvement by metabolic engineering: lysine production as a case study for systems biology. Curr Opin Biotechnol 16:361–366. doi:10.1016/j.copbio.2005.04.010
Wittmann C, Becker J (2007) The l-lysine story: from metabolic pathways to industrial production. Microbiol Monographs 5:39–70. doi:10.1007/7171_2006_089
Becker J, Klopprogge C, Zelder O, Heinzle E, Wittmann C (2005) Amplified expression of fructose 1,6-bisphosphatase in Corynebacterium glutamicum increases in vivo flux through the pentose phosphate pathway and lysine production on different carbon sources. Appl Environ Microbiol 71:8587–8596. doi:10.1128/AEM.71.12.8587-8596.2005
Georgi T, Rittmann D, Wendisch VF (2005) Lysine and glutamate production by Corynebacterium glutamicum on glucose, fructose and sucrose: roles of malic enzyme and fructose-1,6-bisphosphatase. Metab Eng 7:291–301. doi:10.1016/j.ymben.2005.05.001
Lindner SN, Seibold GM, Henrich A, Krämer R, Wendisch VF (2011) Phosphotransferase system (PTS) independent glucose utilization in Corynebacterium glutamicum by inositol permeases and glucokinases. Appl Environ Microbiol 77:3571–3581. doi:10.1128/AEM.02713-10
Dominguez H, Rollin C, Guyonvarch A, Guerquin-Kern JL, Cocaign-Bousquet M, Lindley ND (1998) Carbon-flux distribution in the central metabolic pathways of Corynebacterium glutamicum during growth on fructose. Eur J Biochem 254:96–102. doi:10.1046/j.1432-1327.1998.2540096.x
Wittmann C, Kiefer P, Zelder O (2004) Metabolic fluxes in Corynebacterium glutamicum during lysine production with sucrose as carbon source. Appl Environ Microbiol 70:7277–7287. doi:10.1128/AEM.70.12.7277-7287.2004
Kiefer P, Heinzle E, Zelder O, Wittmann C (2004) Comparative metabolic flux analysis of lysine-producing Corynebacterium glutamicum cultured on glucose or fructose. Appl Environ Microbiol 70:229–239. doi:10.1128/AEM.70.1.229-239.2004
Pons A, Dussap CG, Pequinot C, Gros JB (1996) Metabolic flux distribution in Corynebacterium melassecola ATCC 17965 for various carbon sources. Biotechnol Bioeng 51:77–189. doi:10.1002/(SICI)1097-0290(19960720)51:2<177:AID-BIT7>3.0.CO;2-G
Peng X, Okai N, Vertès AA, Inatomi K, Inui M, Yukawa H (2011) Characterization of the mannitol catabolic operon of Corynebacterium glutamicum. Appl Microbiol Biotechnol 91:1375–1387. doi:10.1007/s00253-011-3352-x
Kopsahelis N, Nisiotou A, Kourkoutas Y, Panas P, Nycha GJE, Kanellaki M (2009) Molecular characterization and molasses fermentation performance of a wild yeast strain operating in an extremely wide temperature range. Bioresour Technol 100:4854–4862. doi:10.1016/j.biortech.2009.05.011
Olbrich H (2006) The molasses. Biotechnologie-Kempe GmbH, Berlin
Kopsahelis N, Agouridis N, Bekatorou A, Kanellaki M (2007) Comparative study of spent grains and delignified spent grains as yeast supports for alcohol production from molasses. Bioresour Technol 98:1440–1447. doi:10.1016/j.biortech.2006.03.030
Becker J, Klopprogge C, Herold A, Zelder O, Bolten CJ, Wittmann C (2007) Metabolic flux engineering of l-lysine production in Corynebacterium glutamicum—over expression and modification of G6P dehydrogenase. J Biotechnol 138:99–109. doi:10.1016/j.jbiotec.2007.05.026
Becker J, Zelder O, Häfner S, Schröder H, Wittmann C (2011) From zero to hero—design-based systems metabolic engineering of Corynebacterium glutamicum for l-lysine production. Metab Eng 13:159–168. doi:10.1016/j.ymben.2011.01.003
Sambrook J, Russel DV (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory Press, New York
van der Rest ME, Lange C, Molenaar D (1999) A heat shock following electroporation induces highly efficient transformation of Corynebacterium glutamicum with xenogeneic plasmid DNA. Appl Microbiol Biotechnol 52:541–545
Hou XH, Ge XY, Wu D, Qian H, Zhang WG (2012) Improvement of l-valine production at high temperature in Brevibacterium flavum by overexpressing ilvEBN r C genes. J Ind Microbiol Biotechnol 39:63–72. doi:10.1007/s10295-011-1000-1
Porter EV, Chassy BM, Holmlund CE (1980) Partial purification and properties of a mannofructokinase from Streptococcus mutans SL-1. Infect Immun 30:43–50
Periyasamy S, Venkatachalam S, Ramasamy S, Srinivasan V (2009) Production of bio-ethanol from sugar molasses using Saccharomyces cerevisiae. Modern Appl Sci 3:32–37
Moon MW, Kim HJ, Oh TK, Shin CS, Lee JS, Kim SJ, Lee JK (2005) Analyses of enzyme II gene mutants for sugar transport and heterologous expression of fructokinase gene in Corynebacterium glutamicum ATCC 13032. FEMS Microbiol Lett 244:259–266. doi:10.1016/j.femsle.2005.01.053
Dominguez H, Lingdley ND (1996) Complete sucrose metabolism requires fructose phosphotransferase activity in Corynebacterium glutamicum to ensure phosphorylation of liberated fructose. Appl Environ Microbiol 62:3878–3880
Aulkemeyer P, Ebner R, Heilenmann G, Jahreis K, Schmid K, Wrieden S, Lengeler JW (1991) Molecular analysis of two fructokinases involved in sucrose metabolism of enteric bacteria. Mol Microbiol 5:2913–2922. doi:10.1111/j.1365-2958.1991.tb01851.x
Myung S, Wang Y, Zhang YHP (2010) Fructose-1,6-bisphosphatase from a hyper-thermophilic bacterium Thermotoga maritima: characterization, metabolite stability, and its implications. Process Biochem 45:1882–1887. doi:10.1016/j.procbio.2010.03.017
Rittmann D, Schaffer S, Wendisch VF, Sahm H (2003) Fructose-1,6-bisphosphatase from Corynebacterium glutamicum: expression and deletion of the fbp gene and biochemical characterization of the enzyme. Arch Microbiol 180:285–292. doi:10.1007/s00203-003-0588-6
Schaftingen EV, Hers HG (1981) Inhibition of fructose-1,6-bisphosphatase by fructose 2,6-biphosphate. Proc Natl Acad Sci USA 78(5):2861–2863
Navas MA, Gancedo JM (1996) The regulatory characteristics of yeast fructose-1,6-bisphosphatase confer only a small selective advantage. J Bacteriol 178:1809–1812
Ohnishi J, Katahira R, Mitsuhashi S, Kakita S, Ikeda M (2005) A novel gnd mutation leading to increased l-lysine production in Corynebacterium glutamicum. FEMS Microbiol Lett 242:265–274. doi:10.1016/j.femsle.2004.11.014
Marx A, Eikmanns BJ, Sahm H, de Graaf AA, Eggeling L (1999) Response of the central metabolism in Corynebacterium glutamicum to the use of an NADH-dependent glutamate dehydrogenase. Metab Eng 1:35–48. doi:10.1006/mben.1998.0106
Xu DQ, Tan YZ, Huan XJ, Hu XQ, Wang XY (2010) Construction of a novel shuttle vector for use in Brevibacterium flavum, an industrial amino acid producer. J Microbiol Methods 80:86–92. doi:10.1016/j.mimet.2009.11.003
Acknowledgments
This work was financially supported by the Program of Chinese 863 National High-Tech Research and Development Plan Project (No. 2008AA02Z212).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xu, J., Zhang, J., Guo, Y. et al. Improvement of cell growth and l-lysine production by genetically modified Corynebacterium glutamicum during growth on molasses. J Ind Microbiol Biotechnol 40, 1423–1432 (2013). https://doi.org/10.1007/s10295-013-1329-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-013-1329-8