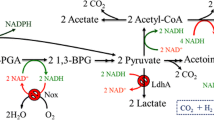
Abstract
Acetoin, a major extracellular catabolic product of Bacillus subtilis cultured on glucose, is widely used to add flavor to food and also serves as a precursor for chemical synthesis. The biosynthesis of acetoin from pyruvate requires the enzymes α-acetolactate synthase (ALS) and α-acetolactate decarboxylase (ALDC), both of which are encoded by the alsSD operon. The transcriptional regulator ALsR is essential for the expression of alsSD. Here we focused on enhancing the production of acetoin by B. subtilis using different promoters to express ALsR. The expression of reporter genes was much higher under the control of the HpaII promoter than under control of the P bdhA promoter. Although the HpaII promoter highly enhanced transcription of the alsSD operon through overexpression of ALsR, the production of acetoin was not significantly increased. In contrast, moderate enhancement of ALsR expression using the P bdhA promoter significantly improved acetoin production. Compared with the wild-type, the enzyme activities of ALS and ALDC in B. subtilis harboring P bdhA were increased by approximately twofold, and the molar yield of acetoin from glucose was improved by 62.9 % in shake flask fermentation. In a 5-L fermentor, the engineered B. subtilis ultimately yielded 41.5 g/L of acetoin. Based on these results, we conclude that enhanced expression of ALDC and ALS by moderately elevated expression of the transcriptional regulator ALsR could increase acetoin production in recombinant B. subtilis.






Similar content being viewed by others
Abbreviations
- ALDC:
-
α-Acetolactate decarboxylase
- ALS:
-
α-Acetolactate synthase
- AlsR:
-
Transcriptional regulator
- AR/BDH:
-
Acetoin reductase/2,3-butanediol dehydrogenase
- CAT:
-
Chloramphenicol acetyltransferase
- GFP:
-
Green fluorescent protein
- GRAS:
-
Generally recognized as safe
References
Atsumi S, Li Z, Liao JC (2009) Acetolactate synthase from Bacillus subtilis serves as a 2-ketoisovalerate decarboxylase for isobutanol biosynthesis in Escherichia coli. Appl Environ Microbiol 75(19):6306–6311
Biswas R, Yamaoka M, Nakayama H, Kondo T, Yoshida K, Bisaria VS, Kondo A (2012) Enhanced production of 2,3-butanediol by engineered Bacillus subtilis. Appl Microbiol Biotechnol 94(3):651–658. doi:10.1007/s00253-011-3774-5
Cañas A, Owens JD (1999) Acetoin production in growing Leuconostoc mesenteroides. World J Microbiol Biotechnol 15(3):339–344
Cogan TM, O’Dowd M, Mellerick D (1981) Effects of pH and sugar on acetoin production from citrate by Leuconostoc lactis. Appl Environ Microbiol 41(1):1–8
Collier PJ, Hall TJ, Iggo JA, Johnston P, Slipszenko JA, Wells PB, Whyman R (1998) Solvent and substituent effects on the sense of the enantioselective hydrogenation of pyruvate esters catalysed by Pd and Pt in colloidal and supported forms. Chem Commun 14:1451–1452
Dartois V, Coppee JY, Colson C, Baulard A (1994) Genetic analysis and overexpression of lipolytic activity in Bacillus subtilis. Appl Environ Microbiol 60(5):1670–1673
Dettwiler B, Dunn IJ, Heinzle E, Prenosil JE (1993) A simulation model for the continuous production of acetoin and butanediol using Bacillus subtilis with integrated pervaporation separation. Biotechnol Bioeng 41(8):791–800
Dugar D, Stephanopoulos G (2011) Relative potential of biosynthetic pathways for biofuels and bio-based products. Nat Biotechnol 29(12):1074–1078
Fradrich C, March A, Fiege K, Hartmann A, Jahn D, Hartig E (2012) The transcription factor AlsR binds and regulates the promoter of the alsSD operon responsible for acetoin formation in Bacillus subtilis. J Bacteriol 194(5):1100–1112
Gaspar P, Neves AR, Gasson MJ, Shearman CA, Santos H (2011) High yields of 2,3-butanediol and mannitol in Lactococcus lactis through engineering of NAD(+) cofactor recycling. Appl Environ Microbiol 77(19):6826–6835
Haavik HI (1974) Studies on the formation of bacitracin by Bacillus licheniformis: role of catabolite repression and organic acids. J Gen Microbiol 84(2):321–326
Hilmi A, Belgsir EM, Léger JM, Lamy C (1997) Electrocatalytic oxidation of aliphatic diols Part V. Electro-oxidation of butanediols on platinum based electrodes. J Electroanal Chem 435(1–2):69–75
Holtzclaw WD, Chapman LF (1975) Degradative acetolactate synthase of Bacillus subtilis: purification and properties. J Bacteriol 121(3):917–922
Ji XJ, Xia ZF, Fu NH, Nie ZK, Shen MQ, Tian QQ, Huang H (2013) Cofactor engineering through heterologous expression of an NADH oxidase and its impact on metabolic flux redistribution in Klebsiella pneumoniae. Biotechnol Biofuels 6(1):7
Kaneko T, Takahashi M, Suzuki H (1990) Acetoin fermentation by citrate-positive Lactococcus lactis subsp. lactis 3022 grown aerobically in the presence of hemin or Cu2+. Appl Environ Microbiol 56(9):2644–2649
Martin MG, Magni C, de Mendoza D, Lopez P (2005) CitI, a transcription factor involved in regulation of citrate metabolism in lactic acid bacteria. J Bacteriol 187(15):5146–5155
McFall SM, Montville TJ (1989) pH-mediated regulation of pyruvate catabolism in Lactobacillus plantarum chemostat cultures. J Ind Microbiol Biotechnol 4(5):335–340
Nicholson WL (2008) The Bacillus subtilis ydjL (bdhA) gene encodes acetoin reductase/2,3-butanediol dehydrogenase. Appl Environ Microbiol 74(22):6832–6838
Qin JY, Xiao ZJ, Ma CQ, Xie NZ, Liu PH, Xu P (2006) Production of 2,3-butanediol by Klebsiella pneumoniae using glucose and ammonium phosphate. Chin J Chem Eng 14(1):132–136
Renna MC, Najimudin N, Winik LR, Zahler SA (1993) Regulation of the Bacillus subtilis alsS, alsD, and alsR genes involved in post-exponential-phase production of acetoin. J Bacteriol 175(12):3863–3875
Rydstrom J (1979) Assay of nicotinamide nucleotide transhydrogenases in mammalian, bacterial, and reconstituted systems. Methods Enzymol 55:261–275
Shaw WV (1975) Chloramphenicol acetyltransferase from chloramphenicol-resistant bacteria. Methods Enzymol 43:737–755
Starrenburg MJ, Hugenholtz J (1991) Citrate Fermentation by Lactococcus and Leuconostoc spp. Appl Environ Microbiol 57(12):3535–3540
Sun JA, Zhang LY, Rao B, Shen YL, Wei DZ (2012) Enhanced acetoin production by Serratia marcescens H32 with expression of a water-forming NADH oxidase. Bioresour Technol 119:94–98. doi:10.1016/j.biortech.2012.05.108
Sun JN, Zhang LY, Rao B, Han YB, Chu J, Zhu JW, Shen YL, Wei DZ (2012) Enhanced acetoin production by Serratia marcescens H32 using statistical optimization and a two-stage agitation speed control strategy. Biotechnol Bioprocess Eng 17(3):598–605
Xiao Z, Xu P (2007) Acetoin metabolism in bacteria. Crit Rev Microbiol 33(2):127–140
Xiao ZJ, Liu PH, Qin JY, Xu P (2007) Statistical optimization of medium components for enhanced acetoin production from molasses and soybean meal hydrolysate. Appl Microbiol Biotechnol 74(1):61–68
Xu H, Jia SR, Liu JJ (2011) Development of a mutant strain of Bacillus subtilis showing enhanced production of acetoin. Afr J Biotechnol 10(5):779–788
Yang SJ, Dunman PM, Projan SJ, Bayles KW (2006) Characterization of the Staphylococcus aureus CidR regulon: elucidation of a novel role for acetoin metabolism in cell death and lysis. Mol Microbiol 60(2):458–468
Yu EK, Saddler JN (1983) Fed-batch approach to production of 2,3-butanediol by Klebsiella pneumoniae grown on high substrate concentrations. Appl Environ Microbiol 46(3):630–635
Zhang L, Chen S, Xie H, Tian Y, Hu K (2012) Efficient acetoin production by optimization of medium components and oxygen supply control using a newly isolated Paenibacillus polymyxa CS107. J Chem Technol Biotechnol 87:1551–1557
Zhang X, Yang TW, Lin Q, Xu MJ, Xia HF, Xu ZH, Li HZ, Rao ZM (2011) Isolation and identification of an acetoin high production bacterium that can reverse transform 2,3-butanediol to acetoin at the decline phase of fermentation. World J Microbiol Biotechnol 27(12):2785–2790
Acknowledgments
This work was supported by the Program for New Century Excellent Talents in University (NCET-10-0459), the National Basic Research Program of China (973 Program) (2012CB725202), the National Natural Science Foundation of China (30970056, 21276110), the Research Fund for the Doctoral Program of Higher Education of China (20110093120001), the Fundamental Research Funds for the Central Universities (JUSRP51306A and JUSRP21121), the 111 Project (111-2-06), and a Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institution.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhang, X., Zhang, R., Bao, T. et al. Moderate expression of the transcriptional regulator ALsR enhances acetoin production by Bacillus subtilis . J Ind Microbiol Biotechnol 40, 1067–1076 (2013). https://doi.org/10.1007/s10295-013-1303-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-013-1303-5