Abstract
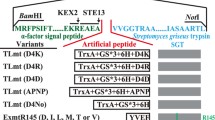
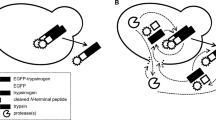
In the present study, the genes encoding trypsinogen and active trypsin from Streptomyces griseus were both cloned and expressed in the methylotrophic yeast Pichia pastoris with the α-factor secretion signal under the control of the alcohol oxidase promoter. The mature trypsin was successfully accumulated extracellularly in soluble form with a maximum amidase activity of 6.6 U ml−1 (batch cultivation with flask cultivation) or 14.4 U ml−1 (fed-batch cultivation with a 3-l fermentor). In contrast, the recombinant trypsinogen formed inclusion bodies and no activity was detected. Replacement of the trypsin propeptide Ala-Pro-Asn-Pro confirmed that its physiological function was as a repressor of activity. More importantly, our results proved that the propeptide inhibited the activity of trypsinogen after its successful folding.







Similar content being viewed by others
References
Bergmeyer HU, Gawehn K (1974) In: Bergmeyer HU (ed) Methods of enzymatic analysis, 2nd edn, vol 1. Academic, New York, pp 1013–1018
Cereghino JL, Cregg JM (2000) Heterologous protein expression in the methylotrophic yeast Pichia pastoris. FEMS Microbiol Rev 24:45–66
Chi WJ, Song JH, Oh EA, Park SW, Chang YK, Kim ES, Hong SK (2009) Medium optimization and application of an affinity column chromatography for Streptomyces griseus trypsin production from the recombinant Streptomyces griseus. J Microbiol Biotechnol 19:1191–1196
Choi SS, Chi WJ, Lee JH, Kang SS, Jeong BC, Hong SK (2001) Overexpression of the sprD gene encoding Streptomyces griseus protease D stimulates actinorhodin production in Streptomyces lividans. J Microbiol 39:305–313
Corey DR, Shiau AK, Yang Q, Janowski BA, Craik CS (1993) Trypsin display on the surface of bacteriophage. Gene 128:129–134
Craik CS, Largman C, Fletcher T, Roczniak S, Barr PJ, Fletterick R, Rutter WJ (1985) Redesigning trypsin: alteration of substrate specificity. Science 228:291–297
Cregg JM, Cereghino JL, Shi J, Higgins DR (2000) Recombinant protein expression in Pichia pastoris. Mol Biotechnol 16:23–52
Daly R, Hearn MT (2005) Expression of heterologous proteins in Pichia pastoris: a useful experimental tool in protein engineering and production. J Mol Recognit 18:119–138
Erlanger BF, Kokowsky N, Cohen W (1961) The preparation and properties of two new chromogenic substrates of trypsin. Arch Biochem Biophys 95:271–278
Ghosalkar A, Sahai V, Srivastava A (2008) Optimization of chemically defined medium for recombinant Pichia pastoris for biomass production. Bioresour Technol 99:7906–7910
Hanquier J, Sorlet Y, Desplancq D, Baroche L, Ebtinger M, Lefevre JF, Pattus F, Hershberger CL, Vertes AA (2003) A single mutation in the activation site of bovine trypsinogen enhances its accumulation in the fermentation broth of the yeast Pichia pastoris. Appl Environ Microbiol 69:1108–1113
Hartner FS, Glieder A (2006) Regulation of methanol utilisation pathway genes in yeasts. Microb Cell Fact 5:39
Hohenblum H, Vorauer-Uhl K, Katinger H, Mattanovich D (2004) Bacterial expression and refolding of human trypsinogen. J Biotechnol 109:3–11
Horinouchi S (2002) A microbial hormone, A-factor, as a master switch for morphological differentiation and secondary metabolism in Streptomyces griseus. Front Biosci 7:2045–2057
Jungo C, Rerat C, Marison IW, von Stockar U (2006) Quantitative characterization of the regulation of the synthesis of alcohol oxidase and of the expression of recombinant avidin in a Pichia pastoris Mut(+) strain. Enzyme Microb Tech 39:936–944
Kato JY, Chi WJ, Ohnishi Y, Hong SK, Horinouchi S (2005) Transcriptional control by A-factor of two trypsin genes in Streptomyces griseus. J Bacteriol 187:286–295
Kim JC, Cha SH, Jeong ST, Oh SK, Byun SM (1991) Molecular cloning and nucleotide sequence of Streptomyces griseus trypsin gene. Biochem Biophys Res Commun 181:707–713
Kim NS, Yu HY, Chung ND, Shin YJ, Kwon TH, Yang MS (2011) Production of functional recombinant bovine trypsin in transgenic rice cell suspension cultures. Protein Expr Purif 76:121–126
Kossiakoff AA, Chambers JL, Kay LM, Stroud RM (1977) Structure of bovine trypsinogen at 1.9 Å resolution. Biochemistry 16:654–664
Macouzet M, Simpson BK, Lee BH (2005) Expression of a cold-adapted fish trypsin in Pichia pastoris. FEMS Yeast Res 5:851–857
Niles AL, Maffitt M, Haak-Frendscho M, Wheeless CJ, Johnson DA (1998) Recombinant human mast cell tryptase beta: stable expression in Pichia pastoris and purification of fully active enzyme. Biotechnol Appl Biochem 28:125–131
Oh EA, Kim MS, Chi WJ, Kim JH, Hong SK (2007) Characterization of the sgtR1 and sgtR2 genes and their role in regulating expression of the sprT gene encoding Streptomyces griseus trypsin. FEMS Microbiol Lett 276:75–82
Ohnishi Y, Ishikawa J, Hara H, Suzuki H, Ikenoya M, Ikeda H, Yamashita A, Hattori M, Horinouchi S (2008) Genome sequence of the streptomycin-producing microorganism Streptomyces griseus IFO 13350. J Bacteriol 190:4050–4060
Olafson RW, Jurasek L, Carpenter MR, Smillie LB (1975) Amino acid sequence of Streptomyces griseus trypsin. Cyanogen bromide fragments and complete sequence. Biochemistry 14:1168–1177
Olafson RW, Smillie LB (1975) Enzymic and physicochemical properties of Streptomyces griseus trypsin. Biochemistry 14:1161–1167
Read RJ, James MN (1988) Refined crystal structure of Streptomyces griseus trypsin at 1.7 Å resolution. J Mol Biol 200:523–551
Salamin K, Sriranganadane D, Lechenne B, Jousson O, Monod M (2010) Secretion of an endogenous subtilisin by Pichia pastoris strains GS115 and KM71. Appl Environ Microbiol 76:4269–4276
Sidhu SS, Kalmar GB, Willis LG, Borgford TJ (1995) Protease evolution in Streptomyces griseus. Discovery of a novel dimeric enzymes. J Biol Chem 270:7594–7600
Sinha J, Plantz BA, Inan M, Meagher MM (2005) Causes of proteolytic degradation of secreted recombinant proteins produced in methylotrophic yeast Pichia pastoris: case study with recombinant ovine interferon-tau. Biotechnol Bioeng 89:102–112
Stroud RM, Kossiakoff AA, Chambers JL (1977) Mechanisms of zymogen activation. Annu Rev Biophys Bioeng 6:177–193
Trop M, Birk Y (1968) The trypsin-like enzyme from Streptomyces griseus (pronase). Biochem J 109:475–476
Trop M, Birk Y (1970) The specificity of proteinases from Streptomyces griseus (pronase). Biochem J 116:19–25
Vasquez JR, Evnin LB, Higaki JN, Craik CS (1989) An expression system for trypsin. J Cell Biochem 39:265–276
Villatte F, Hussein AS, Bachmann TT, Schmid RD (2001) Expression level of heterologous proteins in Pichia pastoris is influenced by flask design. Appl Microbiol Biot 55:463–465
Walsh KA (1970) Trypsinogens and trypsins of various species. In: Perlmann G, Lorand L (eds) Methods of enzymology. Academic, New York, pp 41–63
Wang Z, Wang Y, Zhang D, Li J, Hua Z, Du G, Chen J (2010) Enhancement of cell viability and alkaline polygalacturonate lyase production by sorbitol co-feeding with methanol in Pichia pastoris fermentation. Bioresour Technol 101:1318–1323
Yao LY, Man CX, Zhao F, Qu XG, Han XY, Sun DQ, Jiang YJ (2010) Expression of bovine trypsin in Lactococcus lactis. Int Dairy J 20:806–809
Yee L, Blanch HW (1993) Recombinant trypsin production in high cell density fed-batch cultures in Escherichia coli. Biotechnol Bioeng 41:781–790
Acknowledgments
We thank Professor Byong Lee at Jiangnan University for his discussion and revision. We also thank Professor Jian Jin and Ph.D. candidate Ying Hou at the School of Medicine and Pharmaceutics of Jiangnan University for their support with the mammalian cell digestions. This work was financially supported by the National High Technology Research and Development Program of China (863 Program, 2011AA100905), Program for Changjiang Scholars and Innovative Research Team in University (No. IRT1135), and the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ling, Z., Ma, T., Li, J. et al. Functional expression of trypsin from Streptomyces griseus by Pichia pastoris . J Ind Microbiol Biotechnol 39, 1651–1662 (2012). https://doi.org/10.1007/s10295-012-1172-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-012-1172-3