Abstract
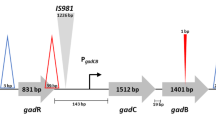
γ-aminobutyric acid (GABA) is generated from glutamate by the action of glutamic acid decarboxylase (GAD) and characterized by hypotensive, diuretic, and tranquilizing effects in humans and animals. The production of GABA by lactic acid starter bacteria would enhance the functionality of fermented dairy foods including cheeses and yogurt. The survey of 42 strains of the yogurt starter culture Streptococcus thermophilus by PCR techniques indicated the presence of a glutamate decarboxylase gene (gadB) in 16 strains. DNA sequencing data indicated that the GAD/GABA antiporter locus (gadB/gadC) in GAD+ S. thermophilus strains is flanked by transposase elements (5′ and 3′) and positioned between the luxS (5′) and the HD-superfamily hydrolase genes (3′). The PCR amplification product of a ca. 2-kb genomic fragment that included the gadB and its putative promoter region was inserted into a shuttle vector, which was used to transform Escherichia coli DH5α. Subsequently, the recombinant plasmid pMEU5a-1/gadB (7.24 kb) was electrotransformed into the GAD-negative strain S. thermophilus ST128. The ST128 transformants carrying the plasmid-encoded gadB produced functional GAD enzyme as evidenced by the conversion of glutamate to GABA at a rate similar to strains with the gadB/gadC operon located on the chromosome. The results demonstrated the potential to impart to non-GABA-producing strains of S. thermophilus and other lactic acid bacteria the GAD+ phenotype that improves their appeal in possible applications in the development of health-promoting functional foods.



Similar content being viewed by others
References
Adeghate E, Ponery AS (2004) GABA in the endocrine pancreas: cellular localization and function in normal and diabetic rats. Tissue Cell 34:1–6
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Coderre PE, Somkuti GA (1999) Cloning and expression of the pediocin operon in Streptococcus thermophilus and other lactic fermentation bacteria. Curr Microbiol 39:295–301
Cotter PD, Hill C (2003) Surviving the acid test: responses of Gram-positive bacteria to low pH. Microbiol Mol Biol Rev 67:429–453
Gilbreth SE, Somkuti GA (2005) Thermophilin 110: a bacteriocin of Streptococcus thermophilus ST110. Curr Microbiol 51:175–182
Hagiwara H, Seki T, Ariga T (2004) The effect of pre-germinated brown rice intake on blood glucose and PAI-1 levels in streptozotocin-induced diabetic rats. Biosci Biotechnol Biochem 68:444–447
Higuchi T, Hayashi H, Abe K (1997) Exchange of glutamate and γ-aminobutyrate in a Lactobacillus strain. J Bacteriol 179:3362–3364
Hiraga K, Ueno Y, Oda K (2008) Glutamate decarboxylase from Lactobacillus brevis: activation by ammonium sulfate. Biosci Biotechnol Biochem 72:1299–1306
Huang J, Mei LH, Wu H, Liu DQ (2007) Biosynthesis of γ-aminobutyric acid (GABA) using immobilized whole cells of Lactobacillus brevis. World J Microbiol Biotechnol 23:865–871
Inoue K, Shirai T, Ochiai H, Kasao M, Hayakawa K, Kimura M, Sansawa H (2003) Blood-pressure lowering effect of a novel fermented milk containing gamma-aminobutyric acid (GABA) in mild hypertensives. Eur J Clin Nutr 57:490–495
Jakobs C, Jaeken J, Gibson KM (1993) Inherited disorders of GABA metabolism. J Inher Metab Dis 16:704–715
Komatsuzaki N, Shima J, Kawamoto S, Momose H, Kimura T (2005) Production of γ-aminobutyric acid (GABA) by Lactobacillus paracasei isolated from traditional fermented foods. Food Microbiol 22:497–504
Komatsuzaki N, Nakamura T, Kimura T, Shima J (2008) Characterization of glutamate decarboxylase from a high γ-aminobutyric acid (GABA)-producer, Lactobacillus paracasei. Biosci Biotechnol Biochem 72:278–285
Li H, Cao Y (2010) Lactic acid bacterial cell factories for gamma-aminobutyric acid. Amino Acids 39:1107–1116
Li H, Qui T, Gao D, Cao Y (2010) Medium optimization for production of gamma-aminobutyric acid by Lactobacillus brevis NCL912. Amino Acids 38:1439–1445
Lin Q, Yang S, Lu F, Bie X, Jiao Y, Zou X (2009) Cloning and expression of glutamate decarboxylase gene from Streptococcus thermophilus Y2. J Gen Appl Microbiol 55:305–310
Nomura M, Nakajima I, Fujita Y, Kobayashi M, Kimoto H, Suzuki I, Aso H (1999) Lactococcus lactis contains only one glutamate decarboxylase gene. Microbiology 145:1375–1380
Park KB, Oh SH (2007) Cloning, sequencing and expression of a novel glutamate decarboxylase gene from a newly isolated lactic acid bacterium, Lactobacillus brevis OPK-3. Bioresour Technol 98(312):319
Renye JA, Somkuti GA (2008) Cloning of milk-derived bioactive peptides in Streptococcus thermophilus. Biotechnol Lett 30:723–730
Sanders JW, Leehouts K, Burghoom J, Brands J, Venema G, Kok J (1998) A chloride-inducible acid resistance mechanism in Lactococcus lactis and its regulation. Mol Microbiol 27:299–310
Schuller H, Al-Wadei HAN, Majidi M (2008) Gamma-aminobutyric acid, a potential tumor suppressor for small airway-derived lung adenocarcinoma. Carcinogenesis 29:1979–1985
Small PC, Waterman SR (1998) Acid stress, anaerobiosis and gadCB: lessons from Lactococcus lactis and Escherichia coli. Trends Microbiol 6:214–216
Solaiman DKY, Somkuti GA (1993) Shuttle vectors developed from Streptococcus thermophilus native plasmid. Plasmid 30:67–78
Somkuti GA, Steinberg DH (1988) Genetic transformation of Streptococcus thermophilus by electroporation. Biochemie 70:579–585
Somkuti GA, Dominiecki ME, Steinberg DH (1998) Permeabilization of Streptococcus thermophilus and Lactobacillus delbrueckii subsp. bulgaricus with ethanol. Curr Microbiol 36:202–206
Somkuti GA, Solaiman DKY (1997) STP2201, a chromosomal promoter sequence of Streptococcus thermophilus. Curr Microbiol 35:180–185
Stougaard P, Molin S (1981) Vertical dye-buoyant density gradients for rapid analysis and preparation of plasmid DNA. Anal Biochem 118:191–193
Sun Z, Chen X, Wang J, Zhao W, Shao Y, Wu L, Zhou Z, Sun T, Wang L, Meng L, Zhang H, Chen W (2011) Complete genome sequence of Streptococcus thermophilus strain ND03. J Bacteriol 193:793–794
Tujioka K, Ohshumi M, Horie K, Kim M, Hayase K, Yokogoshi H (2009) Dietary γ-aminobutyric acid affects the brain protein synthesis rate in ovariectomized female rats. J Nutr Sci Vitaminol 55:75–80
Ueno H (2000) Enzymatic and structural aspects of glutamate decarboxylase. J Mol Catal B Enzym 10:67–79
Yang SY, Yu B, Lu ZX, Bie XM, Lin Q, Sun LJ (2008) Optimization of culture conditions for production of glutamate decarboxylase by Streptococcus salivarius ssp. thermophilus. J Chem Technol Biotechnol 83:389–392
Yang SY, Lu EX, Lu ZX, Bie XM, Jiao Y, Sun LJ, Wu B (2008) Production of γ-aminobutyric acid by Streptococcus thermophilus subsp. thermophilus Y2 under submerged fermentation. Amino Acids 34:473–478
Zhang G, Brown AW (1997) The rapid determination of γ-aminobutyric acid. Phytochemistry 44:1007–1009
Acknowledgments
We thank Jaileene Hernandez for providing technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Rights and permissions
About this article
Cite this article
Somkuti, G.A., Renye, J.A. & Steinberg, D.H. Molecular analysis of the glutamate decarboxylase locus in Streptococcus thermophilus ST110. J Ind Microbiol Biotechnol 39, 957–963 (2012). https://doi.org/10.1007/s10295-012-1114-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-012-1114-0