Abstract
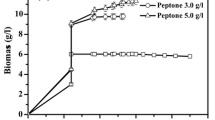
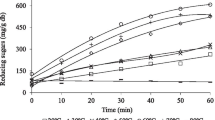
The constant-rate fed-batch production of the polygalacturonic acid bioflocculant REA-11 was studied. A controlled sucrose-feeding strategy resulted in a slight improvement in biomass and a 7% reduction in flocculating activity compared with the batch process. When fed with a 3 g l−1 urea solution, the flocculating activity was enhanced to 720 U ml−1 in 36 h. High cell density (2.12 g l−1) and flocculating activity (820 U ml−1) were obtained in a 10-l fermentor by feeding with a sucrose-urea solution, with values of nearly two times and 50% higher than those of the batch process, respectively. Moreover, the residual sucrose declined to 2.4 g l−1, and residual urea decreased to 0.03 g l−1. Even higher flocculating activity of 920 U ml−1 and biomass of 3.26 g l−1 were obtained by feeding with a sucrose-urea solution in a pilot scale fermentation process, indicating the potential industrial utility of this constant-rate feeding strategy in bioflocculant production by Corynebacterium glutamicum.








Similar content being viewed by others
Abbreviations
- μ:
-
Specific cell growth rate
- qs :
-
Specific sucrose consumption rate
- qp :
-
Specific bioflocculant production rate
- GLC:
-
Glucose
- G6p:
-
Glucose-6-phosphate
- G1P:
-
Glucose-1-phosphate
- UDPG:
-
UDP-glucose
- UDP-GAL:
-
UDP-galactose
- UDP-GALA:
-
UDP-galacturonic acid
- polyGALA:
-
Polygalacturonic acid
- PEP:
-
Phosphoenolpyruvate
- PYR:
-
Pyruvic acid
- AcCoA:
-
AcetylCoA
- ICI:
-
Isocitric acid
- OAA:
-
Oxalacetic acid
- α-KG:
-
α-ketoglutaric acid
- SUC:
-
Succinic acid
- MAL:
-
Malic acid
- LAC:
-
Lactic acid
- AC:
-
Acetic acid
References
Cooney CL, Wang DIC, Mateles RI (1968) Measurement of heat evolution and correlation with oxygen consumption during microbial growth. Biotechnol Bioeng 11:269–281
Deng SB, Bai RB, Hu XM, Luo Q (2003) Characteristics of a bioflocculant produced by Bacillus mucilaginosus and its use in starch wastewater. J Colloid Interface Sci 260:265–272
Fujita M, Ike M, Jang JH, Kim SM, Hirao T (2001) Bioflocculant production from lower-molecular fatty acids as a novel strategy for utilization of sludge digestion liquor. Water Sci Technol 44:237–243
Fujita M, Ike M, Tachibana S, Kitada G, Kim SM, Inoue Z (2000) Characterization of a bioflocculant produced by Citrobacter TKF04 from acetic and propionic acids. J Biosci Bioeng 89:40–46
He N, Du GC, Li Y, Chen J (2001) Flocculating conditions of bioflocculant. J Wuxi U Light Ind 20:248–251
He N, Li Y, Chen J (2004) Production of a novel polygalacturonic acid bioflocculant REA-11 by Corynebacterium glutamicum. Biores Technol 94:99–105
He N, Li Y, Chen J (2005) Foundation of metabolic models and metabolic flux analysis on the production of a novel bioflocculant REA-11. J Chem Industry Eng 56:681–688
He N, Li Y, Chen J, Lun SY (2002) Identification of a novel bioflocculant from a newly isolated Corynebacterium glutamicum. Biochem Eng J 11:137–148
He N, Wu XJ, Deng X, Lu YH, Li QB (2004) Two-stage pH control mode in batch fermentation of a novel bioflocculant from Corynebacterium glutamicum. Chem Res Chinese U 20:152–155
Hermann T (2003) Industrial production of amino acids by coryneform bacteria. J Biotechnol 104:155–172
Ikeda M, Katsumata R (1999) Hyperproduction of tryptophan by Corynebacterium glutamicum with the modified pentose phosphate pathway. Appl Environment Microbiol 65:2497–2502
Roe Joseph H (1934) A colorimetric method for the determination of fructose in blood and urine. J Biol Chem 107:15–22
Kim LS, Hong SJ, Son MK, Lee YH (2006) Polymeric and compositional properties of novel extracellular microbial polyglucosamine biopolymer from new strain of Citrobacter sp BL-4. Biotechnol Lett 28:241–245
Kurane R, Hatamochi K, Kakuno T, Kiyohara M, Hirano M, Taniguchi Y (1994) Production of a bioflocculant by Rhodococcus erythropolis S-1 grown on alcohols. Biosci Biotechnol Biochem 58:428–429
Kurane R, Nohata Y (1991) Microbial flocculation of waste liquids and oil emulsion by a bioflocclant from Alcaligenes latus. Agric Biol Chem 55:1127–1129
Kwon GS, Moon SH, Hong SD, Lee HM, Kim HS, Oh HM, Yoon BD (1996) A novel flocculant biopolymer produced by Pestalotiopsis sp. KCTC 8637p. Biotechnol Lett 18:1459–1464
Levy N, Magdasi S, Bar-Or Y (1992) Physico-chemical aspects in flocculation of bentonite suspensions by a cyanobacterial. Water Res 262:49–54
Li M, Zheng CL, Zhang Y (2005) Determination of carbamide in waste stripping liquid by colorimetry method of paradimethylaminobezaldehyde. Technol Development Chem Ind 34:40–43
Li Y, He N, Guan HQ, Du GC, Chen J (2003) A novel polygalacturonic acid bioflocculant REA-11 produced by Corynebacterium glutamicum: a proposed biosynthetic pathway and experimental confirmation. Appl Microbiol Biotechnol 63:200–206
Luo P, Luo GY, Ji FY (2005) Characterization of an extracellular polysaccharide produced by Bacillus sp. RL-2. J Chongqing U 4:33–37 (English version)
Meng L (2005) Studies on the flocculating activity and fermentation conditions of complex microbial flocculant. Dissertation, Haerbin Industry University
Salehizadeh H, Shojaosadati SA (2001) Extracellular biopolymeric flocculants: recent trends and biotechnological importance. Biotechnol Adv 19:371–385
Sassi AH, Fauvart L, Deschamps AM, Lebeault JM (1998) Fed-batch production of L-lysine by Corynebacterium glutamicum. Biochem Eng J 1:85–90
Takeda M, Kurane R, Nakamura I (1991) Localization of a biopolymer produced by Rhodococcus erythropolis grown on n-pentadecane. Agric Biol Chem 55:2665–2666
Takagi H, Kadowaki K (1985) Purification and chemical properties of a flocculant produced by Paecilomyces. Agric Biol Chem 49:3159–3164
Tieda K, Kurane R (1991) Microbial flocculant from Alcaligenes cupidus KT201. Agric Biol Chem 55:2793–2799
WeusterBotz D, Kelle R, Frantzen M, Wandrey C (1997) Substrate controlled fed-batch production of L-lysine with Corynebacterium glutamicum. Biotechnol Prog 13:387–393
Yamane T, Shmizu S (1984) Fed-batch techniques in microbial processes. Adv Biochem Eng Biotechnol 30:148–194
Zhang ZQ, Lin B, Xia SQ, Wang XJ, Yang AM (2007) Production and application of a novel bioflocculant by multiple-microorganism consortia using brewery wastewater as carbon source. J Environment Sci 19:667–673
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (no. 30700020), the Xiamen Science and Technology Committee (no. 3502Z20093006), and Fujian Provincial Department of Science and Technology, China (key project no. 2008Y0060).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, H., Li, Q., Lu, R. et al. Fed-batch production of a bioflocculant from Corynebacterium glutamicum . J Ind Microbiol Biotechnol 37, 1203–1209 (2010). https://doi.org/10.1007/s10295-010-0767-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-010-0767-9