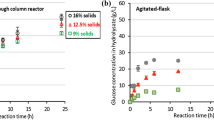
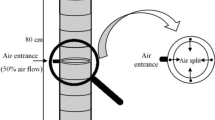
Abstract
Invertase from Aspergillus niger C28B25 was produced by solid-state fermentation (SSF). Fermented solids were used directly as a biocatalyst for batch and continuous hydrolysis of sucrose in a packed-bed reactor under different operational conditions with various temperatures, sucrose concentrations, and feed flow rates. The SSF allowed obtaining a biocatalyst with an invertase activity of 82.2 U/g db. The biocatalyst maintained its activity in the range of 40 to 70 °C for at least 70 h of continuous operation. In a 20-mL packed bed reactor, the highest hydrolysis rate (12.3 g/g db h) was obtained at 40 °C with 2 M sucrose. Continuous hydrolysis in 20-mL and 200-mL reactors at 60 °C led to sucrose hydrolysis above 60% (8.5 residence times) and above 55% (4.5 residence times), respectively. The auto-immobilised biocatalyst produced by SSF without recovery, purification, and immobilisation stages offers an economical alternative for developing accessible biocatalysts that can be applied in batch or continuous sucrose hydrolysis processes. This study shows the potential of biocatalyst production by SSF for other enzymatic systems.




Similar content being viewed by others

Data Availability
Not applicable.
References
Mansfeld, J., Schellenberger, A., & Römbach, J. (1992). Application of polystyrene-bound invertase to continuous sucrose hydrolysis. Biotechnology and Bioengineering, 40, 997–1003.
Cabrera, M. P., Assis, C. R. D., Neri, D. F. M., Pereira, C. F., Soria, F., & Carvalho, L. B. (2017). High sucrolytic activity by invertase immobilized onto magnetic diatomaceous earth nanoparticles. Biotechnology Reports, 14, 38–46.
Moreau, C., Durand, R., Aliès, F., Cotillon, M., Frutz, T., & Théoleyre, M. (2000). Hydrolysis of sucrose in the presence of H-forms zeolites. Industrial Crops and Products, 11, 237–242.
Nasef, M., Saidi, H., & Senna, M. (2005). Hydrolysis of sucrose by radiation grafted sulfonic acid membranes. Chemical Engineering Journal, 108, 13–17.
Krastanov, A. (1997). Continuous sucrose hydrolysis by yeast cells immobilized to wool. Applied Microbiology and Biotechnology, 47, 476–481.
Slavko, K., Chris, L., Estefanía, I. S., Błażej, K., Veli, A., & Marc, K. (2018). Synthesis of fructooligosaccharides (FosA) and inulin (InuO) by GH68 fructosyltransferases from Bacillus agaradhaerens strain WDG185. Carbohydrate Polymers, 179, 350–359.
Nooshin, M., Kamahldin, H., Omid, R., Seyed, S. A., & Sorour, H. (2017). Catalytic efficiency and thermostability improvement of Suc2 invertase through rational site-directed mutagenesis. Enzyme and Microbial Technology, 96, 14–22.
Lee, W. C., & Huang, C. T. (2000). Modelling of ethanol production using Zymomonasmobilis ATCC 10988 grown on the media containing glucose and fructose. Biochemical Engineering Journal, 4, 217–227.
Vargas, L., Pião, A., Domingos, R., & Carmona, E. (2004). Ultrasound effects on invertase from Aspergillus niger. World Journal of Microbiology and Biotechnology, 20, 137–142.
Patel, A. K., Singhania, R. R., & Pandey, A. (2016). Novel enzymatic processes applied to the food industry. Current Opinion in Food Science, 7, 64–72.
Dalagnol, L. M. G., Silveira, V. C. C., Baron, H., Manfroi, V., & Rodrigues, R. C. (2017). Improvement of pectinase, xylanase and cellulase activities by ultrasound: Effects on enzymes and substrates, kinetics and thermodynamic parameters. Process Biochemistry, 61, 80–87.
Martínez-Ruiz, A., Tovar-Castro, L., García, H. S., Saucedo-Castañeda, G., & Favela-Torres, E. (2018). Continuous ethyl oleate synthesis by lipases produced by solid-state fermentation by Rhizopus microspores. Bioresource Technology, 265, 52–58.
Soares, A., Duarte, P. E., Leite, B. R. C., Nogueira, C. A., Rufino, E. N., Ribeiro, F. A., Stringheta, P. C., & Mota, R. A. (2019). Ultrasound assisted enzymatic hydrolysis of sucrose catalyzed by invertase: Investigation on substrate, enzyme and kinetics parameters. LWT, 107, 164–170.
Hölker, U., Höfer, M., & Lenz, J. (2004). Biotechnological advantages of laboratory-scale solid-state fermentation with fungi. Applied Microbiology and Biotechnology, 64, 175–186.
Romero-Gómez, S. J., Augur, C., & Viniegra-González, G. (2000). Invertase production by Aspergillus niger in submerged and solid-state fermentation. Biotechnology Letters, 22, 1255–1258.
Montiel-González, A., Fernández, F., Viniegra-González, G., & Loera, O. (2002). Invertase production on solid-state fermentation by Aspergillus niger strains improved by parasexual recombination. Applied Biochemistry and Biotechnology, 102, 63–70.
Flores-Flores, T. C., Gutiérrez-Rojas, M., Revah, S., & Favela-Torres, E. (2011). Comparative study for oxygenases produced by Aspergillus niger ATCC 9642, in solid-state and submerged fermentation. Revista Mexicana de Ingeniería Quimica, 10, 189–207.
Miller, G. (1960). Measurement of carboxymethylcellulase activity. Analytical Biochemistry, 2, 127–132.
Manoochehri, H., Hosseini, N. F., Saidijam, M., Taheri, M., Rezaee, H., & Nouri, F. (2020). A review on invertase: Its potentials and applications. Biocatalysis and Agricultural Biotechnology, 25, 101599.
Dinarvand, M., Rezaee, M., & Foroughi, M. (2017). Optimizing culture conditions for production of intra and extracellular inulinase and invertase from Aspergillus niger ATCC 20611 by response surface methodology (RSM). Brazilian Journal of Microbiology, 48, 427–441.
Bhalla, T. C., Bansuli, T., Savitri, N., & Thakur, N. (2017). Invertase of Saccharomyces cerevisiae SAA-612: Production, characterization and application in synthesis of fructo-oligosaccharides. LWT, 77, 178–185.
Ghasemi, Y., Mohkam, M., Ghasemian, A., & Rasoul-Amini, S. (2014). Experimental design of medium optimization for invertase production by Pichia sp. Journal of Food Science and Technology, 51, 267–275.
Jafari, R., Naghavi, N. S., Khosravi-Darani, K., Doudi, M., & Shahanipour, K. (2020). Kombucha microbial starter with enhanced production of antioxidant compounds and invertase. Biocatalysis and Agricultural Biotechnology, 29, 101789.
Aydogan, M. N., Taskin, M., Canli, O., Arslan, N. P., & Ortucu, S. (2014). Tris–sucrose buffer system: A new specially designed medium for extracellular invertase production by immobilized cells of isolated yeast Cryptococcus laurentii MT-61. Folia Microbiologica, 59, 9–16.
de Almeida, M. N., Guimaraes, V. M., Falkoski, D. L., de Camargo, B. R., Fontes-Santana, G. C., Maitan-Alfenas, G. P., & de Rezende, S. T. (2018). Purification and characterization of an invertase and a transfructosylase from Aspergillus terreus. Journal of Food Biochemistry, 42, 12551.
Oyedeji, O., Bakare, M. K., Adewale, I. O., Olutiola, P. O., & Omoboye, O. O. (2017). Optimized production and characterization of thermostable invertase from Aspergillus niger IBK1, using pineapple peel as alternate substrate. Biocatalysis and Agricultural Biotechnology, 9, 218–223.
Taskin, M., Ortucu, S., Unver, Y., Tasar, O. C., Ozdemir, M., & Kaymak, H. C. (2016). Invertase production and molasses decolourization by cold-adapted filamentous fungus Cladosporiumherbarum ER-25 in non-sterile molasses medium. Process Safety Environmental Protection, 103, 136–143.
Prodanovic, R., Jovanovic, S., & Vujcic, Z. (2001). Immobilization of invertase on a new type of macroporous glycidyl methacrylate. Biotechnology Letters, 23, 1171–1174.
Szymanska, K., Pudlo, W., Mrowiec-Bialon, J., Czardybon, A., Kocurek, J., & Jarzebski, A. (2013). Immobilization of invertase on silica monoliths with hierarchical pore structure to obtain continuous flow enzymatic microreactors of high performance. Microporous and Mesoporous Materials, 170, 75–82.
Belcarz, A., Ginalska, G., Lobarzewskai, J., & Penel, C. (2002). The novel non-glycosylated invertase from Candida utilis (the properties and the conditions of production and purification). Biochimica et Biophysica Acta, 1594, 40–53.
De la Vega, M., Cejudo, F., & Paneque, A. (1991). Purification and properties of an extracellular invertase from Azotobacterchroococcum. Enzyme and Microbial Technology, 13, 267–271.
L’Hocine, L., Wang, Z., Jiang, B., & Xu, S. (2000). Purification and partial characterization of fructosyltransferase and invertase from Aspergillus niger AS0023. Journal of Biotechnology, 81, 73–84.
Santana, A., Costa, L., Mendes, A., Moraes, C. A., Gomes, M. A., & Perez, M. (2005). Sucrose hydrolysis catalyzed by auto-immobilized invertase into intact cell of Cladosporiumcladosporioides. Electronic Journal of Biotechnology, 8, 54–62.
Mukherjee, S., Basak, S., & Khowala, S. (2002). Regulation (Alteration) of activity and conformation of sucrase by coaggregation with cellobiose in culture of medium of Termitomycesclypeatus. Biotechnology Progress, 18, 404–408.
Buenrostro-Figueroa, J., Huerta-Ochoa, S., Prado-Barragan, A., Ascacio-Valdés, J., Sepúlveda, L., Rodríguez, R., Aguilera-Carbó, A., & Aguilar, C. (2014). Continuous production of ellagic acid in a packed-bed reactor. Process Biochemistry, 49, 1595–1600.
Acknowledgements
JAMR thanks the National Council of Science and Technology (CONACyT) Mexico for the scholarship granted to carry out postgraduate studies.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
The study was conceived, done, and written by AMR and EFT. All authors: AMR, LTC, CNA, GSC, and EFT participated in the preparation of the study, discussion, critical review of the draft of the manuscript, and revision of the final version of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
All authors gave their consent to participate in this research.
Consent for Publication
All authors gave their consent to submit this research for publication.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Martínez-Ruiz, A., Tovar-Castro, L., Aguilar, C.N. et al. Sucrose Hydrolysis in a Continuous Packed-Bed Reactor with Auto-immobilise Aspergillus niger Biocatalyst Obtained by Solid-State Fermentation. Appl Biochem Biotechnol 194, 1327–1339 (2022). https://doi.org/10.1007/s12010-021-03737-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-021-03737-z