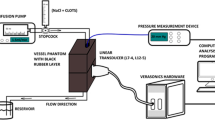
Abstract
Targeted drug delivery (TDD) to abdominal aortic aneurysm (AAA) using a controlled and efficient approach has recently been a significant challenge. In this study, by using magnetic microbubbles (MMBs) under a magnetic field, we investigated the MMBs performance in TDD to AAA based on the amount of surface density of MMBs (SDMM) adhered to the AAA lumen. The results showed that among the types of MMBs studied in the presence of the magnetic field, micromarkers are the best type of microbubble with a −\(50\%\) increase in SDMM adhered to the critical area of AAA. The results show that applying a magnetic field causes the amount of SDMM adhered to the whole area of AAA to increase −\(1.54\) times compared to the condition in which the magnetic field is absent. This optimal and maximum value occurs for Definity MMBs with − 3.3 μm diameter. Applying a magnetic field also increases the adhesion surface density by − \(1.27\), − \(1.14\), and −\(1.11\) times for the Micromarker, Optison, and Sonovue microbubbles, respectively, relative to the condition in which the magnetic field is absent. It was shown that using MBBs under magnetic field has the best performance in delivery to AAA for patients with negative inlet blood flow. Also, we have exposed that in an efficient TDD to AAA using MMBs, decreasing the density of MMBs increases drug delivery efficiency and performance. When density is − \(3500 {\text{Kg}}.{\text{m}}^{ - 3}\), there is the highest difference (about − 75%) between the SDMM adhered to AAA in the presence of a magnetic field and in the absence of a magnetic field.










Similar content being viewed by others
Data Availability
All data, models, or code generated or used during the study are available from the corresponding author upon reasonable request.
Abbreviations
- AAA:
-
Abdominal aortic aneurysm
- ALE:
-
Arbitrary Lagrange–Eulerian
- AMG:
-
Algebraic multigrid
- BDF:
-
Backward differentiation formula
- CT:
-
Computerized tomography
- DICOM:
-
Digital imaging and communications in medicine
- FSI:
-
Fluid–structure interaction
- GNF:
-
Generalized Newtonian fluid
- GMRES:
-
Generalized minimal residual
- MMBs:
-
Magnetic microbubbles
- MUMPS:
-
Multifrontal massively parallel sparse
- NPs:
-
Nanoparticles
- SDMM:
-
Surface density of magnetic microbubbles
- TDD:
-
Targeted drug delivery
- \({\varvec{A}}\) :
-
Magnetic vector potential (T.m)
- \(a\) :
-
Power law index
- \({\varvec{B}}\) :
-
Magnetic flux density (T)
- \({{\varvec{B}}}_{\mathrm{rem}}\) :
-
Remanent magnetic flux (T)
- \({C}_{10}\) :
-
Material constant (Pa)
- \({C}_{20}\) :
-
Material constant (Pa)
- \(d\) :
-
Parameter of non-residency of material
- \({d}_{p}\) :
-
Particle diameter (m)
- \({\varvec{E}}\) :
-
Electrical field (V.m−1)
- \({\varvec{F}}\) :
-
Volumetric force (N)
- \({F}^{s}\) :
-
Dislodge force parameter
- \({{\varvec{F}}}_{B}\) :
-
Brownian force (N)
- \({{\varvec{F}}}_{D}\) :
-
Drag force (N)
- \({{\varvec{F}}}_{G}\) :
-
Gravitational force (N)
- \({{\varvec{F}}}_{L}\) :
-
Lift force (N)
- \({{\varvec{F}}}_{\mathrm{mp}}\) :
-
Magnetophoretic force (N)
- \({\varvec{H}}\) :
-
Magnetic field intensity (A.m−1)
- \({h}_{0}\) :
-
Maximum distance of particle from the wall (m)
- \({I}_{1}\) :
-
First strain variable (Pa)
- \({\varvec{J}}\) :
-
Current density (A.m−2)
- \({{\varvec{J}}}_{\mathrm{e}}\) :
-
External current density (A.m−2)
- \({\varvec{J}}_{{{\text{el}}}}\) :
-
Elastic volume ratio (A.m−2)
- \({K}_{a}^{0}\) :
-
Continuity constant
- \({K}_{B}\) :
-
Boltzmann constant (m2.kg.s−2.k−1)
- \(l\) :
-
Separation distance (m)
- \({\varvec{M}}\) :
-
Magnetization vector (A.m−1)
- \({m}_{l}\) :
-
Surface density of ligand (m−2)
- \({m}_{m}\) :
-
Particle mass (Kg)
- \({{\varvec{m}}}_{\mathrm{eff}}\) :
-
Effective dipole moment (A.m2)
- \({m}_{r}\) :
-
Surface density of receptor (m−2)
- \(p\) :
-
Static pressure (Pa.s)
- \({P}_{a}\) :
-
Adhesion probability
- \({r}_{0}\) :
-
Radius of circular section (m)
- \(T\) :
-
Temperature (K)
- \({T}^{s}\) :
-
Dislodge torque parameter
- \({\varvec{u}}\) :
-
Fluid velocity vector (m.s−1)
- \({\varvec{v}}\) :
-
Particle velocity vector (m.s−1)
- \(V\) :
-
Volume (m3)
- \({{\varvec{W}}}_{\mathrm{iso}}\) :
-
Strain energy density (MPa)
- \(\beta \) :
-
Reactive compliance (Å)
- \(\gamma \) :
-
Aspect ratio
- \(\dot{\gamma }\) :
-
Local shear rate (s−1)
- \({\delta }_{\mathrm{eq}}\) :
-
Equilibrium bond length (m)
- \({\eta }_{0}\) :
-
Zero-shear viscosity (Pa.s)
- \({\eta }_{\infty }\) :
-
Infinite-shear viscosity (Pa.s)
- \({\eta }_{\mathrm{app}}S\) :
-
Wall shear stress (MPa)
- \(\lambda \) :
-
Time constant (s)
- \({\mu }_{0}\) :
-
Magnetic permeability of vacuum (N.A−2)
- \({\mu }_{p}\) :
-
Magnetic permeability of particle (N.A−2)
- \({\mu }_{r}\) :
-
Magnetic permeability (N.A−2)
- \(\rho \) :
-
Density (Kg.m−3)
- \(\sigma \) :
-
Electrical conductivity (Ω.m)
- \({\varvec{\tau}}\) :
-
Stress tensor of the fluid (MPa)
- \(\chi \) :
-
Magnetic susceptibility
References
Alishiri M, Ebrahimi S, Shamloo A et al (2021) Drug delivery and adhesion of magnetic nanoparticles coated nanoliposomes and microbubbles to atherosclerotic plaques under magnetic and ultrasound fields. Eng Appl Comput Fluid Mech 15:1703–1725. https://doi.org/10.1080/19942060.2021.1989042
Ascher UM, Petzold LR (2011) Computer methods for ordinary differential equations and differential-algebraic equations
Asmatulu R, Zalich MA, Claus RO, Riffle JS (2005) Synthesis, characterization and targeting of biodegradable magnetic nanocomposite particles by external magnetic fields. J Magn Magn Mater. https://doi.org/10.1016/j.jmmm.2004.10.103
Babinec P, Krafčík A, Babincová M, Rosenecker J (2010) Dynamics of magnetic particles in cylindrical Halbach array: Implications for magnetic cell separation and drug targeting. Med Biol Eng Comput. https://doi.org/10.1007/s11517-010-0636-8
Biglari H, Razaghi R, Ebrahimi S, Karimi A (2019) A computational dynamic finite element simulation of the thoracic vertebrae under blunt loading: spinal cord injury. J Brazilian Soc Mech Sci Eng 41:84. https://doi.org/10.1007/s40430-019-1588-z
Canchi T, Saxena A, Ng EYK et al (2018) Application of fluid-structure interaction methods to estimate the mechanics of rupture in asian abdominal aortic aneurysms. Bionanoscience 8:1035–1044. https://doi.org/10.1007/s12668-018-0554-z
Cheng J, Zhang R, Li C et al (2018) A targeting nanotherapy for abdominal aortic aneurysms. J Am Coll Cardiol. https://doi.org/10.1016/j.jacc.2018.08.2188
Chertok B, Langer R (2018) Circulating magnetic microbubbles for localized real-time control of drug delivery by ultrasonography-guided magnetic targeting and ultrasound. Theranostics. https://doi.org/10.7150/thno.20781
Chung J, Hulbert GM (2008) A time integration algorithm for structural dynamics with improved numerical dissipation: the generalized-α method. J Appl Mech Doi 10(1115/1):2900803
Cohen Stuart D.C (2009) The development of a discrete particle model for 3D unstructured grids: application to magnetic drug targeting. M.Sc thesis, Delft Univ Technol Title
Curtiss CF, Hirschfelder JO (2006) Integration of stiff equations. Proc Natl Acad Sci. https://doi.org/10.1073/pnas.38.3.235
Davis FM, Rateri DL, Daugherty A (2015) Abdominal aortic aneurysm: Novel mechanisms and therapies. Curr Opin Cardiol 30:566–573. https://doi.org/10.1097/HCO.0000000000000216
Decuzzi P, Ferrari M (2006) The adhesive strength of non-spherical particles mediated by specific interactions. Biomaterials. https://doi.org/10.1016/j.biomaterials.2006.05.024
Dhamacharoen A (2016) Efficient numerical methods for solving differential algebraic equations. J Appl Math Phys. https://doi.org/10.4236/jamp.2016.41007
Duan L, Yang F, He W et al (2016) A multi-gradient targeting drug delivery system based on RGD-l-TRAIL-labeled magnetic microbubbles for cancer theranostics. Adv Funct Mater. https://doi.org/10.1002/adfm.201603637
Ebrahimi S, Shamloo A, Alishiri M et al (2021a) Targeted pulmonary drug delivery in coronavirus disease (COVID-19) therapy: A patient-specific in silico study based on magnetic nanoparticles-coated microcarriers adhesion. Int J Pharm. https://doi.org/10.1016/j.ijpharm.2021.121133
Ebrahimi S, Vatani P, Amani A, Shamloo A (2021b) Drug delivery performance of nanocarriers based on adhesion and interaction for abdominal aortic aneurysm treatment. Int J Pharm. https://doi.org/10.1016/j.ijpharm.2020.120153
El-Boubbou K, Ali R, Al-Zahrani H et al (2019) Preparation of iron oxide mesoporous magnetic microparticles as novel multidrug carriers for synergistic anticancer therapy and deep tumor penetration. Sci Rep. https://doi.org/10.1038/s41598-019-46007-z
Fong SW, Klaseboer E, Khoo BC (2008) Interaction of microbubbles with high intensity pulsed ultrasound. J Acoust Soc Am Doi 10(1121/1):2836746
Furlani EP (2007) Magnetophoretic separation of blood cells at the microscale. J Phys D Appl Phys. https://doi.org/10.1088/0022-3727/40/5/001
Gao Y, Chan CU, Gu Q et al (2016) Controlled nanoparticle release from stable magnetic microbubble oscillations. NPG Asia Mater. https://doi.org/10.1038/am.2016.37
Goldman AJ, Cox RG, Brenner H (1967) Slow viscous motion of a sphere parallel to a plane wall-II Couette flow. Chem Eng Sci. https://doi.org/10.1016/0009-2509(67)80048-4
Golledge J (2019) Abdominal aortic aneurysm: update on pathogenesis and medical treatments. Nat Rev Cardiol 16(4):225–242
Gori F, Boghi A (2011) Two new differential equations of turbulent dissipation rate and apparent viscosity for non-newtonian fluids. Int Commun Heat Mass Transf. https://doi.org/10.1016/j.icheatmasstransfer.2011.03.003
Gori F, Boghi A (2012) A three dimensional exact equation for the turbulent dissipation rate of Generalised Newtonian Fluids. Int Commun Heat Mass Transf. https://doi.org/10.1016/j.icheatmasstransfer.2012.02.010
Joldes GR, Miller K, Wittek A, Doyle B (2016) A simple, effective and clinically applicable method to compute abdominal aortic aneurysm wall stress. J Mech Behav Biomed Mater. https://doi.org/10.1016/j.jmbbm.2015.07.029
Jorgensen WL, Tirado-Rives J (1988) The OPLS potential functions for proteins. energy minimizations for crystals of cyclic peptides and crambin. J Am Chem Soc https://doi.org/10.1021/ja00214a001
Karthikesalingam A, Vidal-Diez A, Holt PJ et al (2016) Thresholds for abdominal aortic aneurysm repair in England and the United States. N Engl J Med. https://doi.org/10.1056/NEJMoa1600931
Kayal S, Bandyopadhyay D, Mandal TK, Ramanujan RV (2011) The flow of magnetic nanoparticles in magnetic drug targeting. RSC Adv. https://doi.org/10.1039/c1ra00023c
Kelly M, Yeoh GH, Timchenko V (2015) On computational fluid dynamics study of magnetic drug targeting. J Comput Multiph Flows. https://doi.org/10.1260/1757-482X.7.1.43
Kim CS, Iglesias AJ, Garcia L (1989) Deposition of inhaled particles in bifurcating airway models: II. Expiratory deposition. J Aerosol Med Depos Clear Eff Lung 2(1):15–27. https://doi.org/10.1089/jam.1989.2.15
Kirby BJ (2010) Micro- and nanoscale fluid mechanics, Transport in Microfluidic Devices. Cambridge University Press, Cambridge
Krafcik A, Babinec P, Frollo I (2015) Computational analysis of magnetic field induced deposition of magnetic particles in lung alveolus in comparison to deposition produced with viscous drag and gravitational force. J Magn Magn Mater. https://doi.org/10.1016/j.jmmm.2014.10.018
Kwon O (2008) Effect of blood viscosity on oxygen transport in residual stenosed artery following angioplasty. J Biomech Eng Doi 10(1115/1):2838029
Lai WM, Rubin D, Krempl E (2010) Introduction to continuum mechanics (Fourth Edition)
Larimi MM, Ramiar A, Ranjbar AA (2014) Numerical simulation of magnetic nanoparticles targeting in a bifurcation vessel. J Magn Magn Mater. https://doi.org/10.1016/j.jmmm.2014.03.002
Lederle FA, Freischlag JA, Kyriakides TC et al (2012) Long-term comparison of endovascular and open repair of abdominal aortic aneurysm. N Engl J Med. https://doi.org/10.1056/NEJMoa1207481
Leung JHJH, Wright ARARAR, Cheshire NN et al (2006) Fluid structure interaction of patient specific abdominal aortic aneurysms a comparison with solid stress models. Biomed Eng Online 5:33. https://doi.org/10.1186/1475-925X-5-33
Lu YJ, Chuang EY, Cheng YH et al (2019) Thermosensitive magnetic liposomes for alternating magnetic field-inducible drug delivery in dual targeted brain tumor chemotherapy. Chem Eng J. https://doi.org/10.1016/j.cej.2019.05.055
Lunnoo T, Puangmali T (2015) Capture efficiency of biocompatible magnetic nanoparticles in arterial flow: a computer simulation for magnetic drug targeting. Nanoscale Res Lett. https://doi.org/10.1186/s11671-015-1127-5
Mannell H, Pircher J, Fochler F et al (2012) Site directed vascular gene delivery in vivo by ultrasonic destruction of magnetic nanoparticle coated microbubbles. Nanomedicine Nanotechnology, Biol Med. https://doi.org/10.1016/j.nano.2012.03.007
Manshadi MKD, Saadat M, Mohammadi M et al (2018) Delivery of magnetic micro/nanoparticles and magnetic-based drug/cargo into arterial flow for targeted therapy. Drug Deliv. https://doi.org/10.1080/10717544.2018.1497106
Manzoori A, Fallah F, Sharzehee M, Ebrahimi S (2020) Computational Investigation of the stability of stenotic carotid artery under pulsatile blood flow using a fluid-structure interaction approach. Int J Appl Mech 12:1758–8251. https://doi.org/10.1142/S1758825120501100
McPhee JT, Hill JS, Eslami MH (2007) The impact of gender on presentation, therapy, and mortality of abdominal aortic aneurysm in the United States, 2001–2004. J Vasc Surg. https://doi.org/10.1016/j.jvs.2007.01.043
McQuareue DA (1963) Kinetics of small systems. I J Chem Phys. https://doi.org/10.1063/1.1733676
Michel M, Becquemin J-P, Marzelle J et al (2018) Editor’s choice – a study of the cost-effectiveness of fenestrated/branched EVAR compared with open surgery for patients with complex aortic aneurysms at 2 years. Eur J Vasc Endovasc Surg 56:15–21. https://doi.org/10.1016/J.EJVS.2017.12.008
Mills CJ, Gabe IT, Gault JH et al (1970) Pressure-flow relationships and vascular impedance in man. Cardiovasc Res. https://doi.org/10.1093/cvr/4.4.405
Mirzababaei SN, Gorji TB, Baou M et al (2017) Investigation of magnetic nanoparticle targeting in a simplified model of small vessel aneurysm. J Magn Magn Mater. https://doi.org/10.1016/j.jmmm.2016.11.044
Moran CS, McCann M, Karan M et al (2005) Association of osteoprotegerin with human abdominal aortic aneurysm progression. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.104.464727
Nourbakhsh E, Mohammadi A, Salemizadeh Parizi M et al (2021) Role of myeloid-derived suppressor cell (MDSC) in autoimmunity and its potential as a therapeutic target. Inflammopharmacology 29:1307–1315
Park SM, Hong MK, Kim SH et al (2019) Comparison of efficacy between ramipril and carvedilol on limiting the expansion of abdominal aortic aneurysm in mouse model. J Cardiovasc Pharmacol Ther. https://doi.org/10.1177/1074248418798631
Peng H, Qian X, Mao L et al (2019) Ultrafast ultrasound imaging in acoustic microbubble trapping. Appl Phys Lett Doi 10(1063/1):5124437
Piper JW, Swerlick RA, Zhu C (1998) Determining force dependence of two-dimensional receptor-ligand binding affinity by centrifugation. Biophys J. https://doi.org/10.1016/S0006-3495(98)77807-5
Pourmehran O, Rahimi-Gorji M, Gorji-Bandpy M, Gorji TB (2015) Simulation of magnetic drug targeting through tracheobronchial airways in the presence of an external non-uniform magnetic field using Lagrangian magnetic particle tracking. J Magn Magn Mater. https://doi.org/10.1016/j.jmmm.2015.05.086
Powell JT, Sweeting MJ, Ulug P et al (2017) Meta-analysis of individual-patient data from EVAR-1, DREAM, OVER and ACE trials comparing outcomes of endovascular or open repair for abdominal aortic aneurysm over 5 years. Br J Surg 104(3):166–178
Raghavan ML, Vorp DA (2000) Toward a biomechanical tool to evaluate rupture potential of abdominal aortic aneurysm: Identification of a finite strain constitutive model and evaluation of its applicability. J Biomech. https://doi.org/10.1016/S0021-9290(99)00201-8
Raut SS, Chandra S, Shum J et al (2013) Biological, geometric and biomechanical factors influencing abdominal aortic aneurysm rupture risk: a comprehensive review. Recent Patents Med Imaging 2(2):44–59
Saffman PG (1965) The lift on a small sphere in a slow shear flow. J Fluid Mech. https://doi.org/10.1017/S0022112065000824
Salemizadeh Parizi M, Salemizadeh Parizi F, Abdolhosseini S et al (2021) Myeloid-derived suppressor cells (MDSCs) in brain cancer: challenges and therapeutic strategies. Inflammopharmacology 29(6):1613–1624
Sampson UKA, Norman PE, Fowkes FGR et al (2014) Global and regional burden of aortic dissection and aneurysms: Mortality trends in 21 world regions, 1990 to 2010. Glob Heart 9(1):171–180
Schenck JF (1996) The role of magnetic susceptibility in magnetic resonance imaging: MRI magnetic compatibility of the first and second kinds. Med Phys 10(1118/1):597854
Scotti CM, Finol EA (2007) Compliant biomechanics of abdominal aortic aneurysms: A fluid-structure interaction study. Comput Struct 85:1097–1113. https://doi.org/10.1016/j.compstruc.2006.08.041
Scotti CM, Shkolnik AD, Muluk SC, Finol EA (2005) Fluid-structure interaction in abdominal aortic aneurysms: Effects of asymmetry and wall thickness. Biomed Eng Online 4:1–22. https://doi.org/10.1186/1475-925X-4-64
Shamloo A, Amani A, Forouzandehmehr M, Ghoytasi I (2019) In Silico study of patient-specific magnetic drug targeting for a coronary LAD atherosclerotic plaque. Int J Pharm. https://doi.org/10.1016/j.ijpharm.2018.12.088
Shamloo A, Ebrahimi S, Amani A, Fallah F (2020) Targeted drug delivery of microbubble to arrest abdominal aortic aneurysm development: a simulation study towards optimized microbubble design. Sci Rep. https://doi.org/10.1038/s41598-020-62410-3
Shamloo A, Forouzandehmehr M (2019) Personalised deposition maps for micro- and nanoparticles targeting an atherosclerotic plaque: attributions to the receptor-mediated adsorption on the inflamed endothelial cells. Biomech Model Mechanobiol 18(3):813–828
Shamloo A, Manuchehrfar F, Rafii-Tabar H (2015a) A viscoelastic model for axonal microtubule rupture. J Biomech 48:1241–1247. https://doi.org/10.1016/j.jbiomech.2015.03.007
Shamloo A, Mohammadaliha N, Mohseni M (2015b) integrative utilization of microenvironments, biomaterials and computational techniques for advanced tissue engineering. J Biotechnol 212:71–89
Shamloo A, Nejad MA, Saeedi M (2017) Fluid–structure interaction simulation of a cerebral aneurysm: Effects of endovascular coiling treatment and aneurysm wall thickening. J Mech Behav Biomed Mater 74:72–83. https://doi.org/10.1016/j.jmbbm.2017.05.020
Shamloo A, Pedram MZ, Heidari H, Alasty A (2016) Computing the blood brain barrier (BBB) diffusion coefficient: a molecular dynamics approach. J Magn Magn Mater. https://doi.org/10.1016/j.jmmm.2016.03.030
Shamsi M, Sedaghatkish A, Dejam M et al (2018) Magnetically assisted intraperitoneal drug delivery for cancer chemotherapy. Drug Deliv. https://doi.org/10.1080/10717544.2018.1455764
Sharifi A, Motlagh SY, Badfar H (2019) Numerical investigation of magnetic drug targeting using magnetic nanoparticles to the Aneurysmal Vessel. J Magn Magn Mater. https://doi.org/10.1016/j.jmmm.2018.10.147
Simsek FG, Kwon YW (2015) Investigation of material modeling in fluid–structure interaction analysis of an idealized three-layered abdominal aorta: aneurysm initiation and fully developed aneurysms. J Biol Phys 41:173–201. https://doi.org/10.1007/s10867-014-9372-x
Stride E, Porter C, Prieto AG, Pankhurst Q (2009) Enhancement of microbubble mediated gene delivery by simultaneous exposure to ultrasonic and magnetic fields. Ultrasound Med Biol. https://doi.org/10.1016/j.ultrasmedbio.2008.11.010
Tan J, Wang S, Yang J, Liu Y (2013) Coupled particulate and continuum model for nanoparticle targeted delivery. Comput Struct. https://doi.org/10.1016/j.compstruc.2012.12.019
Tehrani MD, Yoon JH, Kim MO, Yoon J (2015) A novel scheme for nanoparticle steering in blood vessels using a functionalized magnetic field. IEEE Trans Biomed Eng. https://doi.org/10.1109/TBME.2014.2351234
Thakur SS, Chen YS, Houston ZH et al (2019) Ultrasound-responsive nanobubbles for enhanced intravitreal drug migration: an ex vivo evaluation. Eur J Pharm Biopharm. https://doi.org/10.1016/j.ejpb.2019.01.014
Umbarkar TS, Kleinstreuer C (2015) Computationally efficient fluid-particle dynamics simulations of arterial systems. Commun Comput Phys. https://doi.org/10.4208/cicp.160114.120914a
Wang X, Searle AK, Hohmann JD et al (2018a) Dual-targeted theranostic delivery of mirs arrests abdominal aortic aneurysm development. Mol Ther 26:1056–1065. https://doi.org/10.1016/j.ymthe.2018.02.010
Wang Y, Chen C, Wang Q et al (2018b) Inhibitory effects of cycloastragenol on abdominal aortic aneurysm and its related mechanisms. Br J Pharmacol. https://doi.org/10.1111/bph.14515
Zhang B, Kim H, Wu H et al (2019) Sonothrombolysis with magnetic microbubbles under a rotational magnetic field. Ultrasonics. https://doi.org/10.1016/j.ultras.2019.06.004
Acknowledgements
The authors declare that they have no known competing for financial interests or personal relationships that could have influenced the work reported in this paper.
Funding
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript.
Author information
Authors and Affiliations
Contributions
Amir Shamloo initiated the idea, designed the simulations, analyzed the data, and wrote the paper; Sina Ebrahimi designed the simulations, analyzed the data, and wrote the paper; Ghazal Ghorbani ran the simulations and wrote the paper. Mojgan Alishiri, ran the simulations and revised the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical Approval
Not applicable.
Consent to participate
Not applicable.
Consent to publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shamloo, A., Ebrahimi, S., Ghorbani, G. et al. Targeted drug delivery of magnetic microbubble for abdominal aortic aneurysm: an in silico study. Biomech Model Mechanobiol 21, 735–753 (2022). https://doi.org/10.1007/s10237-022-01559-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-022-01559-4