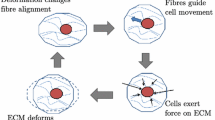
Abstract
Collagen is the main load-bearing component of many soft tissues and has a large influence on the mechanical behavior of tissues when exposed to mechanical loading. Therefore, it is important to increase our understanding of collagen remodeling in soft tissues to understand the mechanisms behind pathologies and to control the development of the collagen network in engineered tissues. In the present study, a constitutive model was developed by coupling a recently developed model describing the orientation and contractile stresses exerted by cells in response to mechanical stimuli to physically motivated collagen remodeling laws. In addition, cell-mediated contraction of the collagen fibers was included as a mechanism for tissue compaction. The model appeared to be successful in predicting a range of experimental observations, which are (1) the change in transition stretch of periosteum after remodeling at different applied stretches, (2) the compaction and alignment of collagen fibers in tissue-engineered strips, (3) the fiber alignment in cruciform gels with different arm widths, and (4) the alignment of collagen fibers in engineered vascular grafts. Moreover, by changing the boundary conditions, the model was able to predict a helical architecture in the vascular graft without assuming the presence of two helical fiber families a priori. Ultimately, this model may help to increase our understanding of collagen remodeling in physiological and pathological conditions, and it may provide a tool for determining the optimal experimental conditions for obtaining native-like collagen architectures in engineered tissues.







Similar content being viewed by others
References
Ambrosi D, Ateshian GA, Arruda EM, Cowin SC, Dumais J, Goriely A, Holzapfel GA, Humphrey JD, Kemkemer R, Kuhl E, Olberding JE, Taber LA, Garikipati K (2011) Perspectives on biological growth and remodeling. J Mech Phys Solids 59(4):863–883
Ateshian GA, Rajan V, Chahine NO, Canal CE, Hung CT (2009) Modeling the matrix of articular cartilage using a continuous fiber angular distribution predicts many observed phenomena. J Biomech Eng 131(6):061003
Baek S, Rajagopal KR, Humphrey JD (2006) A theoretical model of enlarging intracranial fusiform aneurysms. J Biomech Eng 128(1):142–149
Bhole AP, Flynn BP, Liles M, Saeidi N, Dimarzio CA, Ruberti JW (2009) Mechanical strain enhances survivability of collagen micronetworks in the presence of collagenase: implications for load-bearing matrix growth and stability. Philos Trans A Math Phys Eng Sci 367(1902):3339–3362
Billiar KL, Sacks MS (2000a) Biaxial mechanical properties of the natural and glutaraldehyde treated aortic valve cusp—Part I: Experimental results. J Biomech Eng 122:23–30
Billiar KL, Sacks MS (2000b) Biaxial mechanical properties of the natural and glutaraldehyde-treated aortic valve cusp—Part II: A structural constitutive model. J Biomech Eng 122:327–335
Canty EG, Lu Y, Meadows RS, Shaw MK, Holmes DF, Kadler KE (2004) Coalignment of plasma membrane channels and protrusions (fibripositors) specifies the parallelism of tendon. J Cell Biol 165(4):553–563
Deshpande VS, McMeeking RM, Evans AG (2006) A bio-chemo-mechanical model for cell contractility. Proc Natl Acad Sci USA 103(38):14015–14020
Deshpande VS, McMeeking RM, Evans AG (2007) A model for the contractility of the cytoskeleton including the effects of stress-fibre formation and dissociation. Proc R Soc A 463:787–815
Driessen NJB, Wilson W, Bouten CVC, Baaijens FPT (2004) A computational model for collagen fibre remodelling in the arterial wall. J Theor Biol 226(1):53–64
Driessen NJB, Mol A, Bouten CVC, Baaijens FPT (2007) Modeling the mechanics of tissue-engineered human heart valve leaflets. J Biomech 40(2):325–334
Driessen NJB, Cox MAJ, Bouten CVC, Baaijens FPT (2008) Remodelling of the angular collagen fiber distribution in cardiovascular tissues. Biomech Model Mechanobiol 7(2):93–103
Fan R, Bayoumi AS, Chen P, Hobson CM, Wagner WR, Mayer JE Jr, Sacks MS (2013) Optimal elastomeric scaffold leaflet shape for pulmonary heart valve leaflet replacement. J Biomech 46(4):662–669
Foolen J, Van Donkelaar CC, Soekhradj-Soechit S, Ito K (2010) An adaptation mechanism for fibrous tissue to sustained shortening. J Biomech 43(16):3168–3176
Gasser TC, Ogden RW, Holzapfel GA (2006) Hyperelastic modelling of arterial layers with distributed collagen fibre orientations. J R Soc Interface 3(6):15–35
Gasser TC, Gallinetti S, Xing X, Forsell C, Swedenborg J, Roy J (2012) Spatial orientation of collagen fibers in the abdominal aortic aneurysm’s wall and its relation to wall mechanics. Acta Biomater 8(8):3091–3103
Gealy C, Hayes AJ, Buckwell R, Young RD, Caterson B, Quantock AJ, Ralphs JR (2009) Actin and type I collagen propeptide distribution in the developing chick cornea. Invest Ophthalmol Vis Sci 50(4):1653–1658
Hariton I, deBotton G, Gasser TC, Holzapfel GA (2007) Stress-driven collagen fiber remodeling in arterial walls. Biomech Model Mechanobiol 6(3):163–175
Holzapfel GA, Gasser TC, Stadler M (2002) A structural model for the viscoelastic behavior of arterial walls: continuum formulation and finite element analysis. Eur J Mech A Solids 21(3):441–463
Hsu HJ, Lee CF, Locke A, Vanderzyl SQ, Kaunas R (2010) Stretch-induced stress fiber remodeling and the activations of JNK and ERK depend on mechanical strain rate, but not FAK. PloS One 5(8):e12470
Huang C, Yannas IV (1977) Mechanochemical studies of enzymatic degradation of insoluble collagen fibers. J Biomed Mater Res 11(1):137–154
Humphrey JD (2001) Stress, strain, and mechanotransduction in cells. J Biomech Eng 123(6):638–641
Jhun CS, Evans MC, Barocas VH, Tranquillo RT (2009) Planar biaxial mechanical behavior of bioartificial tissues possessing prescribed fiber alignment. J Biomech Eng 131(8):081006
Kuhl E, Holzapfel GA (2007) A continuum model for remodeling in living structures. J Mater Sci 42:8811–8823
Loerakker S, Argento G, Oomens CWJ, Baaijens FPT (2013) Effects of valve geometry and tissue anisotropy on the radial stretch and coaptation area of tissue-engineered heart valves. J Biomech 46(11):1792–1800
Martin C, Sun W (2012) Biomechanical characterization of aortic valve tissue in humans and common animal models. J Biomed Mater Res Part A 100:1591–1599
Martufi G, Gasser TC (2012) Turnover of fibrillar collagen in soft biological tissue with application to the expansion of abdominal aortic aneurysms. J R Soc Interface 9(77):3366–3377
Menzel A, Kuhl E (2012) Frontiers in growth and remodeling. Mech Res Commun 42:1–14
Meshel AS, Wei Q, Adelstein RS, Sheetz MP (2005) Basic mechanism of three-dimensional collagen fibre transport by fibroblasts. Nat Cell Biol 7(2):157–164
Nagel T, Kelly DJ (2012) Remodelling of collagen fibre transition stretch and angular distribution in soft biological tissues and cell-seeded hydrogels. Biomech Model Mechanobiol 11(3–4):325– 339
Neidert MR, Tranquillo RT (2006) Tissue-engineered valves with commissural alignment. Tissue Eng 12(4):891–903
Obbink-Huizer C, Oomens CWJ, Loerakker S, Foolen J, Bouten CVC, Baaijens FPT (2013) Computational model predicts cell orientation in response to a range of mechanical stimuli. Biomech Model Mechanobiol. doi:10.1007/s10237-013-0501-4
Pierce DM, Ricken T, Holzapfel GA (2013) Modeling sample/patient-specific structural and diffusional responses of cartilage using DT-MRI. Int J Numer Methods Biomed Eng 29(8):807–821
Robinson PS, Johnson SL, Evans MC, Barocas VH, Tranquillo RT (2008) Functional tissue-engineered valves from cell-remodeled fibrin with commissural alignment of cell-produced collagen. Tissue Eng Part A 14(1):83–95
Rodriguez EK, Hoger A, McCulloch AD (1994) Stress-dependent finite growth in soft elastic tissues. J Biomech 27(4):455–467
Rubbens MP, Driessen-Mol A, Boerboom RA, Koppert MMJ, Van Assen HC, Baaijens FPT, Bouten CVC (2009) Quantification of the temporal evolution of collagen orientation in mechanically conditioned engineered cardiovascular tissues. Ann Biomed Eng 37(7):1263–1272
Ruberti JW, Hallab NJ (2005) Strain-controlled enzymatic cleavage of collagen in loaded matrix. Biochem Biophys Res Commun 336(2):483–489
Sander EA, Barocas VH, Tranquillo RT (2011) Initial fiber alignment pattern alters extracellular matrix synthesis in fibroblast-populated fibrin gel cruciforms and correlates with predicted tension. Ann Biomed Eng 39(2):714–729
Schriefl AJ, Zeindlinger G, Pierce DM, Regitnig P, Holzapfel GA (2012) Determination of the layer-specific distributed collagen fibre orientations in human thoracic and abdominal aortas and common iliac arteries. J R Soc Interface 9(71):1275–1286
Schriefl AJ, Wolinski H, Regitnig P, Kohlwein SD, Holzapfel GA (2013) An automated approach for three-dimensional quantification of fibrillar structures in optically cleared soft biological tissues. J R Soc Interface 10(80):20120760
Shi Y, Vesely I (2003) Fabrication of mitral valve chordae by directed collagen gel shrinkage. Tissue Eng 9(6):1233–1242
Soares ALF, Stekelenburg M, Baaijens FPT (2011) Remodeling of the collagen fiber architecture due to compaction in small vessels under tissue engineered conditions. J Biomech Eng 133(7):071002
Soares ALF, Oomens CWJ, Baaijens FPT (2012) A computational model to describe the collagen orientation in statically cultured engineered tissues. Comput Methods Biomech Biomed Eng. doi:10.1080/10255842.2012.680192
Syedain ZH, Meier LA, Reimer JM, Tranquillo RT (2013) Tubular heart valves from decellularized engineered tissue. Ann Biomed Eng. doi:10.1007/s10439-013-0872-9
Valentín A, Humphrey JD, Holzapfel GA (2013) A finite element-based constrained mixture implementation for arterial growth, remodeling and adaptation: theory and numerical verification. Int J Numer Methods Biomed Eng 29(8):822–849
Van Donkelaar CC, Heck TAM, Wilson W, Foolen J, Ito K (2013) Versatility of a collagen adaptation model that includes strain-dependent degeneration and cell traction. In: Proceedings of the ASME 2013 summer bioengineering conference (SBC2013-14214), Sunriver, OR, USA
Van Vlimmeren MAA, Driessen-Mol A, Oomens CWJ, Baaijens FPT (2011) An in vitro model system to quantify stress generation, compaction, and retraction in engineered heart valve tissue. Tissue Eng Part C Methods 17(10):983–991
Vernerey FJ, Farsad M (2011) A constrained mixture approach to mechano-sensing and force generation in contractile cells. J Mech Behav Biomed Mater 4(8):1683–1699
Wan W, Hansen L, Gleason RL Jr (2010) A 3-D constrained mixture model for mechanically mediated vascular growth and remodeling. Biomech Model Mechanobiol 9(4):403–419
Wang JHC, Jia F, Gilbert TW, Woo SLY (2003) Cell orientation determines the alignment of cell-produced collagenous matrix. J Biomech 36(1):97–102
Watton PN, Hill NA (2009) Evolving mechanical properties of a model of abdominal aortic aneurysm. Biomech Model Mechanobiol 8(1):25–42
Weidenhamer NK, Tranquillo RT (2013) Influence of cyclic mechanical stretch and tissue constraints on cellular and collagen alignment in fibroblast-derived cell sheets. Tissue Eng Part C Methods 19(5):186–395
Wyatt KEK, Bourne JW, Torzilli PA (2009) Deformation-dependent enzyme mechanokinetic cleavage of type I collagen. J Biomech Eng 131(5):051004
Acknowledgments
The research leading to these results has received funding from the European Union’s Seventh Framework Programme (FP7/2007-2013) under Grant Agreement No. 242008.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Loerakker, S., Obbink-Huizer, C. & Baaijens, F.P.T. A physically motivated constitutive model for cell-mediated compaction and collagen remodeling in soft tissues. Biomech Model Mechanobiol 13, 985–1001 (2014). https://doi.org/10.1007/s10237-013-0549-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-013-0549-1