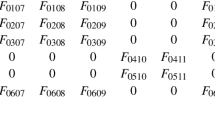Abstract
Mechanical interactions between cells and the fibrous extracellular matrix (ECM) in which they reside play a key role in tissue development. Mechanical cues from the environment (such as stress, strain and fibre orientation) regulate a range of cell behaviours, including proliferation, differentiation and motility. In turn, the ECM structure is affected by cells exerting forces on the matrix which result in deformation and fibre realignment. In this paper we develop a mathematical model to investigate this mechanical feedback between cells and the ECM. We consider a three-phase mixture of collagen, culture medium and cells, and formulate a system of partial differential equations which represents conservation of mass and momentum for each phase. This modelling framework takes into account the anisotropic mechanical properties of the collagen gel arising from its fibrous microstructure. We also propose a cell–collagen interaction force which depends upon fibre orientation and collagen density. We use a combination of numerical and analytical techniques to study the influence of cell–ECM interactions on pattern formation in tissues. Our results illustrate the wide range of structures which may be formed, and how those that emerge depend upon the importance of cell–ECM interactions.



















Similar content being viewed by others
References
Barocas VH, Tranquillo RT (1997) An anisotropic biphasic theory of tissue-equivalent mechanics: the interplay among cell traction, fibrillar network deformation, fibril alignment and cell contact guidance. J Biomech Eng 119:137–145
Barocas VH, Moon AG, Tranquillo RT (1995) The fibroblast-populated collagen microsphere assay of cell traction force—Part 2: measurement of the cell traction parameter. J Biomech Eng 117:161–170
Bissell MJ, Radisky D (2001) Putting tumours in context. Nat Rev Cancer 1:46–54
Breward CJW, Byrne HM, Lewis CE (2002) The role of cell–cell interactions in a two-phase model for avascular tumour growth. J Math Biol 45(2):125–152
Byfield FJ, Reen RK, Shentu TP, Levitan I, Gooch KJ (2009) Endothelial actin and cell stiffness is modulated by substrate stiffness in 2D and 3D. J Biomech 42(8):1114–1119
Byrne HM, Preziosi L (2003) Modelling solid tumour growth using the theory of mixtures. Math Med Biol 20:341–366
Byrne HM, King JR, McElwain DLS, Preziosi L (2003) A two-phase model of solid tumour growth. Appl Math Lett 16(4):567–573
Chauviere A, Hillen T, Preziosi L (2007) Modelling cell movement in anisotropic and heterogeneous network tissues. Netw Heterog Media 2(2):333–357
Cockburn B, Shu CW (1998) The Runge–Kutta discontinuous Galerkin method for conservation laws V. J Comput Phys 141:199–224
Cook J (1995) Mathematical models for dermal wound healing: wound contraction and scar formation. PhD thesis, University of Washington
Cukierman E, Bassi DE (2010) Physico-mechanical aspects of extracellular matrix influences on tumourigenic behaviors. Semin Cancer Biol 20(3):139–145
Dhimolea E, Maffini MV, Soto AM, Sonnenschein C (2010) The role of collagen reorganization on mammary epithelial morphogenesis in a 3d culture model. Biomaterials 31:3622–3630
Drew DA (1983) Mathematical modelling of two-phase flow. Ann Rev Fluid Mech 15:261–291
Dyson RJ, Jensen OE (2010) A fibre-reinforced fluid model of anisotropic plant cell growth. J Fluid Mech 655:472–503
Engler AJ, Sen S, Sweeney HL, Discher DE (2006) Matrix elasticity directs stem cell lineage specification. Cell 126:677–689
Ericksen JL (1960) Transversely isotropic fluids. Colloid Polym Sci 173(2):117–122
Eriksson K, Estep D, Hansbo P, Johnson C (1996) Computational differential equations. Cambridge University Press, Cambridge
Gerisch A, Chaplain MAJ (2008) Mathematical modelling of cancer cell invasion of tissue: local and non-local models and the effect of adhesion. J Theor Biol 250(4):684–704
Green JEF, Friedman A (2008) The extensional flow of a thin sheet of incompressible, transversely isotropic fluid. Euro J Appl Math 19(3):225–257
Green JEF, Waters SL, Shakesheff KM, Byrne HM (2009) A mathematical model of liver cell aggregation in vitro. Bull Math Biol 71:906–930
Green JEF, Waters SL, Shakesheff KM, Edelstein-Keshet L, Byrne HM (2010) Non-local models for the interactions of hepatocytes and stellate cells during aggregation. J Theor Biol 267(1):106–120
Green JEF, Bassom AP, Friedman A (2013) A mathematical model for cell-induced gel compaction in vitro. Math Models Methods Appl Sci 23(1):127–163. doi:10.1142/S0218202512500479
Häcker A (2012) A mathematical model for mesenchymal and chemosensitive cell dynamics. J Math Biol 64:361–401
Hillen T (2006) M\(^5\) mesoscopic and macroscopic models for mesenchymal motion. J Math Biol 53(4):585–616
Hinch EJ, Leal LG (1975) Constitutive equations in suspension mechanics. Part 1. General formulation. J Fluid Mech 71(3):481–495
Hinch EJ, Leal LG (1976) Constitutive equations in suspension mechanics. Part 2. Approximate forms for a suspension of rigid particles affected by Brownian rotations. J Fluid Mech 76(1):187–208
Holloway C, Dyson R, Smith D (2015) Linear Taylor-Couette stability of a transversely isotropic fluid. Proc R Soc A 471:20150141. doi:10.1098/rspa.2015.0141
Ingber DE (2006) Mechanical control of tissue morphogenesis during embryological development. Int J Dev Biol 50:255–266
Ingber DE (2008) Can cancer be reversed by engineering the tumour microenvironment? Semin Cancer Biol 18(5):356–364
Jaalouk DE, Lammerding J (2009) Mechanotransduction gone awry. Nat Rev Mol Cell Biol 10:63–73
Kabla A, Mahadevan L (2007) Nonlinear mechanics of soft fibre networks. J R Soc Interface 4(12):99–106
Kirkpatrick ND, Andreou S, Hoying JB, Utzinger U (2007) Live imaging of collagen remodeling during angiogenesis. Am J Physiol Heart Circ Physiol 292(6):H3198–H3206
Knapp DM, Barocas VH, Moon AG, Yoo K, Petzold LR, Tranquillo RT (1997) Rheology of reconstituted type i collagen gel in confined compression. J Rheol 41:971–933
Korff T, Augustin HG (1999) Tensional forces in fibrillar extracellular matrices control directional capillary sprouting. J Cell Sci 112:3249–3258
Krause S, Maffini MV, Soto AM, Sonnenschein C (2008) A novel 3d in vitro culture model to study stromal–epithelial interactions in the mammary gland. Tissue Eng 14:261–271
Kumar S, Weaver VM (2009) Mechanics, malignancy, and metastasis: the force journey of a tumour cell. Cancer Metastasis Rev 28:113–127
Lee MEM (2001) Mathematical models of the carding process. PhD thesis, University of Oxford
Lee MEM, Ockendon H (2005) A continuum model for entangled fibres. Euro J Appl Math 16:145–160
Lemon G, King JR, Byrne HM, Jensen OE, Shakesheff KM (2006) Mathematical modelling of engineered tissue growth using a multiphase porous flow mixture theory. J Math Biol 52:571–594
Lopez JI, Mouw JK, Weaver VM (2008) Biomechanical regulation of cell orientation and fate. Oncogene 27:6981–6993
Manoussaki D, Lubkin S, Vemon R, Murray J (1996) A mechanical model for the formation of vascular networks in vitro. Acta Biotheor 44(3–4):271–282
Martins-Green M, Bissell MJ (1995) Cell–ECM interactions in development. Semin Dev Biol 6:149–159
Murray JD (1993) Mathematical biology, 2nd edn. Springer, New York
Namy P, Ohayon J, Tracqui P (2004) Critical conditions for pattern formation and in vitro tubulogenesis driven by cellular traction fields. J Theor Biol 227:103–120
Nelson CM, Bissell MJ (2006) Of extracellular matrix, scaffolds, and signalling: tissue architecture regulates development, homeostasis and cancer. Ann Rev Cell Dev Biol 22:287–309
O’Dea RD, Waters SL, Byrne HM (2008) A two-fluid model for tissue growth within a dynamic flow environment. Euro J Appl Math 19(06):607–634
O’Dea RD, Waters SL, Byrne HM (2010) A multiphase model for tissue construct growth in a perfusion bioreactor. Math Med Biol 27(2):95–127
Olsen L, Maini PK, Sherratt JA, Dallon J (1999) Mathematical modelling of anisotropy in fibrous connective tissue. Math Biosci 158(2):145–170
Osborne JM, Whiteley JP (2010) A numerical method for the multiphase viscous flow equations. Comput Methods Appl Mech Eng 199:3402–3417
Oster GF, Murray JD, Harris AK (1983) Mechanical aspects of mesenchymal morphogenesis. J Embryol Exp Morphol 78:83–125
Painter KJ (2009) Modelling cell migration strategies in the extracellular matrix. J Math Biol 58:511–543
Petersen OW, Ronnov-Jessen L, Howlett AR, Bissell MJ (1992) Interaction with basement membrane serves to rapidly distinguish growth and differentiation pattern of normal and malignant human breast epithelial cells. Proc Natl Acad Sci USA 89(19):9064–9068. doi:10.1073/pnas.89.19.9064
Petrie CJS (1999) The rheology of fibre suspensions. J Non-Newton Fluid Mech 87:369–402
Peyton SR, Ghajar CM, Khatiwala CB, Putnam AJ (2007) The emergence of ECM mechanics and cytoskeletal tension as important regulators of cell function. Cell Biochem Biophys 47:300–320
Ronnov-Jessen L, Bissell MJ (2008) Breast cancer by proxy: can the microenvironment be both the cause and consequence? Trends Mol Med 15(1):5–13
Schreiber DI, Barocas VH, Tranquillo RT (2003) Temporal variations in cell migration and traction during fibroblast-mediated gel compaction. Biophys J 84:4102–4114
Soto AM, Sonnenschein C (2004) The somatic mutation theory of cancer: growing problems with the paradigm? BioEssays 26:1097–1107
Spain B (1953) Tensor calculus. Oliver and Boyd, Edinburgh
Stevenson MD, Sieminski AL, McLeod CM, Byfield FJ, Barocas VH, Gooch KJ (2010) Pericellular conditions regulate extent of cell-mediated compaction of collagen gels. Biophys J 99:19–28
Strand DW, Franco OE, Basanta D, Anderson ARA, Hayward SW (2010) Perspectives on tissue interactions in development and disease. Curr Mol Med 10:95–112
Szymanska Z, Morales-Rodrigo C, Lachowicz M, Chaplain MAJ (2009) Mathematical modelling of cancer invasion of tissue: the role and effect of nonlocal interactions. Math Models Methods Appl Sci 19(2):257–281
Takakuda K, Miyairi H (1996) Tensile behaviour of fibroblasts cultured in collagen gel. Biomaterials 17(14):1393–1397
Thompson DW (1942) On growth and form, 2nd edn. Cambridge University Press, Cambridge
Tosin A, Ambrosi D, Preziosi L (2006) Mechanics and chemotaxis in the morphogenesis of vascular networks. Bull Math Biol 68(7):1819–1836
Tranquillo RT, Murray JD (1993) Mechanistic model of wound contraction. J Surg Res 55:233–247
Vader D, Kabla A, Weitz D, Mahadevan L (2009) Strain-induced alignment in collagen gels. PLoS One 4(6):e5902. doi:10.1371/journal.pone.0005902
Weigelt B, Bissell MJ (2008) Unravelling the microenvironmental influences on the normal mammary gland and breast cancer. Semin Cancer Biol 18:311–321
Wipff PJ, Rifkin DB, Meister JJ, Hinz B (2007) Myofibroblast contraction activates latent TGF-\(\beta 1\) from the extracellular matrix. J Cell Biol 179(6):1311–1323
Acknowledgments
We thank A.M. Soto and C. Sonnenschein (Tufts University) for the initial discussions which led to the development of the model, and D.J. Smith (University of Birmingham) for assistance with aspects of the numerics. RJD gratefully acknowledges the support of the University of Birmingham’s System Science for Health initiative and the hospitality of the School of Mathematical Sciences at the University of Adelaide. JEFG is supported by a Discovery Early Career Researcher Award (DE130100031) from the Australian Research Council. The work of HMB was supported in part by award KUK-C1-013-04, made by King Abdullah University of Science and Technology (KAUST).
Author information
Authors and Affiliations
Corresponding author
Additional information
R. J. Dyson and J. E. F. Green are joint first authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (avi 18260 KB)
Supplementary material 2 (avi 19655 KB)
Supplementary material 3 (avi 18876 KB)
Appendix A: Approximation of the cell force, \(\varvec{F}_c\)
Appendix A: Approximation of the cell force, \(\varvec{F}_c\)
In this appendix, we give details of the calculation that leads to the leading-order expression for the cell force given in Eq. (14). Note that we suppress time dependence within this section for notational convenience. We begin by using the fact that \(\varvec{x}' = \varvec{x} + \eta \varvec{\xi }\), where \(\eta \ll 1\), to expand the terms in Eq. (13) which are evaluated at \(\varvec{x}'\) as follows:
where the notation \((\varvec{\xi } \cdot \nabla ) \varvec{a}\vert _{\varvec{ x}}\) is intended to emphasise the fact that the directional derivatives are evaluated at the point \(\varvec{x}\).
On integration, the contribution of the leading-order terms in the integral is zero by symmetry. Proceeding to next order, we find
where \(\hat{\varvec{\xi }} = \varvec{\xi } / | \varvec{\xi }|\), and \(\phi _n\), \(\phi _c\) and \(\varvec{a}\) are evaluated at \(\varvec{x}\) (unless otherwise stated). In component form we have
or, equivalently
where \(T_{j k l}\) is independent of \(\varvec{\xi }\), and is given by
and
Since \(A_{i j k l}\) is an isotropic integral, it must be of the form (Spain 1953)
Furthermore, since \(A_{i j k l} = A_{i k j l} = A_{i l j k}\), we deduce that
From Eqs. (36) and (37) we note that
and contracting over the remaining indices we obtain
Hence, on substituting Eq. (37) into Eq. (34), and on using the well-known properties of the Kronecker delta and the fact \(|\varvec{a}|=1\), we find
where we have assumed that \(\lambda = \eta ^{N+2} \lambda ^* = O(1)\). A little algebra then yields
Rights and permissions
About this article
Cite this article
Dyson, R.J., Green, J.E.F., Whiteley, J.P. et al. An investigation of the influence of extracellular matrix anisotropy and cell–matrix interactions on tissue architecture. J. Math. Biol. 72, 1775–1809 (2016). https://doi.org/10.1007/s00285-015-0927-7
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-015-0927-7