Abstract
Background
Oxidative stress (OS) is a strong risk factor for cardiovascular disease (CVD). The incidence of CVD is lower among kidney transplantation (KT) recipients than hemodialysis patients, and the reduction in OS may be one reason for this difference. Recently, serum sulfatides were recognized as a candidate inhibitory factor of CVD affected by OS. However, the long-term changes in OS and serum sulfatide levels in KT recipients are unknown.
Methods
We investigated the long-term changes in a serum OS marker, malondialdehyde (MDA), and the serum sulfatide levels in 17 KT recipients. Multiple regression analysis was used to analyze the factors correlated with serum sulfatide levels.
Results
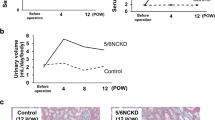
The high serum levels of MDA in the KT recipients decreased dramatically but were still high 1 year after KT surgery. MDA levels decreased further and reached near-normal levels more than 3 years after the surgery. Similarly, over the same 3 years, the low serum sulfatide levels increased to near-normal levels, reaching saturation. Multiple regression analysis showed that the most significant factors influencing serum sulfatide levels were MDA and total cholesterol content.
Conclusions
The current results show that over the long term, the internal improvement brought about by successful KT can normalize OS. Oxidative normalization was significantly correlated with the restoration of serum sulfatide levels, which were also influenced by lipoprotein metabolism. The amelioration of serum sulfatide levels might contribute to the low incidence of CVD in KT recipients.



Similar content being viewed by others
References
Sarnak MJ, Levey AS, Schoolwerth AC, Coresh J, Culleton B, Hamm LL, et al. Kidney disease as a risk factor for development of cardiovascular disease: a statement from the American Heart Association Councils on Kidney in Cardiovascular Disease, High Blood Pressure Research, Clinical Cardiology, and Epidemiology and Prevention. Circulation. 2003;108:2154–69.
Himmelfarb J, Stenvinkel P, Ikizler TA, Hakim RM. The elephant in uremia: oxidant stress as a unifying concept of cardiovascular disease in uremia. Kidney Int. 2002;62:1524–38.
Ishizuka I. Chemistry and functional distribution of sulfoglycolipids. Prog Lipid Res. 1997;36:245–319.
Hara A, Taketomi T. Occurrence of sulfatide as a major glycosphingolipid in WHHL rabbit serum lipoproteins. J Biochem. 1987;102:83–92.
Zhu XH, Hara A, Taketomi T. The existence of galactosylceramide I3-sulfate in serums of various mammals and its anticoagulant activity. J Biochem. 1991;110:241–5.
Hara A, Uemura K, Taketomi T. Sulfatide prolongs blood-coagulation time and bleeding time by forming a complex with fibrinogen. Glycoconj J. 1996;13:187–94.
Roberts DD, Rao CN, Magnani JL, Spitalnik SL, Liotta LA, Ginsburg V. Laminin binds specifically to sulfated glycolipids. Proc Natl Acad Sci USA. 1985;82:1306–10.
Roberts DD, Haverstick DM, Dixit VM, Frazier WA, Santoro SA, Ginsburg V. The platelet glycoprotein thrombospondin binds specifically to sulfated glycolipids. J Biol Chem. 1985;260:9405–11.
Roberts DD, Williams SB, Gralnick HR, Ginsburg V. Von Willebrand factor binds specifically to sulfated glycolipids. J Biol Chem. 1986;261:3306–9.
Merten M, Thiagarajan P. Role for sulfatides in platelet aggregation. Circulation. 2001;104:2955–60.
Kyogashima M. The role of sulfatide in thrombogenesis and haemostasis. Arch Biochem Biophys. 2004;426:157–62.
Hu R, Li G, Kamijo Y, Aoyama T, Nakajima T, Inoue T, et al. Serum sulfatides as a novel biomarker for cardiovascular disease in patients with end-stage renal failure. Glycoconj J. 2007;24:565–71.
Zhang X, Nakajima T, Kamijo Y, Li G, Hu R, Kannagi R, et al. Acute kidney injury induced by protein-overload nephropathy down-regulates gene expression of hepatic cerebroside sulfotransferase in mice, resulting in reduction of liver and serum sulfatides. Biochem Biophys Res Commun. 2009;390:1382–8.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53:982–92.
Li G, Hu R, Kamijo Y, Nakajima T, Aoyama T, Inoue T, et al. Establishment of a quantitative, qualitative, and high-throughput analysis of sulfatides from small amounts of sera by matrix-assisted laser desorption ionization-time of flight mass spectrometry. Anal Biochem. 2007;362:1–7.
Aoyama T, Peters JM, Iritani N, Nakajima T, Furihata K, Hashimoto T, et al. Altered constitutive expression of fatty acid-metabolizing enzymes in mice lacking the peroxisome proliferator-activated receptor alpha (PPARalpha). J Biol Chem. 1998;273:5678–84.
Aoyama T, Uchida Y, Kelley RI, Marble M, Hofman K, Tonsgard JH, et al. A novel disease with deficiency of mitochondrial very-long-chain acyl-CoA dehydrogenase. Biochem Biophys Res Commun. 1993;191:1369–72.
Aoyama T, Yamano S, Waxman DJ, Lapenson DP, Meyer UA, Fischer V, et al. Cytochrome P-450 hPCN3, a novel cytochrome P-450 IIIA gene product that is differentially expressed in adult human liver. cDNA and deduced amino acid sequence and distinct specificities of cDNA-expressed hPCN1 and hPCN3 for the metabolism of steroid hormones and cyclosporine. J Biol Chem. 1989;264:10388–95.
Tanaka M, Ishii H, Aoyama T, Takahashi H, Toriyama T, Kasuga H, et al. Ankle brachial pressure index but not brachial-ankle pulse wave velocity is a strong predictor of systemic atherosclerotic morbidity and mortality in patients on maintenance hemodialysis. Atherosclerosis. 2011;219:643–7.
Pignoli P, Tremoli E, Poli A, Oreste P, Paoletti R. Intimal plus medial thickness of the arterial wall: a direct measurement with ultrasound imaging. Circulation. 1986;74:1399–406.
de Cal M, Silva S, Cruz D, Basso F, Corradi V, Lentini P, et al. Oxidative stress and “monocyte reprogramming” after kidney transplant: a longitudinal study. Blood Purif. 2008;26:105–10.
Campise M, Bamonti F, Novembrino C, Ippolito S, Tarantino A, Cornelli U, et al. Oxidative stress in kidney transplant patients. Transplantation. 2003;76:1474–8.
Minz M, Heer M, Arora S, Sharma A, Khullar M. Oxidative status in stable renal transplantation. Transplant Proc. 2006;38:2020–1.
Antolini F, Valente F, Ricciardi D, Fagugli RM. Normalization of oxidative stress parameters after kidney transplant is secondary to full recovery of renal function. Clin Nephrol. 2004;62:131–7.
Simmons EM, Langone A, Sezer MT, Vella JP, Recupero P, Morrow JD, et al. Effect of renal transplantation on biomarkers of inflammation and oxidative stress in end-stage renal disease patients. Transplantation. 2005;79:914–9.
van den Dorpel MA, Ghanem H, Rischen-Vos J, Man in’t Veld AJ, Jansen H, Weimar W. Conversion from cyclosporine A to azathioprine treatment improves LDL oxidation in kidney transplant recipients. Kidney Int. 1997;51:1608–12.
Apanay DC, Neylan JF, Ragab MS, Sgoutas DS. Cyclosporine increases the oxidizability of low-density lipoproteins in renal transplant recipients. Transplantation. 1994;58:663–9.
Perrea DN, Moulakakis KG, Poulakou MV, Vlachos IS, Papachristodoulou A, Kostakis AI. Correlation between oxidative stress and immunosuppressive therapy in renal transplant recipients with an uneventful postoperative course and stable renal function. Int Urol Nephrol. 2006;38:343–8.
Wanner C, Quaschning T, Weingärnter K. Impact of dyslipidemia in renal transplant recipients. Curr Opin Urol. 2000;10:77–80.
Osorio A, Ortega E, de Haro T, Torres JM, Sánchez P, Ruiz-Requena E. Lipid profiles and oxidative stress parameters in male and female hemodialysis patients. Mol Cell Biochem. 2011;353:59–63.
Acknowledgments
This work was supported in part by GL Sciences (Tokyo, Japan) and the Shinshu Public Utility Foundation (Matsumoto, Japan).
Conflict of interest
The authors have declared that no conflict of interest exists.
Author information
Authors and Affiliations
Corresponding author
Additional information
Y. Kamijo and L. Wang contributed equally to this work.
About this article
Cite this article
Kamijo, Y., Wang, L., Matsumoto, A. et al. Long-term improvement of oxidative stress via kidney transplantation ameliorates serum sulfatide levels. Clin Exp Nephrol 16, 959–967 (2012). https://doi.org/10.1007/s10157-012-0634-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-012-0634-2