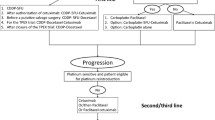
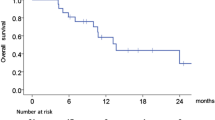
Abstract
S-1 is an oral 5-fluorouracil (5-FU) anticancer agent and has shown promising effects in the treatment of a wide range of carcinomas, including head and neck cancer. In addition to being used as adjuvant chemotherapy, S-1 is a promising agent for palliative treatment. Its ease of administration makes it an ideal drug to treat patients in the outpatient setting while maintaining adequate quality of life. However, the clinical role of S-1 in patients with recurrent/metastatic head and neck cancer is still uncertain. We retrospectively reviewed 16 patients with recurrent/metastatic head and neck cancer who received S-1 monotherapy. Thirteen patients with squamous cell carcinoma (SCC) and 3 patients with non-SCC who had recurrent/metastatic disease received S-1 monotherapy as outpatients. One patient with nasopharyngeal undifferentiated carcinoma and 1 patient with maxillary adenosquamous carcinoma showed complete response (CR), while all SCC patients showed stable disease (SD) or progressive disease (PD). Median time to progression (TTP) was 12 weeks. Five patients showed grade 3 and 4 adverse reactions, all hematological. Except for one episode of grade 4 leucopenia which required hospitalization and granulocyte colony-stimulating factor (GCSF) treatment, all adverse events resolved with dose reduction or dose omission. S-1 was safely administered in outpatients and showed some efficacy in the treatment of recurrent/metastatic head and neck cancer in patients who had received previous chemotherapy. S-1 could be used as palliative treatment in patients with recurrent/metastatic head and neck cancer.
Similar content being viewed by others
References
Parkin DM, Bray F, Ferlay J, Pisani P. Estimating the world cancer burden: Globocan 2000. Int J Cancer 2001;94:153–156.
Seiwert TY, Cohen EE: State-of-the-art management of locally advanced head and neck cancer. Br J Cancer 2005;92:1341–1348.
Alvi A, Johnson JT. Development of distant metastasis after treatment of advanced-stage head and neck cancer. Head Neck 1997;19:500–505.
Cooper JS, Pajak TF, Forastiere A, Jacobs J, Fu KK, Ang KK, et al. Precisely defining high-risk operable head and neck tumors based on RTOG #85-03 and #88-24: targets for postoperative radiochemotherapy? Head Neck 1998;20:588–594.
Kish JA, Weaver A, Jacobs J, Cummings G, Al-Sarraf M. Cisplatin and 5-fluorouracil infusion in patients with recurrent and disseminated epidermoid cancer of the head and neck. Cancer 1984;53:1819–1824.
Mercier RJ, Neal GD, Mattox DE, Gates GA, Pomeroy TC, Von Hoff DD. Cisplatin and 5-fluorouracil chemotherapy in advanced or recurrent squamous cell carcinoma of the head and neck. Cancer 1987;60:2609–2612.
Shirasaka T, Nakano K, Takechi T, Satake H, Uchida J, Fujioka A, et al. Antitumor activity of 1 M tegafur-0.4 M 5-chloro-2,4-dihydroxypyridine-1 M potassium oxonate (S-1) against human colon carcinoma orthotopically implanted into nude rats. Cancer Res 1996;56:2602–2606.
Shirasaka T, Shimamato Y, Ohshimo H, Yamaguchi M, Kato T, Yonekura K, et al. Development of a novel form of an oral 5-fluorouracil derivative (S-1) directed to the potentiation of the tumor selective cytotoxicity of 5-fluorouracil by two biochemical modulators. Anticancer Drugs 1996;7:548–557.
Schoffski P. The modulated oral fluoropyrimidine prodrug S-1, and its use in gastrointestinal cancer and other solid tumors. Anticancer Drugs 2004;15:85–106.
Sakata Y, Ohtsu A, Horikoshi N, Sugimachi K, Mitachi Y, Taguchi, T. Late phase II study of novel oral fluoropyrimidine anticancer drug S-1 (1 M tegafur-0.4 M gimestat-1 M otastat potassium) in advanced gastric cancer patients. Eur J Cancer 1998;34: 1715–1720.
Koizumi W, Kurihara M, Nakano S, Hasegawa K. Phase II study of S-1, a novel oral derivative of 5-fluorouracil, in advanced gastric cancer. For the S-1 Cooperative Gastric Cancer Study Group. Oncology 2000;58:191–197.
Ohtsu A, Baba H, Sakata Y, Mitachi Y, Horikoshi N, Sugimachi K, et al. Phase II study of S-1, a novel oral fluorophyrimidine derivative, in patients with metastatic colorectal carcinoma. S-1 Cooperative Colorectal Carcinoma Study Group. Br J Cancer 2000;83:141–145.
Nishimura G, Yanoma S, Mizuno H, Satake K, Taguchi T, Ikeda Y, et al. Therapeutic effect of 1 M tegafur-0.4 M 5-chloro-2, 4-dihydroxypridine-1 M potassium oxonate (S-1) on head and neck squamous carcinoma cells. Cancer Lett 2000;159:1–7.
Shien T, Shimizu C, Akashi-Tanaka S, Yonemori K, Kohno T, Hojo T, et al. Clinical efficacy of S-1 in pretreated metastatic breast cancer patients. Jpn J Clin Oncol 2008;38:172–175.
Tetzlaff ED, Faust J, Ajani JA. Longterm survival of a Western patient with metastatic gastric cancer treated with S-1 plus cisplatin. Gastric Cancer 2006;9:140–143.
Inuyama Y, Kida A, Tsukuda M, Kohno N, Satake B. Late phase II study of S-1 in patients with advanced head and neck cancer. Gan To Kagaku Ryoho 2001;28:1381–1390.
Pinedo HM, Peters GF. Fluorouracil: biochemistry and pharmacology. J Clin Oncol 1988;6:1653–1664.
Caballero GA, Ausman RK, Quebbeman EJ. Long-term, ambulatory, continuous IV infusion of 5-FU for the treatment of advanced adenocarcinomas. Cancer Treat Rep 1985;69:13–15.
Lokich JJ, Ahlgren JD, Gullo JJ, Philips JA, Fryer JG. A prospective randomized comparison of continuous infusion fluorouracil with a conventional bolus schedule in metastatic colorectal carcinoma: a Mid-Atlantic Oncology Program Study. J Clin Oncol 1989;7:425–432.
Quebbeman E, Ausman R, Hansen R, Becker T, Caballero G, Ritch P, et al. Long-term ambulatory treatment of metastatic colorectal adenocarcinoma by continuous intravenous infusion of 5-fluorouracil. J Surg Oncol 1985;30:60–65.
Tsukuda M, Ogasawara H, Kaneko S, Komiyama S, Horiuchi M, Inuyama Y, et al. A prospective randomized trial of adjuvant chemotherapy with UFT for head and neck carcinoma. Head and Neck UFT Study Group. Gan To Kagaku Ryoho 1994;21: 1169–1177.
Kato H, Ichinose Y, Ohta M, Hata E, Tsubota N, Tada H, et al. A randomized trial of adjuvant chemotherapy with uracil-tegafur for adenocarcinoma of the lung. N Engl J Med 2004;350:1713–1721.
Noguchi S, Koyama H, Uchino J, Abe R, Miura S, Sugimachi K, et al. Postoperative adjuvant therapy with tamoxifen, tegafur plus uracil, or both in women with node-negative breast cancer: a pooled analysis of six randomized controlled trials. J Clin Oncol 2005;23:2172–2184.
Moynihan T, Hansen R, Anderson T, Quebbeman E, Beatty P, Ausman R, et al. Continuous 5-fluorouracil infusion in advanced gastric carcinoma. Am J Clin Oncol 1988;11:461–464.
Morita S, Nakata B, Tsuji A, Mitachi Y, Shirasaka T, Saji S, et al. A phase I study of combination therapy of the oral fluorinated pyrimidine compound S-1 with low-dose cisplatin twice-a-week administration (JFMC27-9902 Step2) in patients with advanced gastric cancer using a continual reassessment method. Jpn J Clin Oncol 2007;37:924–929.
Morise Z, Sugioka A, Fujita J, Hoshimoto S, Kato T, Ikeda M. S-1 plus cisplatin combination therapy for the patients with primary liver carcinomas. Hepatogastroenterology 2007;54: 2315–2318.
Koizumi W, Narahara H, Hara T, Takagane A, Akiya T, Takagi M, et al. S-1 plus cisplatin versus S-1 alone for first-line treatment of advanced gastric cancer (SPIRITS trial): a phase III trial. Lancet Oncol 2008;9:215–221.
Kodaira T, Fuwa N, Tachibana H, Hidano S. Phase I study of S-1 and nedaplatin for patients with recurrence of head and neck cancer. Anticancer Res 2006;26:2265–2268.
Watanabe A, Taniguchi M, Yamashita T, Ueda M, Abe T, Kusumi T, et al. Phase I/II study of S-1 combined with carboplatin in recurrent and/or metastatic head and neck cancer as outpatient chemotherapy. Head Neck 2006;28:620–625.
Spano JP, Busson P, Atlan D, Bourhis J, Pignon JP, Esteban C, et al. Nasopharyngeal carcinomas: an update. Eur J Cancer 2003;39:2121–2135.
Lin JC, Jan JS, Hsu CY, Liang WM, Jiang RS, Wang WY. Phase III study of concurrent chemoradiotherapy versus radiotherapy alone for advanced nasopharyngeal carcinoma: positive effect on overall and progression-free survival. J Clin Oncol 2003;21:631–637.
Alos L, Castillo M, Nadal A, Caballero M, Mallofre C, Palacin A, et al. Adenosquamous carcinoma of the head and neck: criteria for diagnosis in a study of 12 cases. Histopathology 2004;44: 570–579.
Taguchi T, Inuyama Y, Kanamaru R, Hasegawa K, Akazawa S, Niitani H, et al. Phase I study of S-1. S-1 Study Group. Gan To Kagaku Ryoho 1997;24:2253–2264.
Tsukuda M, Kida A, Fujii M, Kono N, Yoshihara T, Hasegawa Y, et al. Randomized scheduling feasibility study of S-1 for adjuvant chemotherapy in advanced head and neck cancer. Br J Cancer 2005;93:884–889.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Suzuki, S., Ishikawa, K. Safety and efficacy of S-1 chemotherapy in recurrent/metastatic head and neck cancer. J Infect Chemother 15, 335–339 (2009). https://doi.org/10.1007/s10156-009-0712-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10156-009-0712-x