Abstract
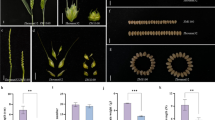
Mutations in Photoperiod-1 (Ppd-1) gene are known to modify flowering time and yield in wheat. We cloned TaPpd-1 from wheat and found high similarity among the three homoeologs of TaPpd-1. To clarify the characteristics of TaPpd-1 homoeologs in different photoperiod conditions for inflorescence architecture and yield, we used CRISPR/Cas9 system to generate Tappd-1 mutant plants by simultaneous modification of the three homoeologs of wheat Ppd-1. Tappd-1 mutant plants showed no off-target mutations. Four T0-edited lines under short-day length and three lines under long-day length conditions with the mutation frequency of 25% and 21%, respectively. These putative transgenic plants of all the lines were self-fertilized and generated T1 and T2 progenies and were evaluated by phenotypic and expression analysis. Results demonstrated that simultaneously edited TaPpd-1- A1, B1, and D1 homoeologs gene copies in T2_SDL-8-4, T2_SDL-4-5, T2_SDL-3-9, and T2_LDL-10-9 showed similar spike inflorescence, flowering time, and significantly increase in 1000-grain weight, grain area, grain width, grain length, plant height, and spikelets per spike due to mutation in both alleles of Ppd-B1 and Ppd-D1 homoeologs but only spike length was decreased in T2_SDL-8-4, T2_SDL-4-5, and T2_LDL-13-3 mutant lines due to mutation in both alleles of Ppd-A1 homoeolog under both conditions. Our results indicate that all TaPpd1 gene homoeologs influence wheat spike development by affecting both late flowering and earlier flowering but single mutant TaPpd-A1 homoeolog affect lowest as compared to the combination with double mutants of TaPpd-B1 and TaPpd-D1, TaPpd-A1 and TaPpd-B1, and TaPpd-A1 and TaPpd-D1 homoeologs for yield enhancement. Our findings further raised the idea that the relative expression of the various genomic copies of TaPpd-1 homoeologs may have an impact on the spike inflorescence architecture and grain morphometric features in wheat cultivars.






Similar content being viewed by others
Data availability
All the data is presented in the main text and supplementary files.
References
Abe F, Haque E, Hisano H, Tanaka T, Kamiya Y, Mikami M, Kawaura K, Endo M, Onishi K, Hayashi T (2019) Genome-edited triple-recessive mutation alters seed dormancy in wheat. Cell Rep 28:1362–1369
Achilli AL, Roncallo PF, Larsen AO, Dreisigacker S, Echenique V (2022) Population structure, allelic variation at Rht-B1 and Ppd-A1 loci and its effects on agronomic traits in Argentinian durum wheat. Sci Rep 12:9629
Amo A, Serikbay D, Song L, Chen L, Hu Y-G (2022) Photoperiod and vernalization alleles and their combinations greatly affected phenological and agronomic traits in bread wheat under autumn and spring sowing conditions. BioRxiv 15:2022–2005
Arjona JM, Royo C, Dreisigacker S, Ammar K, Villegas D (2018) Effect of Ppd-A1 and Ppd-B1 allelic variants on grain number and thousand kernel weight of durum wheat and their impact on final grain yield. Front Plant Sci 9:888
Beales J, Turner A, Griffiths S, Snape JW, Laurie DA (2007) A pseudo-response regulator is misexpressed in the photoperiod insensitive Ppd-D1a mutant of wheat (Triticum aestivum L.). Theor Appl Genet 115:721–733
Bentley A, Turner A, Gosman N, Leigh F, Maccaferri M, Dreisigacker S, Greenland A, Laurie D (2011) Frequency of photoperiod-insensitive Ppd-A1a alleles in tetraploid, hexaploid and synthetic hexaploid wheat germplasm. Plant Breed 130:10–15
Bergeret P (2017) The future of food and agriculture: Trends and challenges [Note de lecture]. Bibliog Pros 29(09):3
Boden SA, Cavanagh C, Cullis BR, Ramm K, Greenwood J, Jean Finnegan E, Trevaskis B, Swain SM (2015) Ppd-1 is a key regulator of inflorescence architecture and paired spikelet development in wheat. Nat Plants 1:1–6
Borrill P, Ramirez-Gonzalez R, Uauy C (2016) expVIP: a customizable RNA-seq data analysis and visualization platform. Plant Physiol 170:2172–2186
Bortiri E, Chuck G, Vollbrecht E, Rocheford T, Martienssen R, Hake S (2006) ramosa2 encodes a lateral organ boundary domain protein that determines the fate of stem cells in branch meristems of maize. Plant Cell 18:574–585
Brown RH, Bregitzer P (2011) A Ds insertional mutant of a barley miR172 gene results in indeterminate spikelet development. Crop Sci 51:1664–1672
Budak H, Appels R, Paux E (2021) Insights on decoding wheat and barley genomes. Func Integ Genom 21:157–159
Chen L, Du Y, Lu Q, Chen H, Meng R, Cui C, Lu S, Yang Y, Chai Y, Li J (2018) The photoperiod-insensitive allele Ppd-D1a promotes earlier flowering in Rht12 dwarf plants of bread wheat. Front Plant Sci 9:1312
Choudhury S, Muthusamy SK, Padaria JC, Dalal M (2021) Genome-wide identification of Ran GTPase family genes from wheat (T. aestivum) and their expression profile during developmental stages and abiotic stress conditions. Func Integ Genom 21:239–250
Chuck G, Meeley R, Hake S (2008) Floral meristem initiation and meristem cell fate are regulated by the maize AP2 genes ids1 and sid1. Development (Cambridge, England) 135(18):3013–3019. https://doi.org/10.1242/dev.024273
Cram D, Kulkarni M, Buchwaldt M, Rajagopalan N, Bhowmik P, Rozwadowski K, Parkin IA, Sharpe AG, Kagale S (2019) WheatCRISPR: a web-based guide RNA design tool for CRISPR/Cas9-mediated genome editing in wheat. BMC Plant Biol 19:1–8
Debernardi JM, Lin H, Chuck G, Faris JD, Dubcovsky J (2017) microRNA172 plays a crucial role in wheat spike morphogenesis and grain threshability. Development 144:1966–1975
Dixon LE, Greenwood JR, Bencivenga S, Zhang P, Cockram J, Mellers G, Ramm K, Cavanagh C, Swain SM, Boden SA (2018) TEOSINTE BRANCHED1 regulates inflorescence architecture and development in bread wheat (Triticum aestivum). Plant Cell 30:563–581
Eaton WM (2021) Understanding the regulation of flowering time through PHOTOPERIOD-1 (Ppd-1) and FLOWERING LOCUS T (FT) genes in bread wheat. MSc by research thesis, University of Leeds
Engler C, Kandzia R, Marillonnet S (2008) A one pot, one step, precision cloning method with high throughput capability. PloS One 3:e3647
Gallavotti A, Long JA, Stanfield S, Yang X, Jackson D, Vollbrecht E, Schmidt RJ (2010) The control of axillary meristem fate in the maize ramosa pathway. Development 137:2849–2856
Gasperini D, Greenland A, Hedden P, Dreos R, Harwood W, Griffiths S (2012) Genetic and physiological analysis of Rht8 in bread wheat: an alternative source of semi-dwarfism with a reduced sensitivity to brassinosteroids. J Ex Botany 63:4419
Gauley A, Boden SA (2021) Stepwise increases in FT1 expression regulate seasonal progression of flowering in wheat (Triticum aestivum). New Phytol 229:1163–1176
Greenwood JR, Finnegan EJ, Watanabe N, Trevaskis B, Swain SM (2017) New alleles of the wheat domestication gene Q reveal multiple roles in growth and reproductive development. Development 144:1959–1965
Guo Z, Song Y, Zhou R, Ren Z, Jia J (2010) Discovery, evaluation and distribution of haplotypes of the wheat Ppd-D1 gene. New Phytol 185:841–851
Houston K, McKim SM, Comadran J, Bonar N, Druka I, Uzrek N, Cirillo E, Guzy-Wrobelska J, Collins NC, Halpin C (2013) Variation in the interaction between alleles of HvAPETALA2 and microRNA172 determines the density of grains on the barley inflorescence. Proc Nat Acad Sci 110:16675–16680
Ishida Y, Tsunashima M, Hiei Y, Komari T (2015) Wheat (Triticum aestivum L.) transformation using immature embryos. In: Agrobacterium protocols. Springer, pp 189–198
Jaeger KE, Wigge PA (2007) FT protein acts as a long-range signal in Arabidopsis. Curr Biol 17:1050–1054
Kamran A, Iqbal M, Spaner D (2014) Flowering time in wheat (Triticum aestivum L.): a key factor for global adaptability. Euphytica 197:1–26
Kim D, Hager M, Brant E, Budak H (2021) Efficient genome editing in wheat using Cas9 and Cpf1 (AsCpf1 and LbCpf1) nucleases. Funct Integr Genomics 21:355–366
Komatsu M, Chujo A, Nagato Y, Shimamoto K, Kyozuka J (2003) FRIZZY PANICLE is required to prevent the formation of axillary meristems and to establish floral meristem identity in rice spikelets. Development (Cambridge, England) 130(16):3841–3850. https://doi.org/10.1242/dev.00564
Lee DY, An G (2012) Two AP2 family genes, supernumerary bract (SNB) and Osindeterminate spikelet 1 (OsIDS1), synergistically control inflorescence architecture and floral meristem establishment in rice. Plant J 69:445–461
Li C, Dubcovsky J (2008) Wheat FT protein regulates VRN1 transcription through interactions with FDL2. Plant J 55:543–554
Li M, Li X, Zhou Z, Wu P, Fang M, Pan X, Lin Q, Luo W, Wu G, Li H (2016) Reassessment of the four yield-related genes Gn1a, DEP1, GS3, and IPA1 in rice using a CRISPR/Cas9 system. Front Plant Sci 7:377
Liang Z, Chen K, Li T, Zhang Y, Wang Y, Zhao Q, Liu J, Zhang H, Liu C, Ran Y (2017) Efficient DNA-free genome editing of bread wheat using CRISPR/Cas9 ribonucleoprotein complexes. Nat Commun 8:1–5
Liu H, Wang K, Tang H, Gong Q, Du L, Pei X, Ye X (2020a) CRISPR/Cas9 editing of wheat TaQ genes alters spike morphogenesis and grain threshability. J Gen Genomics 47:563–575
Liu Y, Zhang L, Melzer M, Shen L, Sun Z, Wang Z, Schnurbusch T, Guo Z (2020b) Ppd-1 Remodels Spike Architecture by Regulating Floral Development in wheat. BioRxiv 12:2020–2005
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25:402–408
Ma X, Zhang Q, Zhu Q, Liu W, Chen Y, Qiu R, Wang B, Yang Z, Li H, Lin Y (2015) A robust CRISPR/Cas9 system for convenient, high-efficiency multiplex genome editing in monocot and dicot plants. Mole Plant 8:1274–1284
Maccaferri M, Sanguineti MC, Corneti S, Ortega JLA, Salem MB, Bort J, DeAmbrogio E, del Moral LFG, Demontis A, El-Ahmed A (2008) Quantitative trait loci for grain yield and adaptation of durum wheat (Triticum durum Desf.) across a wide range of water availability. Genetics 178:489–511
Maphosa L, Langridge P, Taylor H, Parent B, Emebiri LC, Kuchel H, Reynolds MP, Chalmers KJ, Okada A, Edwards J (2014) Genetic control of grain yield and grain physical characteristics in a bread wheat population grown under a range of environmental conditions. Theor Appl Gen 127:1607–1624
McSteen P, Laudencia-Chingcuanco D, Colasanti J (2000) A floret by any other name: control of meristem identity in maize. Trends In Plant Sci 5:61–66
Meyer RS, Purugganan MD (2013) Evolution of crop species: genetics of domestication and diversification. Nat Rev Gen 14:840–852
Miralles D, Richards R (2000) Responses of leaf and tiller emergence and primordium initiation in wheat and barley to interchanged photoperiod. Ann Bot 85:655–663
Nishida H, Yoshida T, Kawakami K, Fujita M, Long B, Akashi Y, Laurie DA, Kato K (2013) Structural variation in the 5′ upstream region of photoperiod-insensitive alleles Ppd-A1a and Ppd-B1a identified in hexaploid wheat (Triticum aestivum L.), and their effect on heading time. Mol Breeding 31:27–37
Pan C, Ye L, Qin L, Liu X, He Y, Wang J, Chen L, Lu G (2016) CRISPR/Cas9-mediated efficient and heritable targeted mutagenesis in tomato plants in the first and later generations. Sci Rep 6:1–9
Park SJ, Jiang K, Tal L, Yichie Y, Gar O, Zamir D, Eshed Y, Lippman ZB (2014) Optimization of crop productivity in tomato using induced mutations in the florigen pathway. Nat Gen 46:1337–1342
Pearce S, Vazquez-Gross H, Herin SY, Hane D, Wang Y, Gu YQ, Dubcovsky J (2015) WheatExp: an RNA-seq expression database for polyploid wheat. BMC Plant Biol 15:1–8
Ramírez-González R, Borrill P, Lang D, Harrington S, Brinton J, Venturini L, Davey M, Jacobs J, Van Ex F, Pasha A (2018) The transcriptional landscape of polyploid wheat. Science 361:eaar6089
Sánchez-León S, Gil-Humanes J, Ozuna CV, Giménez MJ, Sousa C, Voytas DF, Barro F (2018) Low-gluten, nontransgenic wheat engineered with CRISPR/Cas9. Plant Biotechnol J 16:902–910
Sentmanat MF, Peters ST, Florian CP, Connelly JP, Pruett-Miller SM (2018) A survey of validation strategies for CRISPR-Cas9 editing. Sci Rep 8:1–8
Shaw LM, Turner AS, Laurie DA (2012) The impact of photoperiod insensitive Ppd-1a mutations on the photoperiod pathway across the three genomes of hexaploid wheat (Triticum aestivum). Plant J 71:71–84
Shaw LM, Turner AS, Herry L, Griffiths S, Laurie DA (2013) Mutant alleles of Photoperiod-1 in wheat (Triticum aestivum L.) that confer a late flowering phenotype in long days. PLoS One 8:e79459
Shen L, Wang C, Fu Y, Wang J, Liu Q, Zhang X, Yan C, Qian Q, Wang K (2018) QTL editing confers opposing yield performance in different rice varieties. J Integr Plant Biol 60:89–93
Soyk S, Lemmon ZH, Oved M, Fisher J, Liberatore KL, Park SJ, Goren A, Jiang K, Ramos A, van der Knaap E (2017) Bypassing negative epistasis on yield in tomato imposed by a domestication gene. Cell 169:1142–1155
Tang X, Zheng X, Qi Y, Zhang D, Cheng Y, Tang A, Voytas DF, Zhang Y (2016) A single transcript CRISPR-Cas9 system for efficient genome editing in plants. Mol Plant 9:1088–1091
Tanio M, Kato K (2007) Development of near-isogenic lines for photoperiod-insensitive genes, Ppd-B1 and Ppd-D1, carried by the Japanese wheat cultivars and their effect on apical development. Breeding Sci 57:65–72
Tiwari SB, Shen Y, Chang HC, Hou Y, Harris A, Ma SF, McPartland M, Hymus GJ, Adam L, Marion C (2010) The flowering time regulator CONSTANS is recruited to the FLOWERING LOCUS T promoter via a unique cis-element. New Phytol 187:57–66
Turner A, Beales J, Faure S, Dunford RP, Laurie DA (2005) The pseudo-response regulator Ppd-H1 provides adaptation to photoperiod in barley. Science 310:1031–1034
Varkonyi-Gasic E, Lough RH, Moss S, Wu R, Hellens RP (2012) Kiwifruit floral gene APETALA2 is alternatively spliced and accumulates in aberrant indeterminate flowers in the absence of miR172. Plant Mol Biol 78:417–429
Villegas D, Alfaro C, Ammar K, Cátedra M, Crossa J, García del Moral L, Royo C (2016) Daylength, temperature and solar radiation effects on the phenology and yield formation of spring durum wheat. J Agron Crop Sci 202:203–216
Wang Y, Li J (2008) Molecular basis of plant architecture. Annual Rev Plant Biol 59:253
Wang L, Sun S, Jin J, Fu D, Yang X, Weng X, Xu C, Li X, Xiao J, Zhang Q (2015) Coordinated regulation of vegetative and reproductive branching in rice. Proc Nat Acad Sci 112:15504–15509
Wang W, Simmonds J, Pan Q, Davidson D, He F, Battal A, Akhunova A, Trick HN, Uauy C, Akhunov E (2018) Gene editing and mutagenesis reveal inter-cultivar differences and additivity in the contribution of TaGW2 homoeologues to grain size and weight in wheat. Theor Appl Gen 131:2463–2475
Wang W, Pan Q, Tian B, He F, Chen Y, Bai G, Akhunova A, Trick HN, Akhunov E (2019) Gene editing of the wheat homologs of TONNEAU 1-recruiting motif encoding gene affects grain shape and weight in wheat. Plant J 100:251–264
Wilhelm EP, Turner AS, Laurie DA (2009) Photoperiod insensitive Ppd-A1a mutations in tetraploid wheat (Triticum durum Desf.). Theor Appl Gen 118:285–294
Wilhelm EP, Boulton MI, Al-Kaff N, Balfourier F, Bordes J, Greenland AJ, Powell W, Mackay IJ (2013) Rht-1 and Ppd-D1 associations with height, GA sensitivity, and days to heading in a worldwide bread wheat collection. Theor Appl Gen 126:2233–2243
Wu Y, Liu J, Hu G, Xue H, Xu H, Zhao C, Qin R, Cui F, Sun H (2021a) Functional Analysis of the “Green Revolution” Gene Photoperiod-1 and Its Selection Trends During Bread Wheat Breeding. Front Plant Sci 12:745411
Wu Y, Liu J, Hu G, Xue H, Xu H, Zhao C, Qin R, Cui F, Sun H (2021b) Functional analysis of the “Green Revolution” gene Photoperiod-1 and its selection trends during bread wheat breeding. Front Plant Sci 12:745411
Xiao A, Cheng Z, Kong L, Zhu Z, Lin S, Gao G, Zhang B (2014) CasOT: a genome-wide Cas9/gRNA off-target searching tool. Bioinformatics 30:1180–1182
Yoshida A, Sasao M, Yasuno N, Takagi K, Daimon Y, Chen R, Yamazaki R, Tokunaga H, Kitaguchi Y, Sato Y (2013) TAWAWA1, a regulator of rice inflorescence architecture, functions through the suppression of meristem phase transition. Proc Nat Acad Sci 110:767–772
Zadoks JC, Chang TT, Konzak CF (1974) A decimal code for the growth stages of cereals. Weed Res 14:415–421
Zhang D, Yuan Z (2014) Molecular control of grass inflorescence development. Annual Rev Plant Biol 65:553–578
Zhang W, Zhao G, Gao L, Kong X, Guo Z, Wu B, Jia J (2016a) Functional studies of heading date-related gene TaPRR73, a paralog of Ppd1 in common wheat. Front Plant Sci 7:772. https://doi.org/10.3389/fpls.2016.00772
Zhang Y, Liang Z, Zong Y, Wang Y, Liu J, Chen K, Qiu J-L, Gao C (2016b) Efficient and transgene-free genome editing in wheat through transient expression of CRISPR/Cas9 DNA or RNA. Nat Commun 7:1–8
Zhang Y, Bai Y, Wu G, Zou S, Chen Y, Gao C, Tang D (2017) Simultaneous modification of three homoeologs of Ta EDR 1 by genome editing enhances powdery mildew resistance in wheat. Plant J 91:714–724
Zhang Z, Hua L, Gupta A, Tricoli D, Edwards KJ, Yang B, Li W (2019) Development of an Agrobacterium-delivered CRISPR/Cas9 system for wheat genome editing. Plant Biotechnol J 17:1623–1635
Zhao L, Nakazawa M, Takase T, Manabe K, Kobayashi M, Seki M, Shinozaki K, Matsui M (2004) Overexpression of LSH1, a member of an uncharacterised gene family, causes enhanced light regulation of seedling development. Plant J 37:694–706
Acknowledgements
Deeply thankful to the Functional Genomics Lab. at National Institute for Genomics and Advanced Biotechnology (NIGAB) for providing research facilities.
Funding
This research was conducted from the funds of the Agricultural Linkages Program of Pakistan (ALP # CS-551) entitled “Yield enhancement of wheat through improved grain number and spikelet architecture using translational genomics and genome editing approaches”.
Author information
Authors and Affiliations
Contributions
AE and MRK conceptualized the study. AE, NR, and SI performed the research and prepared the initial draft. MU visualized and updated the figures. AE, NR, SI, GMA, MU, and MRK helped in the revised write-up of the original draft. All authors carefully read and approved the article for submission.
Corresponding authors
Ethics declarations
Ethical approval and consent to participate
Not applicable.
Consent to publish
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1:
Supplementary Table S1. Primers used for gene isolation in three homoeologs of TaPpd-1 gene. Supplementary Table S2. Conserved sgRNAs oligonucleotides from A, B, and D genome of TaPpd-1 gene. Supplementary Table S3. Primers used in this study for vector construction and confirmation of CRISPR free plants. Supplementary Table S4. PCR primers used for sequences analysis after mutation in three separate homoeologs of TaPpd-1 gene. Supplementary Table S5. Primer used for Gene expression analysis in three homoeologs of TaPpd-1 gene. Supplementary Table S6. Genotypes of the 20 T0 TaPpd-1 mutants with respect to mutations in the TaPpd-A1, TaPpd-B1, and TaPpd-D1 homoalleles under short-day conditions. Supplementary Table S7. Genotypes of the 16 T0 TaPpd-1 mutants with respect to mutations in the TaPpd-A1, TaPpd-B1, and TaPpd-D1 homoalleles under long-day conditions. Supplementary Table S8. The thousand grain weight spike and seed morphometric parameters for CRISPR-Cas9 induced TaPpd-1 gene mutants under short-day conditions. Supplementary Table S9. The thousand grain weight spike and seed morphometric parameters for CRISPR-Cas9 induced TaPpd-1 gene mutants under long-day condition. Supplementary Figure S1. Multiple sequence alignment of the coding region of wheat Ppd-A1, B1, and D1 homoeologs. Supplementary Figure S2. Multiple sequence alignments of new TaPpd-1 proteins and reference proteins were generated in Bio-Edit software using ClustalW program. Orange line indicates PRR domain and CCT domain. Supplementary Figure S3. PCR amplification from the progenies of T1 mutant lines using the Cas9 specific primers. M: DNA molecular marker, C: T0 plants serve as a positive control. Positive sign indicates CRISPR free plants.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Errum, A., Rehman, N., Uzair, M. et al. CRISPR/Cas9 editing of wheat Ppd-1 gene homoeologs alters spike architecture and grain morphometric traits. Funct Integr Genomics 23, 66 (2023). https://doi.org/10.1007/s10142-023-00989-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10142-023-00989-2