Abstract
Thermal stress can pose a major challenge to salmonid fish. A 4x44K oligonucleotide microarray approach was used to screen for genetically determined variations of a temperature stress response during acclimation in fish gills, a highly specialized and complex organ responsible for gas and electrolyte exchange as well as excretion. The comparison addressed transcriptional changes in the local breeding strain BORN and imported (TCO) rainbow trout after graded 2-week acclimation to 8 and 23 °C. Besides well-characterized mediators of thermoregulation such as genes encoding cold-inducible RNA-binding protein and heat shock proteins, the present microarray study suggests several new candidate genes commonly regulated in gills of the two trout lines. Having identified the differential expression of thermoregulated genes as duplicated paralogues, they were subsequently validated in a gill cell model. Moreover, the comparison of transcriptome profiles provides evidence for distinctively employed expression patterns. The induction of genes encoding factors of the early innate immunity in BORN trout upon warming contrasts with the increased expression of adaptive immune genes in import trout. Cold acclimation induced genes assigned to the functional categories “cell death” and “ion channel activity” in import trout, but repressed “lipid metabolism.” This manuscript provides an overview of the genes of the multifunctional gills in rainbow trout that are mandated after temperature change, suggesting links between the different temperature-dependent pathways and gene networks.




Similar content being viewed by others
References
Amara U, Flierl MA, Rittirsch D, Klos A, Chen H, Acker B, Brueckner UB, Nilsson B, Gebhard F, Lambris JD, Huber-Lang M (2010) Molecular intercommunication between the complement and coagulation systems. J Immunol 185:5628–5636
Anders E (1986) Stand der Zuechtung und Reproduktion brackwasseradaptierter Regenbogenforellenbestaende im Kuestenbereich der DDR. Fischerei-Forschung 24:72
Asea A (2008) Heat shock proteins and toll-like receptors. Handb Exp Pharmacol 111–127
Bailey GS, Poulter RTM, Stockwell PA (1978) Gene duplication in tetraploid fish: model for gene silencing at unlinked duplicated loci. Proc Natl Acad Sci U S A 75:5575–5579
Bols NC, Mosser DD, Steels GB (1992) Temperature studies and recent advances with fish cells in vitro. Comp Biochem Physiol B Biochem Mol Biol 103:1–14
Bols NC, Barlian A, Chirinotrejo M, Caldwell SJ, Goegan P, Lee LEJ (1994) Development of a cell-line from primary cultures of rainbow trout, Oncorhynchus mykiss (Walbaum), gills. J Fish Dis 17:601–611
Bowden TJ (2008) Modulation of the immune system of fish by their environment. Fish Shellfish Immunol 25:373–383
Bowden TJ, Thompson KD, Morgan AL, Gratacap RML, Nikoskelainen S (2007) Seasonal variation and the immune response: a fish perspective. Fish Shellfish Immunol 22:695–706
Bowers RM, Lapatra SE, Dhar AK (2008) Detection and quantitation of infectious pancreatic necrosis virus by real-time reverse transcriptase-polymerase chain reaction using lethal and non-lethal tissue sampling. J Virol Methods 147:226–234
Brett JR (1971) Energetic responses of salmon to temperature—study of some thermal relations in physiology and freshwater ecology of sockeye salmon (Oncorhynchus nerka). Am Zool 11:99
Breves JP, Fox BK, Pierce AL, Hirano T, Grau EG (2010) Gene expression of growth hormone family and glucocorticoid receptors, osmosensors, and ion transporters in the gill during seawater acclimation of Mozambique tilapia, Oreochromis mossambicus. J Exp Zool A 13A:432–441
Brunelli JP, Wertzler KJ, Sundin K, Thorgaard GH (2008) Y-specific sequences and polymorphisms in rainbow trout and Chinook salmon. Genome 51:739–748
Buckley BA, Gracey AY, Somero GN (2006) The cellular response to heat stress in the goby Gillichthys mirabilis: a cDNA microarray and protein-level analysis. J Exp Biol 209:2660–2677
Clague MJ, Urbe S (2010) Ubiquitin: same molecule, different degradation pathways. Cell 143:682–685
Craig EA, Gross CA (1991) Is hsp70 the cellular thermometer? Trends Biochem Sci 16:135–140
David NB, Sapede D, Saint-Etienne L, Thisse C, Thisse B, Dambly-Chaudiere C, Rosa FM, Ghysen A (2002) Molecular basis of cell migration in the fish lateral line: role of the chemokine receptor CXCR4 and of its ligand, SDF1. Proc Natl Acad Sci U S A 99:16297–16302
Engelsma MY, Hougee S, Nap D, Hofenk M, Rombout JHWM, van Muiswinkel WB, Verburg-van Kemenade BML (2003) Multiple acute temperature stress affects leucocyte populations and antibody responses in common carp, Cyprinus carpio L. Fish Shellfish Immunol 15:397–410
Evans DH, Piermarini PM, Choe KP (2005) The multifunctional fish gill: dominant site of gas exchange, osmoregulation, acid-base regulation, and excretion of nitrogenous waste. Physiol Rev 85:97–177
Fevolden SE, Refstie T, Roed KH (1992) Disease resistance in rainbow trout (Oncorhynchus mykiss) selected for stress response. Aquaculture 104:19–29
Godard CAJ, Goldstone JV, Said MR, Dickerson RL, Woodin BR, Stegeman JJ (2005) The new vertebrate CYP1C family: cloning of new subfamily members and phylogenetic analysis. Biochem Biophys Res Commun 331:1016–1024
Goss GG, Perry SF, Wood CM, Laurent P (1992) Mechanisms of ion and acid-base regulation at the gills of fresh-water fish. J Exp Zool 263:143–159
Gracey AY, Fraser EJ, Li WZ, Fang YX, Taylor RR, Rogers J, Brass A, Cossins AR (2004) Coping with cold: an integrative, multitissue analysis of the transcriptome of a poikilothermic vertebrate. Proc Natl Acad Sci U S A 101:16970–16975
Hartl FU, Hayer-Hartl M (2002) Molecular chaperones in the cytosol: from nascent chain to folded protein. Science 295:1852–1858
Hazel JR (1995) Thermal adaptation in biological-membranes—is homeoviscous adaptation the explanation. Annu Rev Physiol 57:19–42
Hokanson KEF, Kleiner CF, Thorslund TW (1977) Effects of constant temperatures and diel temperature-fluctuations on specific growth and mortality-rates and yield of juvenile rainbow trout, Salmo gairdneri. J Fish Res Board Can 34:639–648
Hori TS, Gamperl AK, Afonso LO, Johnson SC, Hubert S, Kimball J, Bowman S, Rise ML (2010) Heat-shock responsive genes identified and validated in Atlantic cod (Gadus morhua) liver, head kidney and skeletal muscle using genomic techniques. BMC Genomics 11:72
Huang DW, Sherman BT, Lempicki RA (2009) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4:44–57
Hurley IA, Mueller RL, Dunn KA, Schmidt EJ, Friedman M, Ho RK, Prince VE, Yang Z, Thomas MG, Coates MI (2007) A new time-scale for ray-finned fish evolution. Proc R Soc A Biol Sci 274:489–498
Jima DD, Shah RN, Orcutt TM, Joshi D, Law J, Litman GW, Trede NS, Yoder JA (2009) Enhanced transcription of complement and coagulation genes in the absence of adaptive immunity. Mol Immunol 46:1505–1516
Ju Z, Dunham RA, Liu Z (2002) Differential gene expression in the brain of channel catfish (Ictalurus punctatus) in response to cold acclimation. Mol Gen Genomics 268:87–95
Kassahn KS, Caley M, Ward AC, Connolly AR, Stone G, Crozier RH (2007) Heterologous microarray experiments used to identify the early gene response to heat stress in a coral reef fish. Mol Ecol 16:1749–1763
Keefer ML, Peery CA, High B (2009) Behavioral thermoregulation and associated mortality trade-offs in migrating adult steelhead (Oncorhynchus mykiss): variability among sympatric populations. Can J Fish Aquat Sci 66:1734–1747
Koban M (1986) Can cultured teleost hepatocytes show temperature-acclimation? Am J Physiol 250:R211–R220
Koide T, Asada S, Nagata K (1999) Substrate recognition of collagen-specific molecular chaperone HSP47—structural requirements and binding regulation. J Biol Chem 274:34523–34526
Kopp A, Bala M, Buechler C, Falk W, Gross P, Neumeier M, Scholmerich J, Schaffler A (2010) C1q/TNF-related protein-3 represents a novel and endogenous lipopolysaccharide antagonist of the adipose tissue. Endocrinology 151:5267–5278
Koppang EO, Fischer U, Moore L, Tranulis MA, Dijkstra JM, Koellner B, Aune L, Jirillo E, Hordvik I (2010) Salmonid T cells assemble in the thymus, spleen and in novel interbranchial lymphoid tissue. J Anat 217:728–739
Kothary RK, Candido EPM (1982) Induction of a novel set of polypeptides by heat-shock or sodium arsenite in cultured-cells of rainbow trout, Salmo gairdnerii. Can J Biochem 60:347–355
Lee L, Dayeh V, Schirmer K, Bols N (2009) Applications and potential uses of fish gill cell lines: examples with RTgill-W1. In Vitro Cell Dev An 45:127–134
Lindquist S (1986) The heat-shock response. Annu Rev Biochem 55:1151–1191
Liu M, Tee C, Zeng F, Sherry JP, Dixon B, Bols NC, Duncker BP (2011) Characterization of p53 expression in rainbow trout. Comp Biochem Physiol C Toxicol Pharmacol 154:326–332
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(T)(−Delta Delta C) method. Methods 25:402–408
Mackay WC (1974) Effect of Temperature on Osmotic and Ionic Regulation in Goldfish, Carassius auratus L. J Comp Physiol 88:1–19
Matthews KR, Berg NH (1997) Rainbow trout responses to water temperature and dissolved oxygen stress in two southern California stream pools. J Fish Biol 50:50–67
Mosser DD, Heikkila JJ, Bols NC (1986) Temperature ranges over which rainbow trout fibroblasts survive and synthesize heat-shock proteins. J Cell Physiol 128:432–440
Murata M, Kodama H, Onuma M (1995) Characterization of rainbow trout C-polysaccharide binding proteins. J Vet Med Sci 57:419–425
Nikoskelainen S, Bylund G, Lilius EM (2004) Effect of environmental temperature on rainbow trout (Oncorhynchus mykiss) innate immunity. Dev Comp Immunol 28:581–592
Nishiyama H, Danno S, Kaneko Y, Itoh K, Yokoi H, Fukumoto M, Okuno H, Millan JL, Matsuda T, Yoshida O, Fujita J (1998) Decreased expression of cold-inducible RNA-binding protein (CIRP) in male germ cells at elevated temperature. Am J Pathol 152:289–296
Ojima N, Yamashita M, Watabe S (2005) Quantitative mRNA expression profiling of heat-shock protein families in rainbow trout cells. Biochem Biophys Res Commun 329:51–57
Pan F, Zarate J, Choudhury A, Rupprecht R, Bradley TM (2004) Osmotic stress of salmon stimulates upregulation of a cold inducible RNA binding protein (CIRP) similar to that of mammals and amphibians. Biochimie 86:451–461
Parsell DA, Lindquist S (1993) The function of heat-shock proteins in stress tolerance: degradation and reactivation of damaged proteins. Annu Rev Genet 27:437–496
Podrabsky JE, Somero GN (2004) Changes in gene expression associated with acclimation to constant temperatures and fluctuating daily temperatures in an annual killifish Austrofundulus limnaeus. J Exp Biol 207:2237–2254
Portner HO (2001) Climate change and temperature-dependent biogeography: oxygen limitation of thermal tolerance in animals. Naturwissenschaften 88:137–146
Qian Y, Zhang J, Yan B, Chen X (2008) DEC1, a basic helix-loop-helix transcription factor and a novel target gene of the p53 family, mediates p53-dependent premature senescence. J Biol Chem 283:2896–2905
Quinn NL, McGowan CR, Cooper GA, Koop BF, Davidson WS (2011a) Identification of genes associated with heat tolerance in Arctic charr exposed to acute thermal stress. Physiol Genomics 43:685–696
Quinn NL, McGowan CR, Cooper GA, Koop BF, Davidson WS (2011b) Ribosomal genes and heat shock proteins as putative markers for chronic, sub-lethal heat stress in Arctic charr: applications for aquaculture and wild fish. Physiol Genomics 43:1056–1064
Raida MK, Buchmann K (2007) Temperature-dependent expression of immune-relevant genes in rainbow trout following Yersinia ruckeri vaccination. Dis Aquat Org 77:41–52
Rebl A, Siegl E, Koellner B, Fischer U, Seyfert HM (2007) Characterization of twin toll-like receptors from rainbow trout (Oncorhynchus mykiss): evolutionary relationship and induced expression by Aeromonas salmonicida salmonicida. Dev Comp Immunol 31:499–510
Rebl A, Anders E, Wimmers K, Goldammer T (2009) Cloning and tissue-specific expression of a delta-COP homologue in a freshwater and a brackish water-adapted strain of rainbow trout (Oncorhynchus mykiss). Genes Genet Syst 84:239–243
Rebl A, Koebis JM, Fischer U, Takizawa F, Verleih M, Wimmers K, Goldammer T (2011) MARCH5 gene is duplicated in rainbow trout, but only fish-specific gene copy is up-regulated after VHSV infection. Fish Shellfish Immunol 31:1041–1050
Rebl A, Verleih M, Korytar T, Kuehn C, Wimmers K, Koellner B, Goldammer T (2012) Identification of differentially expressed protective genes in liver of two rainbow trout strains. Vet Immunol Immunopathol 145:305–315
Richards RC, Short CE, Driedzic WR, Ewart KV (2010) Seasonal changes in hepatic gene expression reveal modulation of multiple processes in rainbow smelt (Osmerus mordax). Mar Biotechnol 12:650–663
Roberts RJ, Agius C, Saliba C, Bossier P, Sung YY (2010) Heat shock proteins (chaperones) in fish and shellfish and their potential role in relation to fish health: a review. J Fish Dis 33:789–801
Roobol A, Carden MJ, Newsam RJ, Smales C (2009) Biochemical insights into the mechanisms central to the response of mammalian cells to cold stress and subsequent rewarming. FEBS J 276:286–302
Ryckaert J, Pasmans F, Tobback E, Duchateau L, Decostere A, Haesebrouck F, Sorgeloos P, Bossier P (2010) Heat shock proteins protect platyfish (Xiphophorus maculatus) from Yersinia ruckeri induced mortality. Fish Shellfish Immunol 28:228–231
Sarasquete C, Segner H (2000) Cytochrome P4501A (CYP1A) in teleostean fishes. A review of immunohistochemical studies. Sci Total Environ 247:313–332
Schild H, Rammensee HG (2000) gp96—the immune system’s Swiss army knife. Nat Immunol 1:100–101
Schneider-Merck T, Pohnke Y, Kempf R, Christian M, Brosens JJ, Gellersen B (2006) Physical interaction and mutual transrepression between CCAAT/enhancer-binding protein beta and the p53 tumor suppressor. J Biol Chem 281:269–278
Sonna LA, Fujita J, Gaffin SL, Lilly CM (2002) Invited review: effects of heat and cold stress on mammalian gene expression. J Appl Physiol 92:1725–1742
Teles M, Mackenzie S, Boltana S, Callol A, Tort L (2011) Gene expression and TNF-alpha secretion profile in rainbow trout macrophages following exposures to copper and bacterial lipopolysaccharide. Fish Shellfish Immunol 30:340–346
Vergnes L, Chin R, Young SG, Reue K (2011) Heart-type fatty acid-binding protein is essential for efficient brown adipose tissue fatty acid oxidation and cold tolerance. J Biol Chem 286:380–390
Verleih M, Rebl A, Koellner B, Korytar T, Köbis JM, Kühn C, Wimmers K, Goldammer T (2013) Iron–sulfur cluster scaffold (ISCU) gene is duplicated in salmonid fish and tissue and temperature dependent expressed in rainbow trout. Gene 512(2):251–258
Wagner F, Heidtke KR, Drescher B, Radelof U (2007) Development and perspectives of scientific services offered by genomic biological resource centres. Brief Funct Genomic Proteomic 6:163–170
Watts M, Munday BL, Burke CM (2001) Immune responses of teleost fish. Aust Vet J 79:570–574
Yamazaki, T (1991) Culture of foreign origin fishes. Farming Japan (25th anniversary) 25-1:41–46
Yiangou M, Paraskeva E, Hsieh CC, Markou E, Victoratos P, Scouras Z, Papaconstantinou J (1998) Induction of a subgroup of acute phase protein genes in mouse liver by hyperthermia. Biochim Biophys Acta 1396:191–206
Acknowledgments
This work is coordinated by the Campus bioFISCH-MV and is funded by the European Fisheries Fund (EFF) and the Ministry of Agriculture, the Environment and Consumer Protection Mecklenburg-Western Pomerania (pilot project: Rainbow trout BORN). We wish to acknowledge I. Hennings and B. Schöpel for expert technical assistance.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resource 1
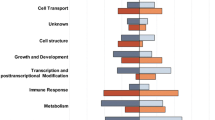
The percentage of overrepresented genes assigned to specific biological functions in either BORN or import trout at both temperatures is presented. Note that neither genes linked with protein synthesis nor genes related to energy production are stronger expressed in BORN trout. The percentage of overrepresented genes involved in selected biological functions is shown for gill tissue of BORN (black columns) and import trout (grey) at 8 °C (filled) and 23 °C (hatched) according to IPA analysis. Most significant biological functions are denoted with asterisks, whereas *** indicate lowest p-values (DOCX 44 kb)
Online Resource 2
(XLSX 120 kb)
Online Resource 3
(TIFF 10430 kb)
Online Resource 4
(DOCX 35 kb)
Online Resource 5
(DOCX 25 kb)
Rights and permissions
About this article
Cite this article
Rebl, A., Verleih, M., Köbis, J.M. et al. Transcriptome Profiling of Gill Tissue in Regionally Bred and Globally Farmed Rainbow Trout Strains Reveals Different Strategies for Coping with Thermal Stress. Mar Biotechnol 15, 445–460 (2013). https://doi.org/10.1007/s10126-013-9501-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-013-9501-8