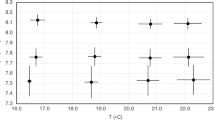
Abstract
Noticeable changes in global temperatures, climate and ocean carbon chemistry are the result of carbon dioxide increase in the atmosphere. This increase has been mitigated by the oceans capacity to absorb one-fourth of the carbon dioxide in the atmosphere, although this CO2 intake affects oceans carbonate chemistry [i.e., ocean acidification—(OA)]. The detrimental effect of OA in the development and shell formation has been studied in several species of bivalves, although no information is available on the wedge shell Donax trunculus, a gastronomically appreciated species and an important economical resource in several southern European countries. We evaluated the effect of pCO2 increase on hatching and early life stages of D. trunculus, considering two ocean acidification scenarios (ΔpH −0.3 and ΔpH −0.6). Our results showed that elevated pCO2 caused a delay in hatching into D-larvae and reduced larvae survival. In the extreme scenario (ΔpH −0.6), some trochophore larvae persisted to day 9 of the experiment and more abnormal larvae were produced than in the ΔpH −0.3 and control treatments. At day 5, normal veligers under extreme acidification were smaller than in other treatments, but by day 9, these differences were attenuated and the average size of normal D-larvae varied inversely to the pH gradient. Possible underlying mechanisms for these complex response patterns are discussed, including the existence of phenotypic plasticity or genetic pre-adaptive capacity in this D. trunculus population to cope with future environmental changes.





Similar content being viewed by others
References
Andersson AJ, Mackenzie FT (2011) Ocean acidification: setting the record straight. Biogeosci Discuss 8:6161–6190. doi:10.5194/bgd-8-6161-2011
Aranda JA, Louzán A, Da Costa F, Cerviño-Otero A, Nóvoa S, Ojea J, Martinez-Patiño D (2009) Desarrollo embrionario y larvario de dos especies de moluscos bivalvos: Donax trunculus (Linné, 1758) y Ruditapes decussatus (Linné, 1758). XII Congreso Nacional de Acuicultura: Con la acuicultura alimentamos tu salud, Daniel Beaz, Morris Villaroel, Salvador Cárdenas. Ministerio de Medio Ambiente, Medio Rural y Marino; Sociedad Española de Acuicultura; Fundación Observatorio Español de Acuicultura, Madrid, pp 516–517
Barros P, Sobral P, Range P, Chícharo L, Matias D (2013) Effects of sea-water acidification on fertilization and larval development of the oyster Crassostrea gigas. J Exp Mar Biol Ecol 440:200–206. doi:10.1016/j.jembe.2012.12.014
Berge JA, Bjerkeng B, Pettersen O, Schaanning MT, Øxnevad S (2006) Effects of increased sea water concentrations of CO2 on growth of the bivalve Mytilus edulis L. Chemosphere 62:681–687. doi:10.1016/j.chemosphere.2005.04.111
Bonsdorff E, Norkko A, Boström C (1995) Recruitment and population maintenance of the bivalve Macoma balthica (L.)—factors affecting settling success and early survival on shallow sandy bottoms. In: Eleftheriou A, Ansell A, Smith C (eds) Biology and ecology of shallow coastal waters. Proceedings 28th European Marine Biology Symposium. Olsen and Olsen, Fredensborg, pp 253–260
Bopp L, Resplandy L, Orr JC, Doney SC, Dunne JP, Gehlen M, Halloran P, Heinze C, Ilyina T, Séférian R, Tjiputra J, Vichi M (2013) Multiple stressors of ocean ecosystems in the 21st century: projections with CMIP5 models. Biogeosciences 10:6225–6245. doi:10.5194/bg-10-6225-2013
Broecker W, Takahashi T (1966) Calcium carbonate precipitation on the Bahama banks. J Geophys Res 71:1575. doi:10.1029/JZ071i006p01575
Byrne M, Przeslawski R (2013) Multistressor impacts of warming and acidification of the ocean on marine invertebrates’ life histories. Integr Comp Biol 53:582–596. doi:10.1093/icb/ict049
Calabrese A, Davis HC (1966) The pH tolerance of embryos and larvae of Mercenaria mercenaria and Crassostrea virginica. Biol Bull 131:427–436. doi:10.2307/1539982
Caldeira K, Wickett M (2003) Anthropogenic carbon and ocean pH. Nature 425:365. doi:10.1038/425365a
Cooley SR, Lucey N, Kite-Powell H, Doney SC (2012) Nutrition and income from molluscs today imply vulnerability to ocean acidification tomorrow. Fish Fish 13:182–215. doi:10.1111/j.1467-2979.2011.00424.x
da Costa González F (ed) (2013) Clam fisheries and aquaculture. Nova Science Publishers, Hauppauge
Fabry V, Seibel B, Feely R, Orr J (2008) Impacts of ocean acidification on marine fauna and ecosystem processes. ICES J Mar Sci 65:414–432. doi:10.1093/icesjms/fsn048
Feely R, Sabine C, Lee K, Berelson W, Kleypas J, Fabry VJ, Millero FJ (2004) Impact of anthropogenic CO2 on the CaCO3 system in the oceans. Science 305:362–366. doi:10.1126/science.1097329
Feely R, Doney S, Cooley S (2009) Ocean acidification: present conditions and future changes in a high-CO2 world. Oceanography 22:36–47. doi:10.5670/oceanog.2009.95
Frankignoulle M, Abril G, Borges A, Bourge I, Canon C, DeLille B, Libert E, Theate JM (1998) Carbon dioxide emission from European estuaries. Science 282:434–436. doi:10.1126/science.282.5388.434
Gaspar M, Ferreira R, Monteiro C (1999) Growth and reproductive cycle of Donax trunculus L., (Mollusca: Bivalvia) off Faro southern Portugal. Fish Res 41:309–316. doi:10.1016/S0165-7836(99)00017-X
Gazeau F, Quiblier C, Jansen JM, Gattuso JP, Middelburg JJ, Heip CHR (2007) Impact of elevated CO2 on shellfish calcification. Geophys Res Lett 34:L07603. doi:10.1029/2006GL028554
Gazeau F, Gatuso JP, Dawber C, Pronker AE, Peene F, Peene J, Heip CHR, Middelburg JJ (2010) Effect of ocean acidification on the early life stages of the blue mussel Mytilus edulis. Biogeosciences 7:2051–2060. doi:10.5194/bg-7-2051-2010
Gazeau F, Parker LM, Comeau S, Gattuso JP, O’Connor WA, Martin S, Pörtner HO, Pauline MR (2013) Impacts of ocean acidification on marine shelled mollusks. Mar Biol 160:2207–2245. doi:10.1007/s00227-013-2219-3
Hart M, Strathann R (1995) Mechanisms and rates of suspension feeding. In: McEdwards L (ed) Ecology of marine invertebrate larvae. CRC Press, Boca Raton, pp 193–222
Hendriks IE, Duarte CM, Álvarez M (2010) Vulnerability of marine biodiversity to ocean acidification: a meta-analysis. Estuar Coast Shelf Sci 86:157–164. doi:10.1016/j.ecss.2009.11.022
Hiebenthal C, Philipp EER, Eisenhauer A, Wahl M (2012) Effects of seawater pCO2 and temperature on shell growth, shell stability, condition and cellular stress of Western Baltic Sea Mytilus edulis (L.) and Arctica islandica (L.). Mar Biol 160:2073–2087. doi:10.1007/s00227-012-2080-9
His E, Seaman MNL, Beiras R (1997) A simplification the bivalve embryogenesis and larval development bioassay method for water quality assessment. Water Res 31(2):351–355. doi:10.1016/S0043-1354(96)00244-8
Hoegh-Guldberg O, Cai R, Poloczanska ES, Brewer PG, Sundby S, Hilmi K, Fabry VJ, and Jung S (2014) The Ocean. In: Barros VR, Field CB, Dokken DJ, Mastrandrea MD, Mach KJ, Bilir TE, Chatterjee M, Ebi KL, Estrada YO, Genova RC, Girma B, Kissel ES, Levy AN, MacCracken S, Mastrandrea PR, White LL (eds) Climate change 2014: impacts, adaptation, and vulnerability. Part B: regional aspects. Contribution of working group II to the fifth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, pp 1655–1731
Ivanina AV, Dickinson GH, Matoo OB, Bagwe R, Dickinson A, Beniash E, Sokolova IM (2013) Interactive effects of elevated temperature and CO2 levels on energy metabolism and biomineralization of marine bivalves Crassostrea virginica and Mercenaria mercenaria. Comp Biochem Physiol A: Mol Integr Physiol 166:101–111. doi:10.1016/j.cbpa.2013.05.016
Jansson A, Norkko J, Norkko A (2013) Effects of reduced pH on Macoma balthica larvae from a system with naturally fluctuating pH-dynamics. PLoS One 8(6):e68198. doi:10.1371/journal.pone.0068198
Kurihara H, Kato S, Ishimatsu A (2007) Effects of increased seawater pCO2 on early development of the oyster Crassostrea gigas. Aquat Biol 1:91–98. doi:10.3354/ab00009
Kurihara H, Asai T, Kato S, Ishimatsu A (2009) Effects of elevated pCO2 on early development in the mussel Mytilus galloprovincialis. Aquat Biol 4:225–233. doi:10.3354/ab00109
Matoo OB, Ivanina AV, Ullstad C, Beniash E, Sokolova IM (2013) Interactive effects of elevated temperature and CO(2) levels on metabolism and oxidative stress in two common marine bivalves (Crassostrea virginica and Mercenaria mercenaria). Comp Biochem Physiol A: Mol Integr Physiol 164:545–553. doi:10.1016/j.cbpa.2012.12.025
McConnaughey TA, Gillikin DP (2008) Carbon isotopes in mollusk shell carbonates. Geo-Mar Lett 28:287–299. doi:10.1007/s00367-008-0116-4
Orr J, Fabry V, Aumont O, Boop L, Doney SC, Feely RA, Gnanadesikan A, Gruber N, Ishida A, Joos F, Key RM, Lindsey K, Maier-Reiner E, Matean R, Montray P, Mouchet A, Najjan RG, Plattner GK, Rodgers KB, Slater RD, Totterdell IJ, Weirig MF, Yamanaka Y (2005) Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature 437:681. doi:10.1038/nature04095
Parker LM, Ross PM, O’Connor WA (2011) Populations of the Sydney rock oyster, Saccostrea glomerata, vary in response to ocean acidification. Mar Biol 158:689–697. doi:10.1007/s00227-010-1592-4
Pedersen TM, Hansen JLS, Josefson AB, Hansen BW (2008) Mortality through ontogeny of soft-bottom marine invertebrates with planktonic larvae. J Mar Syst 73:185–207. doi:10.1016/j.jmarsys.2007.10.008
Pereira AM (2013) The importance of the pelagic larval phase of the wedge shell Donax trunculus (L.): implications for the management of the fishery. Ph.D. dissertation, Universidade do Algarve
Ramón M, Abelló P, Richardson C (1995) Population structure and growth of Donax trunculus (Bivalvia Donacidae) in the western Mediterranean. Mar Biol 121:665–671. doi:10.1007/BF00349302
Range P, Chícharo MA, Ben-Hamadou R, Piló D, Matias D, Joaquim S, Oliveira AP, Chícharo L (2011) Calcification, growth and mortality of juvenile clams Ruditapes decussatus under increased pCO2 and reduced pH: variable responses to ocean acidification at local scales? J Exp Mar Biol Ecol 396:177–184. doi:10.1016/j.jembe.2010.10.020
Range P, Piló D, Ben-Hamadou R, Chícharo MA, Matias D, Joaquim S, Oliveira AP, Chícharo L (2012) Seawater acidification by CO2 in a coastal lagoon environment: effects on life history traits of juvenile mussels Mytilus galloprovincialis. J Exp Mar Biol Ecol 424–425:89–98. doi:10.1016/j.jembe.2012.05.010
Raven J, Caldeira K, Elderfield H, Hoegh-Guldberg O (2005) Ocean acidification due to increasing atmospheric carbon dioxide. Clyvedon Press, Cardiff
Rodolfo-Metalpa R, Houlbrèque F, Tambutté É, Boisson F, Baggini C, Patti FP, Jeffree M, Fine M, Foggo A, Gattuso JP, Hall-Spencer JM (2011) Coral and mollusc resistance to ocean acidification adversely affected by warming. Nat Clim Change 1:308–312. doi:10.1038/nclimate1200
Ruiz-Azcona P, Rodriguez-Sierra R, Martin JB (1996) Culture of coquina clam, Donax trunculus, larvae. Aquaculture 139:151–155. doi:10.1016/0044-8486(95)01154-4
Sabine CL, Feely RA, Gruber N, Key RM, Lee K, Bullister JL, Wanninkhof R, Wong CS, Wallace DWR, Tilbrook B, Millero FS, Peng TH, Kozyr A, Ono T, Rio AF (2004) The oceanic sink for anthropogenic CO2. Science 305:367–371. doi:10.1126/science.1097403
Talmage SC, Gobler CJ (2011) Effects of elevated temperature and carbon dioxide on the growth and survival of larvae and juveniles of three species of northwest Atlantic bivalves. PLoS One 6:e26941. doi:10.1371/journal.pone.0026941
Thomsen J, Casties I, Pansch C, Körtzinger A, Melzner F (2013) Food availability outweighs ocean acidification effects in juvenile Mytilus edulis: laboratory and field experiments. Glob Change Biol 19:1017–1027. doi:10.1111/gcb.12109
Tlili S, Métais I, Boussetta H, Mouneyrac C (2010) Linking changes at sub-individual and population levels in Donax trunculus: assessment of marine stress. Chemosphere 81:692–700. doi:10.1016/j.chemosphere.2010.07.064
Van Colen C, Debusschere E, Braeckman U, Van Gansbeke D, Vincx M (2012) The early life history of the clam Macoma balthica in a high CO2 world. PLoS One 7:e44655. doi:10.1371/journal.pone.0044655
Weiss IM, Tuross N, Addadi L, Weiner S (2002) Mollusc larval shell formation: amorphous calcium carbonate is a precursor phase for aragonite. J Exp Zool 293:478–491. doi:10.1002/jez.90004
White MM, McCorkle DC, Mullineaux LS, Cohen AL (2013) Early exposure of bay scallops (Argopecten irradians) to high CO2 causes a decrease in larval shell growth. PLoS One 8:e61065. doi:10.1371/journal.pone.0061065
Wicks L, Roberts J (2012) Benthic invertebrates in a high—CO2 world. In: Gibson RN, Atkinson RJA, Gordon JDM, Hughes RN (eds) Oceanography and marine biology. CRC Press, Boca Raton, pp 127–188
Wittmann AC, Pörtner HO (2013) Sensitivities of extant animal taxa to ocean acidification. Nat Clim Change 3:995–1001. doi:10.1038/nclimate1982
Acknowledgments
We are grateful to Sr. Mauricio Teixeira for system maintenance assistance. This work was supported by the grants SFRH/BD/40430/2007 and SFRH/BPD/69959/2010, awarded by Fundação para a Ciência e Tecnologia to A.M.P and P.R, respectively.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: Wolfgang Cramer.
Rights and permissions
About this article
Cite this article
Pereira, A.M., Range, P., Campoy, A. et al. Larval hatching and development of the wedge shell (Donax trunculus L.) under increased CO2 in southern Portugal. Reg Environ Change 16, 855–864 (2016). https://doi.org/10.1007/s10113-015-0803-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10113-015-0803-4