Abstract
Previous studies in our laboratory described a new group A streptococcal protective antigen (Spa) in type 18 streptococci that was distinct from the type 18 M protein. This study was undertaken to identify additional serotypes of group A streptococci that express Spa proteins. PCR techniques were used to identify and clone a new spa gene from type 36 streptococci. The 5′ sequence of spa36 was highly variable compared to spa18, while the 3′ sequence was conserved. Antisera against Spa36 opsonized type 36 streptococci but not type 18 streptococci, indicating that the opsonic Spa epitopes were type-specific. Antisera against the conserved carboxy-terminal half of Spa18 were used to identify Spa or Spa-like proteins expressed on the surface of 25 of 70 different serotypes of GAS. Spa proteins may represent a new family of type-specific surface antigens that function in concert with M proteins to elicit protective immune responses.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Group A streptococci (GAS) are strict human pathogens responsible for a wide variety of infections ranging from mild to moderate pharyngitis, impetigo, erysipelas and cellulitis to severe life threatening invasive infections including necrotizing fasciitis and toxic shock syndrome [1]. GAS also cause noninfectious immunological complications such as acute rheumatic fever, rheumatic heart disease and glomerulonephritis. These organisms express a number of surface molecules that function as virulence determinants, some of which confer resistance to opsonization and phagocytic killing. Classic studies by Lancefield [2] demonstrated the central role of the type-specific M protein in mouse virulence, resistance to complement-mediated opsonization and phagocytic killing and the protective efficacy of M protein antibodies. More recent studies have elucidated the complexities of the bacterial surface and the interrelated functions of M proteins, M-related proteins (Mrp) and capsule, among others, in preventing complement-mediated phagocytosis [3–6]. In some strains, the M protein is the dominant virulence factor and primary protective antigen [7]. In others, the M protein, Mrp and capsule play variable roles in protecting the organism from innate complement-mediated defenses and providing targets for antibody-mediated immune responses that protect against infection [6, 8].
Previous studies from our laboratory described a new streptococcal protective antigen (Spa) on the surface of serotype 18 GAS [9, 10]. Spa18 and M18 were both expressed on the bacterial surface and both were shown to contain epitopes that evoked opsonic antibodies. In addition, Spa functioned in concert with M18 to block complement-mediated phagocytic killing in human blood. Moreover, expression of Spa18 and M18 were required for optimal virulence in mice [10]. Our study was undertaken to identify spa genes in other serotypes of GAS. We report the structure and type-specificity of a new Spa protein expressed on the surface of type 36 GAS. In addition, we show that several other serotypes of GAS contain spa-like genes. Our data suggest that Spa proteins may represent a new family of type-specific surface antigens which could have important implications in pathogenesis of GAS infections and in vaccine development because of their ability to evoke opsonic antibodies.
Materials and methods
Bacterial strains
Strains of GAS used in this study were from our laboratory collection and originally obtained from a variety of sources, including Dr. Bernard Beall at the Centers for Disease Control and Prevention, Atlanta, Georgia and Dr. Stan Shulman, Children’s Memorial Medical Center in Chicago, Illinois. E.coli M15 was used for molecular cloning using the plasmid pQE-30 (Qiagen, Valencia, CA), as previously described [10].
DNA techniques
Plasmid DNA was purified with miniprep kits (Qiagen) and chromosomal DNA was prepared from different GAS serotypes as described by Caparon and Scott [11].
ELISA
Microtiter wells (Nunc-immuno modules; Nalge Nunc International, Roskilde, Denmark) were coated with whole bacteria grown overnight in THB and yeast extract, washed with PBS and resuspended in PBS and adjusted to an OD530 of 1.0. In some experiments, ELISA was performed using microtiter wells coated with recombinant proteins representing the N-terminal half of either Spa18 and Spa36 by methods previously described [12]. A combination of 5% bovine serum, 5% horse serum and 5% porcine serum, diluted in PBS (PH 7.4) with 0.05% Tween 20, was used to block unoccupied binding sites of the wells as well as the Fc binding sites on the surface of the bacteria. Rabbit antisera against the C-terminal half of Spa18, the N-terminal half of Spa18 and the N-terminal half of Spa36 were used as primary antibodies. To determine titers, the sera were serially diluted in PBS (PH 7.4) with 0.05% Tween 20, added to the wells, and incubated at 37°C for 2 h. In screening experiments sera were used at a single dilution of 1:500. The wells were washed with 0.15% saline-Tween 20. A 1:2000 dilution of peroxidase-conjugated goat immunoglobulin G (IgG) to rabbit immunoglobulins (IgG, IgM, and IgA) was added and incubated at 37°C for 1 h. After washing, peroxidase substrate was added and A405 was recorded after 15 minutes by a microplate reader. Wells containing pre-immune rabbit serum or PBS instead of immune rabbit serum but containing all other reagents served as negative controls.
Identification of spa or spa-like genes in multiple serotypes of GAS
GAS chromosomal DNA was used as a template to screen for the presence of spa or spa-like genes by PCR. The spa18 gene resides in a locus between relA and nrdI genes [13]. Based on a comparison of several GAS genome sequences upstream of nrdI, and within the gene pstG, a 24 bp sequence (CAATCTTATCAAAACAACCTATTC) was selected as a forward primer and another (AGATTTGTACTCAACTTCACCTAA) located within the relA gene was synthesized as a reverse primer. PCR primers copying sequences within nrdI (ACTGAGGCTAATAAAAACAAGGG) and relA (CACCATCAACGATATCAC) were also used in subsequent experiments. The 3′ half of spa or spa-like genes was amplified using internal forward (GTTGGTAGTCTAGACCGC) and reverse (GTTTTCTTTGCGTTTAGG) primers based on the published spa18 sequence [13]. Amplicons were sequenced directly by the University of Tennessee Molecular Resource Center, using primer walking when required.
Cloning, expression, and purification of recombinant Spa36
The first 603 bp of spa36 encoding the N-terminal half of the mature protein were amplified by PCR and ligated into the expression vector PQE-30 containing a 6-His tag (Qiagen). The resulting construct was used to transform E.coli M15, and clones were selected for ampicillin resistance and screened for spa inserts by plasmid DNA extraction, digestion and DNA electrophoresis. Four clones containing inserts of the appropriate size were selected and the Spa protein (N-Spa36) from one of these clones was expressed using QIA expressionist protocol (Qiagen) and purified over a Ni-column. This was then dialyzed against PBS and purity was assessed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). The recombinant protein migrated as a single band with an approximate molecular mass of 25 kDa. Cloning, expression and purification of the 3′ 741 bp of spa18 encoding the C-terminal half of the mature protein (C-Spa18) was prepared using the same techniques. Cloning, expression and purification of the N-terminus of Spa18 (N-Spa18) was described previously [10].
Preparation of antisera
The purified N-Spa36 fragment (200 µg) was mixed with 500 µg alum (Rehydragel, Reheis, Inc., Berkeley Heights, New Jersey) and used to immunize rabbits via the intramuscular route. Booster injections were given at 4, 8 and 13 weeks, and sera were collected at 0, 5, 9, 10, 14 and 15 weeks. Antiserum against a synthetic peptide copying the N-terminal 30 amino acids of type 36 M protein was raised in a similar fashion except that the peptide was first conjugated to KLH by methods previously described [14]. Preparation of antisera against N-Spa18 and a synthetic peptide of M18 have been previously described [10].
Opsonization assays
In vitro opsonization assays were performed as previously described [15]. Briefly, assays were performed with whole, heparinized, nonimmune human blood. A standard inoculum of streptococci, grown to log-phase, was added to 0.1 ml of test serum and 0.35 ml of blood. The tubes were rotated end-over-end for 45 minutes at 37°C. In some experiments, phagocytosis was assessed using blood that was reconstituted with one part fresh serum or plasma containing heparin and one part cells that had been washed two times in Hanks’ balanced salt solution with no antiserum added to the mixture. Some reaction mixtures contained hyaluronidase (0.2 mg/ml, bovine testicular enzyme obtained from Sigma, St. Louis, Missouri) to remove the GAS capsule. At the end of the incubation period, smears were made on glass slides and stained with modified Wright’s stain. Opsonization was quantitated by counting 50 consecutive neutrophils per slide and calculating the percentage with associated streptococci [15].
Blood passage of GAS
In an effort to identify phenotypic variants of GAS, blood passage was performed with selected strains. The reaction mixtures were similar to those described above for the opsonization assay except that fewer bacteria were added and no immune serum was used. The mixture was rotated for 3 h at 37°C, and 0.02 ml aliquots of the mixture were streaked onto blood agar plates, incubated overnight at 37°C and individual colonies were recovered.
Mouse virulence of type 36 GAS
Mouse virulence of phenotypic variants of type 36 GAS was assessed after intraperitoneal challenge infections of ICR white mice. Two groups of ten mice each were challenged with 107 CFU of either M+/Spa+ or M+/Spa- type 36 GAS (naturally occurring variants of the same strain). Deaths were recorded for 3 days.
Hot acid extraction of surface proteins
Hot acid extracts of type 36 GAS were prepared for semi-quantitative analyses of M and Spa protein expression by Western blot. Type 36 GAS were grown in 10 ml of THB + 1% yeast at 37°C overnight. Bacteria were pelleted, washed once with PBS and then resuspended in PBS to an O.D. of 3.0 at A530. Then, 250 µl of 0.2 M HCl was added to each cell pellet which was resuspended and boiled for 5 minutes. The suspension was neutralized to a pH of 7.4 by adding 0.2 M NaOH. Suspensions were centrifuged and supernatants were used in Western blots described below.
Western blots
Western blots were performed as described previously [16] using surface proteins of GAS extracted by hot acid. The blots were probed with rabbit antisera against recombinant Spa and synthetic M proteins described above at a dilution of 1:200.
GenBank accession number
The sequence of spa36 has been entered into GenBank with the accession number EU559276.
Results
Identification of Spa-like proteins expressed on the surface of multiple serotypes of GAS
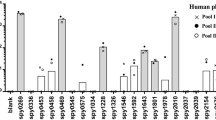
Our previous studies of Spa18 showed that the 3′ sequence of the gene was highly conserved compared to the gene that encodes SeM of Streptococcus equi yet distinct from the 3′ sequence of emm genes of GAS [10, 17]. In addition, the 5′ sequence of the spa18 gene encoding the mature protein shared no sequence similarity with the SeM gene of S. equi or any of the GAS emm genes. We postulated that the spa18 gene was acquired through horizontal transfer and then structurally evolved into a serotypically distinct surface protein through mutation and immunological pressure and selection. Based on the paradigm that M proteins of GAS contain highly conserved C-terminal sequences yet variable and hypervariable N-terminal epitopes, we reasoned that the C-terminus of Spa proteins expressed by other serotypes of GAS may share immunological identity with Spa18. Thus, we cloned, expressed and purified the C-terminal half of Spa18 (C-Spa18) and used it to raise antisera in rabbits. The antisera, which had ELISA titers against the purified protein of >100,000, were used to screen 70 different serotypes of GAS from our laboratory collection using a whole-cell ELISA (Table 1). Twenty-five of the 70 strains tested expressed surface proteins that reacted with the antisera against the putative conserved truncated Spa protein, suggesting that Spa or Spa-like proteins may be broadly represented in GAS. Additionally, the Spa antibodies showed a broad range of reactivity among this collection of GAS (Table 1).
Identification of spa and spa-like genes in GAS
The results above suggested that some of the serotypes tested expressed Spa or Spa-like surface proteins. To identify spa genes, we selected a subset of serotypes from the collection based on antibody reactivity and screened genomic DNA for the presence of 3′ spa gene fragments by PCR using forward and reverse primers from within the putative conserved region of spa. All of the serotypes selected that were reactive with C-Spa18 antibodies contained spa-like gene fragments that were confirmed by sequence analysis (Table 2).
The type 18 streptococcal genome sequence [13] revealed that the spa18 gene is located in a locus that is flanked by the genes relA and nrdI, both of which are conserved in other genomes of GAS. Based on the observation that the GAS emm genes of different serotypes are for the most part located in similar chromosomal locations, we used PCR to screen the genomic DNA from the serotypes listed in Table 2 to identify spa genes located between relA and nrdI. The majority of the PCR products ranged in size from 2 kb to 2.5 kb, whereas the type 18 template yielded a PCR product of ~5 kb, suggesting that no additional open reading frames (ORF) were present in this location in the majority of positive strains. Sequence analysis of PCR amplicons from emm types 15, 65, 67 and 78 confirmed the absence of spa in the region between relA and nrdI. However, the DNA template from type 36 GAS yielded a PCR product of ~5 kb, which was identical in size to the PCR fragment derived from the control M18 template.
Cloning, sequencing and expression of spa36
The sequence of the ~1.7 kb ORF located between relA and nrdi of the type 36 strain indicated the presence of a putative new spa36 gene. Dot matrix analysis of the translated protein sequences of Spa18 and Spa36 showed that both had highly conserved signal peptides and C-terminal regions and divergent sequences in the N-terminal regions of the mature proteins (Fig. 1). The translated Spa36 protein had a predicted molecular weight of 60.6 kDa, a signal peptide predicted to be 32 aa in length, an LPSTGE motif and a hydrophilic tail, consistent with a cell wall location for the protein. An analysis of the secondary structural characteristics revealed a high alpha-helical potential throughout most of the length of the Spa36 protein (data not shown).
Type-specific opsonic epitopes of Spa36
To determine whether Spa36 contained epitopes that evoked antibodies that opsonized type 36 streptococci, we used PCR to clone the 5′ variable region of the spa36 gene and expressed and purified the recombinant truncated protein (N-Spa36) which was used to immunize rabbits. Type 36 streptococci also express an emm36 gene in addition to the spa gene. To investigate the relative contributions of Spa and M antibodies to opsonization and phagocytosis of type 36 streptococci, we also raised rabbit antisera against a synthetic peptide copying the N-terminal 30aa of the M36 protein (sM36). N-Spa18 and sM18 rabbit antisera were available from previous studies [10]. Antiserum against N-Spa36 reacted in high titer with N-Spa36 and showed some cross-reactivity with N-Spa18, as determined by ELISA (Table 3). Likewise, N-Spa18 antisera reacted in high titer with N-Spa18 but showed only minimal cross-reactivity with N-Spa36. The M protein antisera reacted only with the homologous synthetic M antigens by ELISA (Table 3).
In vitro opsonization assays showed that the N-Spa36 protein evoked antibodies that opsonized type 36, but not type 18 streptococci. Similarly, N-Spa18 antibodies opsonized only type 18 organisms. These results indicate that Spa36 represents a new type-specific surface protein of GAS that contains opsonic epitopes. The antiserum against sM18 opsonized type 18 streptococci and not type 36 streptococci. Interestingly, the sM36 antiserum did not promote opsonization of the homologous type 36 organism. Although the rabbit antiserum against the conserved C-terminal region of Spa18 (C-Spa18) reacted with the surface of type 36 streptococci (Table 1), it did not promote opsonization of type 36 or type 18 organisms (data not shown). Taken together, these results indicate that type 36 streptococci express a new serotype-specific Spa36 protein that contains opsonic epitopes, but unlike type 18 streptococci the antiserum against M36 resulted in little to no opsonic activity against the homologous serotype.
Phenotypic variation of spa36 expression
During the course of our studies, we identified two phenotypic variants of type 36 streptococci. The predominant phenotype expressed both M36 and Spa36 (M+/Spa+). A minor variant, accounting for ~10% of the colonies on a blood agar plate after passage in human blood, expressed M36 but not Spa (M+/Spa-), as assessed by whole-cell ELISA with N-Spa36 and sM36 rabbit antisera. Both phenotypic variants were identical on blood agar plates and grew as large mucoid colonies, a finding that was consistent with India ink preparations showing a large hyaluronate capsule. Hot acid extracts of the surface proteins from the variant phenotypes were used in Western blots to confirm the results of whole cell ELISA. Anti-N-Spa36 antibodies showed no reaction with acid extracts in the M+/Spa- strain, whereas multiple immunoreactive bands were present in the extract from the Spa+ phenotype. sM36 antisera reacted with multiple bands in both variants (data not shown).
In functional opsonization assays, an interesting and consistent observation was that the sM36 antiserum was much less effective in promoting phagocytosis of the M+/Spa+ variant compared to the M+/Spa- isolate (Table 4), despite the finding that the sM36 antiserum showed almost equal reactivity with hot acid extracts of both phenotypes, indicating that the amount of M36 expressed by both strains was similar. Both phenotypes appeared to be equally resistant to phagocytosis in human blood in the absence of immune serum and both were equally virulent in mice following intraperitoneal challenge infections (Table 4). Despite multiple attempts to recover revertants of the Spa- phenotype to the Spa+ phenotype, none was detected. PCR amplification and sequencing of the spa gene, the leader sequence and putative upstream promoter regions in the Spa- phenotype revealed no mutations or deletions compared to the Spa+ phenotype (data not shown).
Synergism between Spa36 and M36 antibodies in promoting opsonization of type 36 streptococci
Because the M antibodies promoted only low levels of opsonization of the M+Spa+ organism, we sought to determine if these antibodies could synergize with Spa antibodies to promote phagocytosis of M+Spa+ type 36 GAS (Fig. 2). Anti-sM36 antiserum diluted 1:4 and used alone in this assay resulted in 2% opsonization. Dilution of the anti-N-Spa36 rabbit serum to 1:16 reduced the opsonic activity to 2%. However, the combination of the two diluted antisera resulted in 82% opsonization (Fig. 2). These results are strengthened by the observation that further dilution of the sM36 antiserum to 1:8 and 1:16 mixed with a 1:16 dilution of N-Spa36 antiserum continued to result in high levels of opsonic activity well above the expected additive value of either concentration of antiserum alone. These results suggest that antisera against the two surface antigens of type 36 streptococci synergistically promote opsonization and phagocytosis of the organism.
Relative role of M protein, Spa and capsule in resistance to phagocytosis of type 36 streptococci
These experiments were undertaken to determine the relative importance of the capsule and the M and Spa proteins in conferring resistance to phagocytosis. The M+/Spa+ organism and the M+/Spa- variant, both of which expressed large hyaluronate capsules by the India ink test, were equally resistant to phagocytosis by human blood that was reconstituted with either non-immune serum or non-immune plasma (Table 5). Enzymatic digestion of the capsule resulted in significant increases in phagocytosis of both strains in either serum or plasma (Table 5). These results suggest that the hyaluronate capsule of type 36 streptococci plays a significant role in resistance to phagocytosis. In addition, M36 and Spa36 appear to contribute to resistance to opsonization in human blood. However, unlike some other serotypes of GAS this mechanism appears to be independent of the presence of fibrinogen since the levels of opsonization were similar in blood containing either plasma or serum (Table 5).
Discussion
The virulence of GAS is principally determined by the ability of these organisms to colonize the nasopharyngeal mucosa and/or the skin, invade deeper tissues and then resist opsonization and uptake and killing by phagocytic cells. Many previous studies of virulence determinants have focused on the central role of the surface M protein because of its ability to confer resistance to phagocytosis and to function as a protective antigen that evokes opsonic antibodies. It is now generally accepted that, depending on the serotype and strain of GAS under study, the M protein may share functional characteristics with other surface structures to accomplish the necessary traits of virulence and to express protective epitopes [3, 4, 6, 18]. Our previous studies in which we defined the Spa protein of type 18 streptococci showed that Spa18 and M18 both contained opsonic epitopes and both contributed to resistance to phagocytosis of the organism [10]. In this study, we have extended these observations by describing new Spa proteins, with a focus on Spa36.
The heterogeneity of the Spa36 and Spa18 N-terminal sequences and the almost complete identity of the C-terminal regions indicate that they most likely represent surface proteins with common origins that later diverged under immune pressure. In addition, the C-terminal sequences are highly homologous with the C-terminal region of the S. equi SeM protein [10], suggesting that there may have been horizontal gene transfer at some point in time. The conserved epitopes of the Spa protein were identified on the surface of 25/70 GAS serotypes tested using rabbit antisera against the C-Spa18 recombinant protein. The conserved spa gene sequence was identified in eight serotypes in addition to types 18 and 36. Our finding that the C-Spa18 antisera showed low-level reactions with the surface of type 3 GAS, yet the genome of this serotype does not contain a Spa gene, suggests that in some cases the antibodies may recognize cross-reactive epitopes of other surface proteins. However, the conserved spa gene sequence was identified in eight serotypes of GAS in addition to types 18 and 36. Thus, we believe that Spa proteins potentially represent an entirely new family of type-specific protective surface antigens of some GAS. Studies are now underway to clone and sequence additional members of the Spa family.
In these studies we observed that the type 36 organism had two different phenotypes, namely, M+Spa+ and M+Spa-. Unlike type 18 streptococci, which were opsonized equally by antibodies against Spa and M protein [9], type 36 organisms were effectively opsonized by M antibodies only when Spa was not expressed, the explanation for which is not clear. Precise quantitation of antibody binding to the surface of the M+Spa+ phenotype was complicated by the fact that this strain exhibited very high levels of non-specific immunoglobulin binding that was difficult to block with serum from multiple species. However, the semi-quantitative assessment of M protein and Spa protein in hot acid extracts of both phenotypes indicated that similar levels of M protein were expressed by both strains. Indeed, our results showed that the combination of M and Spa antibodies was actually synergistic in promoting opsonization of the M+Spa+ phenotype. These results suggest that the M antibodies alone may have been insufficient to overcome the “antiopsonic” effect of Spa and successful opsonization required the “neutralization” of this effect by Spa antibodies. The findings that Spa antibodies alone were sufficient to promote opsonization of M+Spa+ and that M antibodies were effective in promoting opsonization of M+Spa- organisms could indicate that Spa and M protein use different mechanisms to confer resistance to opsonization. Although in our previous studies we defined the role of M and Spa in the virulence of M18 streptococci, the precise “antiopsonic” mechanism of Spa remains to be determined. Future studies to unravel the reason for this discordant result between M and Spa antibodies may shed new light on the precise mechanisms of antibody-mediated opsonization of GAS.
The finding that some serotypes of GAS express both M and Spa may have important implications for vaccine development. The 26-valent vaccine that is currently undergoing clinical trials [19] contains peptides from both M18 and Spa18 in an effort to evoke maximum levels of opsonic antibodies against this highly “rheumatogenic” serotype [20]. Identification of additional GAS serotypes that express both surface proteins will potentially provide the information needed to broaden the efficacy of future vaccine constructs. This may be of particular importance if Spa expression varies during the course of infection and, as shown in this study, if M antibodies alone are not sufficient to opsonize M+Spa+ strains. Further studies are needed to define the role of Spa in pathogenesis and determine the significance of the phenotypic variation in the expression of Spa.
References
Stevens DL (1999) The flesh-eating bacterium: what’s next? J Infect Dis 179:366–374
Lancefield RC (1962) Current knowledge of the type specific M antigens of group A streptococci. J Immunol 89:307–313
Podbielski A, Schnitzler N, Beyhs P, Boyle MDP (1996) M-related protein (Mrp) contributes to group A streptococcal resistance to phagocytosis by human granulocytes. Molec Micro 19:429
Courtney HS, Hasty DL, Dale JB (2003) Serum opacity factor (SOF) of Streptococcus pyogenes evokes antibodies that opsonize homologous and heterologous SOF-positive serotypes of group A streptococci. Infect Immun 71(9):5097–5103
Dale JB, Washburn RG, Marques MB, Wessels MR (1996) Hyaluronate capsule and surface M protein in resistance to phagocytosis of group A streptococci. Infec Immun 64:1495–1501
Ji Y, Schnitzler N, DeMaster E, Cleary P (1998) Impact of M49, Mrp, Enn, and C5a peptidase proteins on colonization of the mouse oral mucosa by Streptococcus pyogenes. Infect Immun 66(11):5399–5405
Courtney HS, Bronze MS, Dale JB, Hasty DL (1994) Analysis of the role of M24 protein in streptococcal adhesion and colonization by use of omega-interposon mutagenesis. Infect Immun 62:4868
Moses AE, Wessels MR, Zalcman K, Alberti S, Natanson-Yaron S, Menes T, Hanski E (1997) Relative contributions of hyaluronic acid capsule and M protein to virulence in a mucoid strain of the group A streptococcus. Infect Immun 65:64
Dale JB, Chiang EY, Liu SY, Courtney HS, Hasty DL (1999) New protective antigen of group A streptococci. J Clin Invest 103:1261–1268
McLellan DG, Chiang EY, Courtney HS, Hasty DL, Wei SC, Hu MC, Walls MA, Bloom JJ, Dale JB (2001) Spa contributes to the virulence of type 18 group A streptococci. Infect Immun 69(5):2943–2949
Caparon MG, Scott JR (1991) Genetic manipulation of pathogenic streptococci. Methods Enzymol 204:556–586
Hall MA, Stroop SD, Hu MC, Walls MA, Reddish MA, Burt DS, Lowell GH, Dale JB (2004) Intranasal immunization with multivalent group A streptococcal vaccines protects mice against intranasal challenge infections. Infect Immun 72(5):2507–2512
Smoot JC, Barbian KD, Van Gompel JJ, Smoot LM, Chaussee MS, Sylva GL, Sturdevant DE, Ricklefs SM, Porcella SF, Parkins LD, Beres SB, Campbell DS, Smith TM, Zhang Q, Kapur V, Daly JA, Veasy LG, Musser JM (2002) Genome sequence and comparative microarray analysis of serotype M18 group A Streptococcus strains associated with acute rheumatic fever outbreaks. Proc Natl Acad Sci U S A 99(7):4668–4673
Dale JB, Beachey EH (1986) Localization of protective epitopes of the amino terminus of type 5 streptococcal M protein. J Exp Med 163:1191–1202
Beachey EH, Stollerman GH, Chiang EY, Chiang TM, Seyer JM, Kang AH (1977) Purification and properties of M protein extracted from group A streptococci with pepsin: covalent structure of the amino terminal region of the type 24 M antigen. J Exp Med 145:1469–1483
Dale JB, Beachey EH (1985) Multiple heart-cross-reactive epitopes of streptococcal M proteins. J Exp Med 161:113–122
Timoney JF, Artiushin SC, Boschwitz JS (1997) Comparison of the sequences and functions of Streptococcus equi M-like proteins SeM and SzPSe. Infect Immun 65:3600–3605
Courtney HS, Hasty DL, Dale JB (2006) Anti-phagocytic mechanisms of Streptococcus pyogenes: binding of fibrinogen to M-related protein. Mol Microbiol 59(3):936–947
McNeil SA, Halperin SA, Langley JM, Smith B, Warren A, Sharratt GP, Baxendale DM, Reddish MA, Hu MC, Stroop SD, Linden J, Fries LF, Vink PE, Dale JB (2005) Safety and immunogenicity of 26-valent group a streptococcus vaccine in healthy adult volunteers. Clin Infect Dis 41(8):1114–1122
Veasy LG, Tani LY, Daly JA, Korgenski K, Miner L, Bale J, Kaplan EL, Musser JM, Hill HR (2004) Temporal association of the appearance of mucoid strains of Streptococcus pyogenes with a continuing high incidence of rheumatic fever in Utah. Pediatrics 113(3 Pt 1):e168–172
Acknowledgements
This work was supported by research funds from NIH/NIAID USPHS Grants AI10085 and AI060592 and research funds from the Department of Veterans Affairs. P. Tennant was the recipient of a medical student summer fellowship supported by NIH 5T35DK007405.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ahmed, E.A., Penfound, T.A., Brewer, S.C. et al. Streptococcal protective antigens (Spa): a new family of type-specific proteins of group A streptococci. Eur J Clin Microbiol Infect Dis 29, 51–57 (2010). https://doi.org/10.1007/s10096-009-0819-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-009-0819-0