Abstract
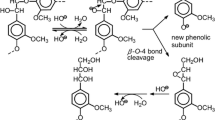
Primary pyrolysis reactions and relative reactivities for depolymerization and condensation/carbonization were evaluated for various lignin model dimers with α-O-4, β-O-4, β-1, and biphenyl substructures by characterizing the tetrahydrofuran (THF)-soluble and THF-insoluble fractions obtained after pyrolysis at 400°C. Reactivity was quite different depending on the model structure: depolymerization: α-O-4 [phenolic (ph), nonphenolic (nonph)], β-O-4 (ph) > β-O-4 (nonph), β-1 (ph, nonph) > biphenyl (ph, nonph); condensation/carbonization: β-1 (ph) > β-O-4 (ph) > α-O-4 (ph) > β-O-4 (nonph), biphenyl (ph, nonph), α-O-4 (nonph), β-1 (nonph). Major degradation pathways were also identified for β-O-4 and β-1 model dimers: β-O-4 types: Cβ-O cleavage to form cinnamyl alcohols and phenols and Cγ-elimination yielding vinyl ethers; β-1 types: Cα-Cβ cleavage yielding benzaldehydes and styrenes and Cγ-elimination yielding stilbenes. Relative reactivities of these pathways were also quite different between phenolic and nonphenolic forms even in the same types; Cβ-O cleavage (β-O-4) and Cγ-elimination (β-1) were substantially enhanced in phenolic forms.
Article PDF
Similar content being viewed by others
References
Kawamoto H, Murayama M, Saka S (2003) Pyrolysis behavior of levoglucosan as an intermediate in cellulose pyrolysis: polymerization into polysaccharide as a key reaction to carbonized product formation. J Wood Sci 49:469–473
Kawamoto H, Hatanaka W, Saka S (2003) Thermochemical conversion of cellulose in polar solvent (sulfolane) into levoglucosan and other low molecular-weight substances. J Anal Appl Pyrolysis 70:303–313
Kawamoto H, Saka S (2006) Heterogeneity in cellulose pyrolysis indicated from the pyrolysis in sulfolane. J Anal Appl Pyrolysis 76:280–284
Kawamoto H, Saito S, Hatanaka W, Saka S (2006) Catalytic pyrolysis of cellulose in sulfolane with some acidic catalysts. J Wood Sci DOI: 10.1007/s10086-006-0835-y
Ramiah MV (1970) Thermogravimetric and differential thermal analysis of cellulose, hemicellulose, and lignin. J Appl Polym Sci 14:1323–1337
Stahl E, Karig F, Brögmann U, Nimz H, Becker H (1973) Thermofractography of lignin and its use for rapid analysis on the ultra-micro-scale. Holzforschung 27:89–92
Karig VF, Stahl E (1974) Über den Einfluß der Thermolysebedingungen auf funktionelle Gruppen bei der Thermofraktographie von Ligninen. Holzforschung 28:201–203
Fenner RA, Lephardt JO (1981) Examination of the thermal decomposition of Kraft lignin by Fourier transform infrared evolved gas analysis. J Agric Food Chem 29:846–849
Jakab E, Faix O, Till F, Székely T (1995) Thermogravimetry/mass spectrometry study of six lignins within the scope of an international round robin test. J Anal Appl Pyrolysis 35:167–179
Jakab E, Faix O, Till F (1997) Thermal decomposition of milled wood lignins studied by thermogravimetry/mass spectrometry. J Anal Appl Pyrolysis 40-41:171–186
Haw JF, Schultz TP (1985) Carbon-13 CP/MAS NMR and FT-IR study of low-temperature lignin pyrolysis. Holzforschung 39:289–296
Domburg GE, Sergeeva VN, Zheibe GA (1970) Thermal analysis of some lignin model compounds. J Therm Anal 2:419–428
Domburg GE, Rossinskaya G, Sergeeva V (1974) Study of thermal stability of b-ether bonds in lignin and its models. Proceedings of the 4th International Conference on Thermogravimetric Analysis, 2:211–220
Domburg GE, Rossinskaya G, Dobele G (1975) Thermoanalytical study of model lignin compounds. V. Thermal decomposition of acetovanillone benzyl ether and pinoresinol. Koksnes Kimija 87-94
Klein MT, Virk PS (1981) Model pathways of lignin thermolysis. Report MIT-EL81-005
Brežný R, Mihálov V, Kváčik V (1983) Low temperature thermolysis of lignins. I. Reactions of β-O-4 model compounds. Holzforschung 37:199–204
Adler E (1977) Lignin chemistry — past, present and future. Wood Sci Technol 11:169–218
Nakatsubo F, Sato K, Higuchi T (1975) Synthesis of guaiacylglycerol-β-guaiacyl ether. Holzforschung 29:165–168
Kristersson P, Lundquist K (1980) A new synthetic route to lignin model compounds of the 1,2-diaryl-1,3-propanediol type. Acta Chem Scand B34:213–234
Li S, Lundquist K, Stomberg R (1993) Synthesis of 1,2-bis(3,4-dimethoxyphenyl)-1,3-propanediol starting from trans-1,3-bis(3,4-dimethoxyphenyl)-2,3-epoxy-1-propanone. Acta Chem Scand 47:867–871
Kratzl K, Vierhapper FW (1971) Synthese von 14C-kernmarkierten Vanillinen und Bikreosolen. Monatsh Chem 102:425–430
Yaguchi T, Hosoya S, Nakano J, Satoh A, Nomura Y, Nakamura M (1979) Mechanism of rapid delignification during alkaline cooking with addition of tetrahydroanthraquinone. Mokuzai Gakkaishi 25:239–240
Gierer J, Lenic J, Norén I, Szabo-Lin I (1974) Lignin chromophores. Part I. Synthesis of chromophores of the 2,4′-and 4,4′-dihydroxystilbene types. Acta Chem Scand B28:717–729
Szabo-Lin I, Teder A (1976) Absorption bands in the electronic spectra of lignin model compounds. Part 2. Stilbenes. Sven Papperstidn 5:153–156
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kawamoto, H., Horigoshi, S. & Saka, S. Pyrolysis reactions of various lignin model dimers. J Wood Sci 53, 168–174 (2007). https://doi.org/10.1007/s10086-006-0834-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10086-006-0834-z