Abstract
The objective was to evaluate time to reach an EDSS of 4, 6, and 7 in NMOSD and MOGAD patients included in the Argentinean MS and NMOSD registry (RelevarEM, NCT 03,375,177).
Methods
NMOSD patients diagnosed according to 2015 criteria and with MOGAD were identified. Patients with at least 3 years of follow-up and periodic clinical evaluations with EDSS outcomes were included. AQP4-antibody and MOG-antibody status was recorded, and patients were stratified as seropositive and seronegative for AQP4-antibody. EDSS of 4, 6, and 7 were defined as dependent variables. Log rank test was used to identify differences between groups.
Results
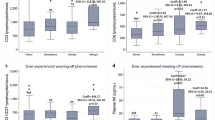
Registry data was provided for a total of 137 patients. Of these, seventy-five presented AQP4-ab-positive NMOSD, 45 AQP4-ab-negative NMOSD, and 11 MOGAD. AQP4-ab status was determined by cell-based assay (CBA) in 72% of NMOSD patients. MOG-ab status was tested by CBA in all cases. Mean time to EDSS of 4 was 53.6 ± 24.5 vs. 63.1 ± 32.2 vs. 44.7 ± 32 months in seropositive, seronegative NMOSD, and MOGAD, respectively (p = 0.76). Mean time to EDSS of 6 was 79.2 ± 44.3 vs. 75.7 ± 48.6 vs. 54.7 ± 50 months in seropositive, seronegative NMOSD, and MOGAD (p = 0.23), while mean time to EDSS of 7 was 86.8 ± 54 vs. 80.4 ± 51 vs. 58.5 ± 47 months in seropositive, seronegative NMOSD, and MOGAD (p = 0.39).
Conclusion
No differences were observed between NMOSD (seropositive and seronegative) and MOGAD in survival curves.

Similar content being viewed by others
References
Wingerchuk DM, Banwell B, Bennett JL et al (2015) International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology 85(2):177–189
Waters PJ, McKeon A, Leite MI, et al. (2012) Serologic diagnosis of NMO: a multicenter comparison of aquaporin-4-IgG assays. Neurology 78(9): 665–71; discussion 9.
Marignier R, Hacohen Y, Cobo-Calvo A et al (2021) Myelin-oligodendrocyte glycoprotein antibody-associated disease. Lancet Neurol 20(9):762–772
Cobo-Calvo A, Ruiz A, Maillart E et al (2018) Clinical spectrum and prognostic value of CNS MOG autoimmunity in adults: the MOGADOR study. Neurology 90(21):e1858–e1869
Carnero Contentti E, Rojas JI, Cristiano E, et al. (2021) Latin American consensus recommendations for management and treatment of neuromyelitis optica spectrum disorders in clinical practice[Mult Scler Relat Disord. 2020 Oct;45:102428]. Mult Scler Relat Disord 52:103026
Jarius S, Paul F, Aktas O et al (2018) MOG encephalomyelitis: international recommendations on diagnosis and antibody testing. Nervenarzt 89(12):1388–1399
Jurynczyk M, Jacob A, Fujihara K, Palace J (2019) Myelin oligodendrocyte glycoprotein (MOG) antibody-associated disease: practical considerations. Pract Neurol 19(3):187–195
Sato DK, Callegaro D, Lana-Peixoto MA et al (2014) Distinction between MOG antibody-positive and AQP4 antibody-positive NMO spectrum disorders. Neurology 82(6):474–481
Kleiter I, Gahlen A, Borisow N et al (2016) Neuromyelitis optica: evaluation of 871 attacks and 1,153 treatment courses. Ann Neurol 79(2):206–216
Rojas JI, Alonso Serena M, Garcea O et al (2020) Multiple sclerosis and neuromyelitis optica spectrum disorders in Argentina: comparing baseline data from the Argentinean MS Registry (RelevarEM). Neurol Sci 41(6):1513–1519
Rojas JI, Carra A, Correale J et al (2019) The Argentinean multiple sclerosis registry (RelevarEM): methodological aspects and directions. Mult Scler Relat Disord 32:133–137
Lennon VA, Wingerchuk DM, Kryzer TJ et al (2004) A serum autoantibody marker of neuromyelitis optica: distinction from multiple sclerosis. Lancet 364(9451):2106–2112
Waters P, Woodhall M, O’Connor KC et al (2015) MOG cell-based assay detects non-MS patients with inflammatory neurologic disease. Neurol Neuroimmunol Neuroinflamm 2(3):e89
Kitley J, Leite MI, Nakashima I et al (2012) Prognostic factors and disease course in aquaporin-4 antibody-positive patients with neuromyelitis optica spectrum disorder from the United Kingdom and Japan. Brain 135(Pt 6):1834–1849
Mealy MA, Kessler RA, Rimler Z et al (2018) Mortality in neuromyelitis optica is strongly associated with African ancestry. Neurol Neuroimmunol Neuroinflamm 5(4):e468
Jarius S, Ruprecht K, Kleiter I, et al. (2016) MOG-IgG in NMO and related disorders: a multicenter study of 50 patients. Part 1: frequency, syndrome specificity, influence of disease activity, long-term course, association with AQP4-IgG, and origin. J Neuroinflammation 13(1): 279
Contentti EC, Lopez PA, Pettinicchi JP et al (2021) Assessing attacks and treatment response rates among adult patients with NMOSD and MOGAD: Data from a nationwide registry in Argentina. Mult Scler J Exp Transl Clin 7(3):20552173211032336
Carnero Contentti E, Daccach Marques V, Soto de Castillo I, et al. (2020) Clinical features and prognosis of late-onset neuromyelitis optica spectrum disorders in a Latin American cohort. J Neurol 267(5): 1260–8
Zhang LJ, Yang LN, Li T et al (2017) Distinctive characteristics of early-onset and late-onset neuromyelitis optica spectrum disorders. Int J Neurosci 127(4):334–338
Mao Z, Lu Z, Hu X (2014) Distinction between MOG antibody-positive and AQP4 antibody-positive NMO spectrum disorders. Neurology 83(12):1122
Cai LJ, Zhang Q, Zhang Y et al (2020) Clinical characteristics of very late-onset neuromyelitis optica spectrum disorder. Mult Scler Relat Disord 46:102515
Carnero Contentti E, Daccach Marques V, Soto de Castillo I, Tkachuk V, Lopez PA, Rojas JI (2020) Age at onset correlate with disability in Latin American aquaporin-4-IgG-positive NMOSD patients. Mult Scler Relat Disord 44: 102258.
Seok JM, Cho HJ, Ahn SW et al (2017) Clinical characteristics of late-onset neuromyelitis optica spectrum disorder: a multicenter retrospective study in Korea. Mult Scler 23(13):1748–1756
Jarius S, Paul F, Weinshenker BG, Levy M, Kim HJ, Wildemann B (2020) Neuromyelitis optica. Nat Rev Dis Primers 6(1):85
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
Ethics committee approval was obtained for each participating center, and a written informed consent (according to each committee, if necessary) was obtained from all participants before data collection.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rojas, J.I., Pappolla, A., Patrucco, L. et al. Disability outcomes in NMOSD and MOGAD patients: data from a nationwide registry in Argentina. Neurol Sci 44, 281–286 (2023). https://doi.org/10.1007/s10072-022-06409-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-022-06409-w