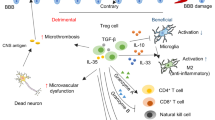
Abstract
CD4+CD25+ regulatory T (Treg) cells and Th17 cells play important roles in peripheral immunity. Immune responses are main elements in the pathogenesis of ischemic stroke (IS). The contribution of Th17 cells in IS patients has not been proved, and whether the balance of Treg/Th17 cells is changed in IS patients remains unidentified. In the present study, we studied Th17 and Treg cell frequency, cytokine secretion, expression of transcription factors, and microRNAs related to Th17 and Treg cells differentiation, which is compared between IS patients and control group. Thirty patients with IS and 30 individuals as control group were enrolled in this study. The frequency of Th17 and Treg lymphocytes, the expression of transcription factors and microRNAs related to these cells, and the serum levels of associated cytokines were assessed by flow cytometry, real-time PCR, and ELISA, respectively. A significant reduction in proportion of peripheral Treg cell frequency and the levels of TGF-β and FOXP3 expression were observed in patients with IS compared with controls, while the proportions of Th17 were increased dramatically, and these effects were along with increases in the levels of IL-17A and RORγt expression in IS patients. The levels of mir-326 and mir-106b-25 expression were increased in patients with IS. These studies suggest that the increase in proportion of Th17 cells and decrease in Treg cells might contribute to the pathogenesis of IS. Manipulating the balance between Tregs and Th17 cells might be helpful for the treatment of IS.





Similar content being viewed by others
Change history
20 February 2018
In the original article the terms RORγt and GF-β were misspelled throughout the text. They should read RORγt and TGF-β instead. We apologize for the inconvenience. The original article has been corrected.
References
Macrez R, Ali C, Toutirais O, Le Mauff B, Defer G, Dirnagl U, Vivien D (2011) Stroke and the immune system: from pathophysiology to new therapeutic strategies. Lancet Neurol 10(5):471–480
Liu X (2012) Beyond the time window of intravenous thrombolysis: standing by or by stenting? Interventional neurology 1(1):3–15. https://doi.org/10.1159/000338389
Park M-G, Kim M-K, Chae S-H, Kim H-K, Han J, Park K-P (2017) Lymphocyte-to-monocyte ratio on day 7 is associated with outcomes in acute ischemic stroke. Neurol Sci 1–7
Xu X, Li M, Jiang Y (2013) The paradox role of regulatory T cells in ischemic stroke. Sci World J 2013:1–8. https://doi.org/10.1155/2013/174373
Shichita T, Sugiyama Y, Ooboshi H, Sugimori H, Nakagawa R, Takada I, Iwaki T, Okada Y, Iida M, Cua DJ (2009) Pivotal role of cerebral interleukin-17-producing γδT cells in the delayed phase of ischemic brain injury. Nat Med 15(8):946–950. https://doi.org/10.1038/nm.1999
Liesz A, Suri-Payer E, Veltkamp C, Doerr H, Sommer C, Rivest S, Giese T, Veltkamp R (2009) Regulatory T cells are key cerebroprotective immunomodulators in acute experimental stroke. Nat Med 15(2):192–199. https://doi.org/10.1038/nm.1927
Kleinschnitz C, Wiendl H (2013) Con: regulatory T cells are protective in ischemic stroke. Stroke 44(8):e87–e88
Dolati S, Babaloo Z, Jadidi-Niaragh F, Ayromlou H, Sadreddini S, Yousefi M (2017) Multiple sclerosis: therapeutic applications of advancing drug delivery systems. Biomed Pharmacother 86:343–353. https://doi.org/10.1016/j.biopha.2016.12.010
Lajoie S, Lewkowich IP, Suzuki Y, Clark JR, Sproles AA, Dienger K, Budelsky AL, Wills-Karp M (2010) Complement-mediated regulation of the IL-17A axis is a central genetic determinant of the severity of experimental allergic asthma. Nat Immunol 11(10):928–935. https://doi.org/10.1038/ni.1926
Swardfager W, Winer DA, Herrmann N, Winer S, Lanctôt KL (2013) Interleukin-17 in post-stroke neurodegeneration. Neurosci Biobehav Rev 37(3):436–447. https://doi.org/10.1016/j.neubiorev.2013.01.021
Schäbitz W-R (2013) Regulatory T cells in ischemic stroke. Stroke 44(8):e84–e84
Kirkham BW, Kavanaugh A, Reich K (2014) Interleukin-17A: a unique pathway in immune-mediated diseases: psoriasis, psoriatic arthritis and rheumatoid arthritis. Immunology 141(2):133–142
Dolati S, Sadreddini S, Rostamzadeh D, Ahmadi M, Jadidi-Niaragh F, Yousefi M (2016) Utilization of nanoparticle technology in rheumatoid arthritis treatment. Biomed Pharmacother 80:30–41. https://doi.org/10.1016/j.biopha.2016.03.004
Magnus T, Wiendl H, Kleinschnitz C (2012) Immune mechanisms of stroke. Curr Opin Neurol 25(3):334–340
Noack M, Miossec P (2014) Th17 and regulatory T cell balance in autoimmune and inflammatory diseases. Autoimmun Rev 13(6):668–677. https://doi.org/10.1016/j.autrev.2013.12.004
Hu Y, Zheng Y, Wu Y, Ni B, Shi S (2014) Imbalance between IL-17A-producing cells and regulatory T cells during ischemic stroke. Mediat Inflamm 2014:1–8. https://doi.org/10.1155/2014/813045
Gan C, Wang C, Tan K (2012) Circulatory microRNA-145 expression is increased in cerebral ischemia. Genet Mol Res 11(1):147–152
Majdi A, Mahmoudi J, Sadigh-Eteghad S, Farhoudi M, Shotorbani SS (2016) The interplay of microRNAs and post-ischemic glutamate excitotoxicity: an emergent research field in stroke medicine. Neurol Sci 37(11):1765–1771
Dolati S, Maleki LA, Ahmadi M, Marofi F, Babaloo Z, Ayramloo H, Jafarisavari Z, Oskouei H, Afkham A, Younesi V (2017) Nanocurcumin restores aberrant miRNA expression profile in multiple sclerosis, randomized, double-blind, placebo-controlled trial. J Cell Physiol
Petrocca F, Vecchione A, Croce CM (2008) Emerging role of miR-106b-25/miR-17-92 clusters in the control of transforming growth factor β signaling. Cancer Res 68(20):8191–8194
De Santis G, Ferracin M, Biondani A, Caniatti L, Tola MR, Castellazzi M, Zagatti B, Battistini L, Borsellino G, Fainardi E (2010) Altered miRNA expression in T regulatory cells in course of multiple sclerosis. J Neuroimmunol 226(1):165–171. https://doi.org/10.1016/j.jneuroim.2010.06.009
de Faria O Jr, Moore CS, Kennedy TE, Antel JP, Bar-Or A, Dhaunchak AS (2012) MicroRNA dysregulation in multiple sclerosis. Front Genet 3
Khoshnam SE, Winlow W, Farzaneh M, Farbood Y, Moghaddam HF (2017) Pathogenic mechanisms following ischemic stroke. Neurol Sci 1–20
Iadecola C, Anrather J (2011) The immunology of stroke: from mechanisms to translation. Nat Med 17(7):796–808. https://doi.org/10.1038/nm.2399
Kleinschnitz C, Schwab N, Kraft P, Hagedorn I, Dreykluft A, Schwarz T, Austinat M, Nieswandt B, Wiendl H, Stoll G (2010) Early detrimental T-cell effects in experimental cerebral ischemia are neither related to adaptive immunity nor thrombus formation. Blood 115(18):3835–3842. https://doi.org/10.1182/blood-2009-10-249078
Planas AM, Chamorro A (2009) Regulatory T cells protect the brain after stroke. Nat Med 15(2):138–139. https://doi.org/10.1038/nm0209-138
Sutton CE, Lalor SJ, Sweeney CM, Brereton CF, Lavelle EC, Mills KH (2009) Interleukin-1 and IL-23 induce innate IL-17 production from γδ T cells, amplifying Th17 responses and autoimmunity. Immunity 31(2):331–341. https://doi.org/10.1016/j.immuni.2009.08.001
Gelderblom M, Weymar A, Bernreuther C, Velden J, Arunachalam P, Steinbach K, Orthey E, Arumugam TV, Leypoldt F, Simova O (2012) Neutralization of the IL-17 axis diminishes neutrophil invasion and protects from ischemic stroke. Blood 120(18):3793–3802. https://doi.org/10.1182/blood-2012-02-412726
Siniscalchi A, Gallelli L, Malferrari G, Pirritano D, Serra R, Santangelo E, De Sarro G (2014) Cerebral stroke injury: the role of cytokines and brain inflammation. J Basic Clin Physiol Pharmacol 25(2):131–137
Yan J, Read SJ, Henderson RD, Hull R, O'Sullivan JD, McCombe PA, Greer JM (2012) Frequency and function of regulatory T cells after ischaemic stroke in humans. J Neuroimmunol 243(1):89–94. https://doi.org/10.1016/j.jneuroim.2011.12.019
Li Q, Wang Y, Yu F, Wang YM, Zhang C, Hu C, Wu Z, Xu X, Hu S (2013) Peripheral Th17/Treg imbalance in patients with atherosclerotic cerebral infarction. Int J Clin Exp Pathol 6(6):1015–1027
Ng HP, Burris RL, Nagarajan S (2011) Attenuated atherosclerotic lesions in apoE-Fcγ-chain-deficient hyperlipidemic mouse model is associated with inhibition of Th17 cells and promotion of regulatory T cells. J Immunol 187(11):6082–6093. https://doi.org/10.4049/jimmunol.1004133
Xiao S, Ma Y, Zhu H, Sun H, Yin Y, Feng G (2015) miRNA functional synergistic network analysis of mice with ischemic stroke. Neurol Sci 36(1):143–148. https://doi.org/10.1007/s10072-014-1904-4
Acknowledgments
This work was financially supported by the grant of Aging Research Institute, Tabriz University of Medical Sciences, and Tabriz, Iran.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interests regarding the publication of this paper.
Additional information
The original version of this article was revised: The terms ROR훾t and TGF-훽 were misspelled throughout the text. They should read RORγt and TGF-β instead.
Rights and permissions
About this article
Cite this article
Dolati, S., Ahmadi, M., Khalili, M. et al. Peripheral Th17/Treg imbalance in elderly patients with ischemic stroke. Neurol Sci 39, 647–654 (2018). https://doi.org/10.1007/s10072-018-3250-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-018-3250-4