Abstract
Introduction
Surgical site infection (SSI) is a frequent complication of abdominal surgery causing increased morbidity. Triclosan-coated sutures are recommended to reduce SSI. The aim of this systematic review and meta-analysis was to evaluate the evidence from randomized controlled trials (RCT) comparing the rate of SSI in abdominal surgery when using triclosan-coated or uncoated sutures for fascial closure.
Methods
A systematic literature search was conducted using Medline, EMBASE, the Cochrane library, CINAHL, Scopus and Web of Science including publications until August 2017. The quality of the RCTs was evaluated using critical appraisal checklists from SIGN. Meta-analyses and trial sequential analysis were performed with Review Manager v5.3 and TSA software, respectively.
Results
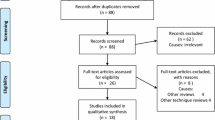
Eight RCTs on abdominal wall closure were included in the meta-analysis. In an overall comparison including both triclosan-coated Vicryl and PDS sutures for fascial closure, triclosan-coated sutures were superior in reducing the rate of SSI (OR 0.67; 0.46–0.98). When evaluating PDS sutures separately, there was no effect of triclosan-coating on the rate of SSI (OR 0.85; 0.61–1.17). Trial sequential analysis showed that the required information size (RIS) of 797 patients for triclosan-coated Vicryl sutures was almost reached with an accrued information size (AIS) of 795 patients. For triclosan-coated PDS sutures an AIS of 2707 patients was obtained, but the RIS was estimated to be 18,693 patients.
Conclusion
Triclosan-coated Vicryl sutures for abdominal fascial closure decrease the risk of SSI significantly and based on the trial sequential analysis further RCTs will not change that outcome. There was no effect on SSI rate with the use of triclosan-coated PDS sutures for abdominal fascial closure, and it is unknown whether additional RCTs will change that.



Similar content being viewed by others
References
Coello R, Charlett A, Wilson J, Ward V, Pearson A, Borriello P (2005) Adverse impact of surgical site infections in English hospitals. J Hosp Infect 60(2):93–103. https://doi.org/10.1016/j.jhin.2004.10.019
Meyhoff CS, Wetterslev J, Jorgensen LN, Henneberg SW, Hogdall C, Lundvall L, Svendsen PE, Molleru H, Lunn TH, Simonsen I, Martinsen KR, Pulawska T, Bundgaard L, Bugge L, Hansen EG, Riber C, Gocht-Jensen P, Walker LR, Bendtsen A, Johansson G, Skovgaard N, Helto K, Poukinski A, Korshin A, Walli A, Bulut M, Carlsson PS, Rodt SA, Lundbech LB, Rask H, Buch N, Perdawid SK, Reza J, Jensen KV, Carlsen CG, Jensen FS, Rasmussen LS, Group PT (2009) Effect of high perioperative oxygen fraction on surgical site infection and pulmonary complications after abdominal surgery: the PROXI randomized clinical trial. JAMA 302(14):1543–1550. https://doi.org/10.1001/jama.2009.1452
Aga E, Keinan-Boker L, Eithan A, Mais T, Rabinovich A, Nassar F (2015) Surgical site infections after abdominal surgery: incidence and risk factors. A prospective cohort study. Infect Dis (Lond) 47(11):761–767. https://doi.org/10.3109/23744235.2015.1055587
Murray BW, Cipher DJ, Pham T, Anthony T (2011) The impact of surgical site infection on the development of incisional hernia and small bowel obstruction in colorectal surgery. Am J Surg 202(5):558–560. https://doi.org/10.1016/j.amjsurg.2011.06.014
Lamarsalle L, Hunt B, Schauf M, Szwarcensztein K, Valentine WJ (2013) Evaluating the clinical and economic burden of healthcare-associated infections during hospitalization for surgery in France. Epidemiol Infect 141(12):2473–2482. https://doi.org/10.1017/S0950268813000253
Graf K, Ott E, Vonberg RP, Kuehn C, Schilling T, Haverich A, Chaberny IF (2011) Surgical site infections—economic consequences for the health care system. Langenbecks Arch Surg 396(4):453–459. https://doi.org/10.1007/s00423-011-0772-0
Sorensen LT (2012) Wound healing and infection in surgery: the pathophysiological impact of smoking, smoking cessation, and nicotine replacement therapy: a systematic review. Ann Surg 255(6):1069–1079. https://doi.org/10.1097/SLA.0b013e31824f632d
Dhurandhar NV, Bailey D, Thomas D (2015) Interaction of obesity and infections. Obes Rev 16(12):1017–1029. https://doi.org/10.1111/obr.12320
Chuah LL, Papamargaritis D, Pillai D, Krishnamoorthy A, le Roux CW (2013) Morbidity and mortality of diabetes with surgery. Nutr Hosp 28(Suppl 2):47–52. https://doi.org/10.3305/nh.2013.28.sup2.6713
Leaper DJ (2010) Risk factors for and epidemiology of surgical site infections. Surg Infect (Larchmt) 11(3):283–287. https://doi.org/10.1089/sur.2010.022
Sessler DI (2016) Perioperative thermoregulation and heat balance. Lancet 387(10038):2655–2664. https://doi.org/10.1016/S0140-6736(15)00981-2
Campbell L, Zirwas MJ (2006) Triclosan. Dermatitis 17(4):204–207
Dhom J, Bloes DA, Peschel A, Hofmann UK (2016) Bacterial adhesion to suture material in a contaminated wound model: comparison of monofilament, braided, and barbed sutures. J Orthop Res. https://doi.org/10.1002/jor.23305
Leaper D, Assadian O, Hubner NO, McBain A, Barbolt T, Rothenburger S, Wilson P (2011) Antimicrobial sutures and prevention of surgical site infection: assessment of the safety of the antiseptic triclosan. Int Wound J 8(6):556–566. https://doi.org/10.1111/j.1742-481X.2011.00841.x
Allegranzi B, Zayed B, Bischoff P, Kubilay NZ, de Jonge S, de Vries F, Gomes SM, Gans S, Wallert ED, Wu X, Abbas M, Boermeester MA, Dellinger EP, Egger M, Gastmeier P, Guirao X, Ren J, Pittet D, Solomkin JS, Group WHOGD (2016) New WHO recommendations on intraoperative and postoperative measures for surgical site infection prevention: an evidence-based global perspective. Lancet Infect Dis. https://doi.org/10.1016/S1473-3099(16)30402-9
Wang ZX, Jiang CP, Cao Y, Ding YT (2013) Systematic review and meta-analysis of triclosan-coated sutures for the prevention of surgical-site infection. Br J Surg 100(4):465–473. https://doi.org/10.1002/bjs.9062
Edmiston CE Jr, Daoud FC, Leaper D (2013) Is there an evidence-based argument for embracing an antimicrobial (triclosan)-coated suture technology to reduce the risk for surgical-site infections?: a meta-analysis. Surgery 154(1):89–100. https://doi.org/10.1016/j.surg.2013.03.008
Apisarnthanarak A, Singh N, Bandong AN, Madriaga G (2015) Triclosan-coated sutures reduce the risk of surgical site infections: a systematic review and meta-analysis. Infect Control Hosp Epidemiol 36(2):169–179. https://doi.org/10.1017/ice.2014.22
Daoud FC, Edmiston CE Jr, Leaper D (2014) Meta-analysis of prevention of surgical site infections following incision closure with triclosan-coated sutures: robustness to new evidence. Surg Infect (Larchmt) 15(3):165–181. https://doi.org/10.1089/sur.2013.177
de Jonge SW, Atema JJ, Solomkin JS, Boermeester MA (2017) Meta-analysis and trial sequential analysis of triclosan-coated sutures for the prevention of surgical-site infection. Br J Surg 104(2):e118–e133. https://doi.org/10.1002/bjs.10445
Leaper DJ, Edmiston CE Jr, Holy CE (2017) Meta-analysis of the potential economic impact following introduction of absorbable antimicrobial sutures. Br J Surg 104(2):e134–e144. https://doi.org/10.1002/bjs.10443
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol 62(10):e1–34. https://doi.org/10.1016/j.jclinepi.2009.06.006
Scottish Intercollegiate Guidelines Network (SIGN). http://www.sign.ac.uk/methodology/index.html
Wetterslev J, Jakobsen JC, Gluud C (2017) Trial Sequential Analysis in systematic reviews with meta-analysis. BMC Med Res Methodol 17(1):39. https://doi.org/10.1186/s12874-017-0315-7
Mattavelli I, Rebora P, Doglietto G, Dionigi P, Dominioni L, Luperto M, La Porta A, Garancini M, Nespoli L, Alfieri S, Menghi R, Dominioni T, Cobianchi L, Rotolo N, Soldini G, Valsecchi MG, Chiarelli M, Nespoli A, Gianotti L (2015) Multi-center randomized controlled trial on the effect of triclosan-coated sutures on surgical site infection after colorectal surgery. Surg Infect (Larchmt) 16(3):226–235. https://doi.org/10.1089/sur.2014.005
Nakamura T, Kashimura N, Noji T, Suzuki O, Ambo Y, Nakamura F, Kishida A (2013) Triclosan-coated sutures reduce the incidence of wound infections and the costs after colorectal surgery: a randomized controlled trial. Surgery 153(4):576–583. https://doi.org/10.1016/j.surg.2012.11.018
Baracs J, Huszar O, Sajjadi SG, Horvath OP (2011) Surgical site infections after abdominal closure in colorectal surgery using triclosan-coated absorbable suture (PDS Plus) vs. uncoated sutures (PDS II): a randomized multicenter study. Surg Infect (Larchmt) 12(6):483–489. https://doi.org/10.1089/sur.2011.001
Rasic Z, Schwarz D, Adam VN, Sever M, Lojo N, Rasic D, Matejic T (2011) Efficacy of antimicrobial triclosan-coated polyglactin 910 (Vicryl* Plus) suture for closure of the abdominal wall after colorectal surgery. Coll Antropol 35(2):439–443
Ruiz-Tovar J, Alonso N, Morales V, Llavero C (2015) Association between triclosan-coated sutures for abdominal wall closure and incisional surgical site infection after open surgery in patients presenting with fecal peritonitis: a randomized clinical trial. Surg Infect (Larchmt) 16(5):588–594. https://doi.org/10.1089/sur.2014.072
Diener MK, Knebel P, Kieser M, Schuler P, Schiergens TS, Atanassov V, Neudecker J, Stein E, Thielemann H, Kunz R, von Frankenberg M, Schernikau U, Bunse J, Jansen-Winkeln B, Partecke LI, Prechtl G, Pochhammer J, Bouchard R, Hodina R, Beckurts KT, Leissner L, Lemmens HP, Kallinowski F, Thomusch O, Seehofer D, Simon T, Hyhlik-Durr A, Seiler CM, Hackert T, Reissfelder C, Hennig R, Doerr-Harim C, Klose C, Ulrich A, Buchler MW (2014) Effectiveness of triclosan-coated PDS Plus versus uncoated PDS II sutures for prevention of surgical site infection after abdominal wall closure: the randomised controlled PROUD trial. Lancet 384(9938):142–152. https://doi.org/10.1016/S0140-6736(14)60238-5
Justinger C, Slotta JE, Ningel S, Graber S, Kollmar O, Schilling MK (2013) Surgical-site infection after abdominal wall closure with triclosan-impregnated polydioxanone sutures: results of a randomized clinical pathway facilitated trial (NCT00998907). Surgery 154(3):589–595. https://doi.org/10.1016/j.surg.2013.04.011
Mingmalairak CUP, Paocharoen V (2009) Efficacy of antimicrobial coating suture coated polyglactin 910 with triclosan (Vicryl Plus) compared with polyglactin 910 (Vicryl) in reduced surgical site infection of appendicitis, double blind randomized control trial, preliminary safety report. J Med Assoc Thai 92(6):770–775
Health CAfDaTi (2014) Antibacterial sutures for wound closure after surgery: a review of clinical and cost-effectiveness and guidelines for use. In. CADTH Rapid Response Reports. Ottawa
Sandini M, Mattavelli I, Nespoli L, Uggeri F, Gianotti L (2016) Systematic review and meta-analysis of sutures coated with triclosan for the prevention of surgical site infection after elective colorectal surgery according to the PRISMA statement. Medicine (Baltimore) 95(35):e4057. https://doi.org/10.1097/MD.0000000000004057
Elsolh B, Zhang L, Patel SV (2017) The effect of antibiotic-coated sutures on the incidence of surgical site infections in abdominal closures: a meta-analysis. J Gastrointest Surg 21(5):896–903. https://doi.org/10.1007/s11605-017-3357-6
Konstantelias AA, Andriakopoulou CS, Mourgela S (2017) Triclosan-coated sutures for the prevention of surgical-site infections: a meta-analysis. Acta Chir Belg 117(3):137–148. https://doi.org/10.1080/00015458.2017.1287396
Sorensen L, Hemmingsen U, Kirkeby L, Kallehave F, Jorgensen L (2005) Smoking is a risk factor for incisional hernia. Arch Surg 140(2):119–123. https://doi.org/10.1001/archsurg.140.2.119
Millbourn D, Cengiz Y, Israelsson LA (2009) Effect of stitch length on wound complications after closure of midline incisions: a randomized controlled trial. Arch Surg 144(11):1056–1059. https://doi.org/10.1001/archsurg.2009.189
Gislason H, Gronbech JE, Soreide O (1995) Burst abdomen and incisional hernia after major gastrointestinal operations—comparison of three closure techniques. Eur J Surg 161(5):349–354
Wissing J, van Vroonhoven TJ, Schattenkerk ME, Veen HF, Ponsen RJ, Jeekel J (1987) Fascia closure after midline laparotomy: results of a randomized trial. Br J Surg 74(8):738–741
Gys T, Hubens A (1989) A prospective comparative clinical study between monofilament absorbable and non-absorbable sutures for abdominal wall closure. Acta Chir Belg 89(5):265–270
Velnar T, Bailey T, Smrkolj V (2009) The wound healing process: an overview of the cellular and molecular mechanisms. J Int Med Res 37(5):1528–1542
Muysoms FE, Antoniou SA, Bury K, Campanelli G, Conze J, Cuccurullo D, de Beaux AC, Deerenberg EB, East B, Fortelny RH, Gillion JF, Henriksen NA, Israelsson L, Jairam A, Janes A, Jeekel J, Lopez-Cano M, Miserez M, Morales-Conde S, Sanders DL, Simons MP, Smietanski M, Venclauskas L, Berrevoet F, European Hernia S (2015) European Hernia Society guidelines on the closure of abdominal wall incisions. Hernia 19(1):1–24. https://doi.org/10.1007/s10029-014-1342-5
Deerenberg EB, Harlaar JJ, Steyerberg EW, Lont HE, van Doorn HC, Heisterkamp J, Wijnhoven BP, Schouten WR, Cense HA, Stockmann HB, Berends FJ, Dijkhuizen FP, Dwarkasing RS, Jairam AP, van Ramshorst GH, Kleinrensink GJ, Jeekel J, Lange JF (2015) Small bites versus large bites for closure of abdominal midline incisions (STITCH): a double-blind, multicentre, randomised controlled trial. Lancet 386(10000):1254–1260. https://doi.org/10.1016/S0140-6736(15)60459-7
Ahmed I, Sutton AJ, Riley RD (2012) Assessment of publication bias, selection bias, and unavailable data in meta-analyses using individual participant data: a database survey. BMJ 344:d7762. https://doi.org/10.1136/bmj.d7762
Funding
No funding was received for the current work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
NH, ED, LV, RF and JG declare no conflict of interest. MM declares conflict of interest not directly related to the submitted work: grants and personal fees (speaker) from Aspide, Bard and Gore and is editor-in-chief of Hernia Journal, Springer. FM declares conflict of interest not directly related to the submitted work: grants and personal fees (consultancy and speaker) from Medtronic and Dynamesh and proctoring for Intuitive Surgical.
Ethical approval
This study did not need approval from the local ethical committee.
Human and animal rights
This study does not contain any studies with participants or animals performed by any of the authors.
Informed consent
Informed consent was not required for this study.
Rights and permissions
About this article
Cite this article
Henriksen, N.A., Deerenberg, E.B., Venclauskas, L. et al. Triclosan-coated sutures and surgical site infection in abdominal surgery: the TRISTAN review, meta-analysis and trial sequential analysis. Hernia 21, 833–841 (2017). https://doi.org/10.1007/s10029-017-1681-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10029-017-1681-0