Abstract
Objective
This meta-analysis aims to determine if antibiotic-impregnated sutures for abdominal fascial closure prevent postoperative surgical site infections (SSIs), hernias, and/or dehiscence.
Methods
MEDLINE and EMBASE databases (1946–2016) were searched. Randomized controlled trials comparing antibiotic-impregnated sutures to standard sutures for abdominal closure were eligible. Risk of bias was evaluated using the Cochrane Handbooks definitions.
Results
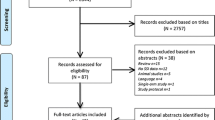
Four-hundred fifty articles were reviewed; five eligible studies (N = 3117) were identified. All studies routinely used prophylactic antibiotics. Overall risk of SSI in the antibiotic-impregnated suture group was 10.4 vs. 13.0% in the control group. Pooled data showed no difference in SSI between suture types (odds ratio 0.79, 95% CI 0.57–1.09, P = 0.15, I 2 = 44%). There was no evidence of subgroup effect by suture material (polydioxanone vs. polyglactin 910; P = 0.19) or by comparing colorectal surgery studies to others (P = 0.67). There was a high risk of bias in two studies, one for high loss to follow-up and one for not using an intent-to-treat analysis.
Conclusion
Our meta-analysis is the most comprehensive review on the utility of antibiotic-impregnated sutures in abdominal surgery to prevent SSI. We found no evidence to support routine use of these sutures.





Similar content being viewed by others
References
Watanabe A, Kohnoe S, Shimabukuro R. Risk factors associated with surgical site infection in upper and lower gastrointestinal surgery. Surgery today. 38(5):404–12. 2008.
Magill SS, Hellinger W, Cohen J. Prevalence of healthcare-associated infections in acute care hospitals in Jacksonville, Florida. Infection control and hospital epidemiology. 33(3):283–91. 2012.
Dumville JC, Gray TA, Walter CJ, Sharp CA, Page T. Dressings for the prevention of surgical site infection. The Cochrane database of systematic reviews. 9:CD003091. 2014.
Mangram AJ, Horan TC, Pearson ML, Silver LC, Jarvis WR. Guideline for Prevention of Surgical Site Infection, 1999. Centers for Disease Control and Prevention (CDC) Hospital Infection Control Practices Advisory Committee. American journal of infection control. 27(2):97–132; quiz 133–4; discussion 96. 1999.
McLaws ML, Taylor PC. The Hospital Infection Standardised Surveillance (HISS) programme: analysis of a two-year pilot. The Journal of hospital infection. 53(4):259–67. 2003.
Leaper DJ, van Goor H, Reilly J. Surgical site infection - a European perspective of incidence and economic burden. International wound journal. 1(4):247–73. 2004.
Seiler CM, Bruckner T, Diener MK. Interrupted or continuous slowly absorbable sutures for closure of primary elective midline abdominal incisions: a multicenter randomized trial (INSECT: ISRCTN24023541). Annals of surgery. 249(4):576–82. 2009.
Barbolt TA. Chemistry and safety of triclosan, and its use as an antimicrobial coating on Coated VICRYL* Plus Antibacterial Suture (coated polyglactin 910 suture with triclosan). Surgical infections. 3 Suppl 1:S45-53. 2002.
Ming X, Rothenburger S, Nichols MM. In vivo and in vitro antibacterial efficacy of PDS plus (polidioxanone with triclosan) suture. Surgical infections. 9(4):451–7. 2008.
Katz S, Izhar M, Mirelman D. Bacterial adherence to surgical sutures. A possible factor in suture induced infection. Annals of surgery. 194(1):35–41. 1981.
Wang ZX, Jiang CP, Cao Y, Ding YT. Systematic review and meta-analysis of triclosan-coated sutures for the prevention of surgical-site infection. The British journal of surgery. 100(4):465–73. 2013.
Baracs J, Huszár O, Sajjadi SG, Horváth OP. Surgical site infections after abdominal closure in colorectal surgery using triclosan-coated absorbable suture (PDS Plus) vs. uncoated sutures (PDS II): a randomized multicenter study. Surgical infections. 12(6):483–9. 2011.
Diener MK, Knebel P, Kieser M. Effectiveness of triclosan-coated PDS Plus versus uncoated PDS II sutures for prevention of surgical site infection after abdominal wall closure: the randomised controlled PROUD trial. Lancet (London, England). 384(9938):142–52. 2014.
Justinger C, Slotta JE, Ningel S, Gräber S, Kollmar O, Schilling MK. Surgical-site infection after abdominal wall closure with triclosan-impregnated polydioxanone sutures: results of a randomized clinical pathway facilitated trial (NCT00998907). Surgery. 154(3):589–95. 2013.
Mattavelli I, Rebora P, Doglietto G. Multi-Center Randomized Controlled Trial on the Effect of Triclosan-Coated Sutures on Surgical Site Infection after Colorectal Surgery. Surgical infections. 16(3):226–35. 2015.
Nakamura T, Kashimura N, Noji T. Triclosan-coated sutures reduce the incidence of wound infections and the costs after colorectal surgery: a randomized controlled trial. Surgery. 153(4):576–83. 2013.
Higgins JPT, Green S(eds). Cochrane Handbook for Systematic Reviews of Interventions. The Cochrane Collaboration, 2011.
Review Manager (RevMan) [Computer program]. Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Guyatt GH, Oxman AD, Vist GE. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ (Clinical research ed.). 336(7650):924–6. 2008.
Kamel C, McGahan L, Mierqwinski-Urban M, Embil J. Preoperative Skin Antiseptic Preparations and Application Techniques for Preventing Surgical Site Infections: A Systematic Review of the Clinical Evidence and Guidelines. CADTH Rapid Response Reports 2011.
Noorani A, Rabey N, Walsh SR, Davies RJ. Systematic review and meta-analysis of preoperative antisepsis with chlorhexidine versus povidone-iodine in clean-contaminated surgery. The British journal of surgery. 97(11):1614–20. 2010.
Nelson RL, Gladman E, Barbateskovic M. Antimicrobial prophylaxis for colorectal surgery. The Cochrane database of systematic reviews. 2014.
Antibacterial sutures for wound closure after surgery: a review of clinical and cost-effectiveness and guidelines for use. Canadian Agency for Drugs and Technologies in Health. CADTH Rapid Response Reports. 2014. https://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0071281/
Author Contribution
All authors contributed to designing the study, acquiring and analyzing data, and drafting/revising the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Sources of Funding/Support
None.
Rights and permissions
About this article
Cite this article
Elsolh, B., Zhang, L. & Patel, S.V. The Effect of Antibiotic-Coated Sutures on the Incidence of Surgical Site Infections in Abdominal Closures: a Meta-Analysis. J Gastrointest Surg 21, 896–903 (2017). https://doi.org/10.1007/s11605-017-3357-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-017-3357-6