Abstract
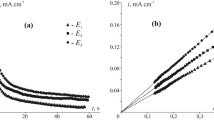
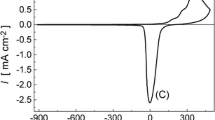
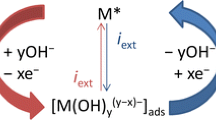
Multilayer oxide films were grown on silver in base by repetitive potential cycling; however, the type of oxide obtained, as assessed on the basis of its reduction behaviour, was dependent on the lower limit of the oxide growth cycles. Using limits of 1.03–2.60 V (RHE) the oxide film produced was assumed to be predominantly Ag2O; reduction of the latter yielded a cathodic peak at ca. 0.8 V and a surface layer of silver microparticles of diameter ranging from ca. 100 to 227 nm which, although relatively stable, were prone to rapid, extensive reoxidation. Altering the oxide growth limits to 0.7–2.60 V resulted in the growth of a different type of oxide deposit which is assumed to be AgOH; reduction of the latter occurred in a negative sweep in a random manner, i.e. in the form of cathodic spikes extending to potentials as low as ca. –0.5 V. Both types of silver oxide species are assumed to be involved in premonolayer oxidation and electrocatalysis at silver in base and the nature of the former process is discussed in some detail.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Nagle, L.C., Ahern, A.J. & Burke, D.L. Some unusual features of the electrochemistry of silver in aqueous base. J Solid State Electrochem 6, 320–330 (2002). https://doi.org/10.1007/s100080100233
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s100080100233