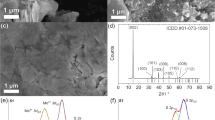
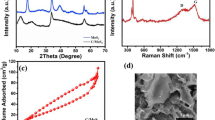
Abstract
Choice of binder and the electrode-making process play a pivotal role in the electrochemical performance of MoS2, when used as lithium-ion battery anode. In this work, MoS2 nanorods are prepared by gas phase synthesis method using molybdenum trioxide (MoO3) nanobelts and sulfur as starting materials. It has been observed that by tuning the reaction conditions, morphology and yield of the final product can be controlled. Carboxymethyl cellulose (CMC) is used as binder to fabricate the MoS2 electrode, and its electrochemical performance is tested against Li/Li+. The performance of electrode can be further improved by incorporating heat treatment to the active material and conductive carbon mixture prior to electrode fabrication. The electrochemical data shows that the optimum temperature for heat treatment is 700 °C. In the current report, we would like to elucidate a detailed study based on electrode fabrication process and their impact on the electrochemical performance.








Similar content being viewed by others
References
Poizot P, Laruelle S, Grugeon S, Dupont L, Tarascon JM (2000) Nature 407:496–499
Sen UK, Srakar S, Veluri PS, Singh S, Mitra S (2013) Nanosci Nanotechnol Asia 3:21–35
Bruce PG, Scrosati B, Tarascon JM (2008) Angew Chem Int Ed 47:2930–2946
Taberna PL, Mitra S, Poizot P, Simon P, Tarascon JM (2006) Nat Mater 5:567–573
Reddy MV, Yu T, Sow CH, Shen ZX, Lim CT, Rao GVS, Chowdari BVR (2007) Adv Funct Mater 17:2792–2799
Chang K, Chen W (2011) ACS Nano 6:4720–4728
Cao X, Shi Y, Shi W, Rui X, Yan Q, Kong J, Zhang H (2013) Small 9:3433–3438
Wang Z, Chen T, Chen W, Chang K, Ma L, Huang G, Chen D, Lee JY (2013) J Mater Chem A 1:2202–2210
Bindumadhavan K, Srivastava SK, Mahanty S (2013) Chem Commun 49:1823–1825
Park SK, Yu SH, Woo S, Quan B, Lee DC, Kim MK, Sung YE, Piao Y (2013) Dalton Trans 42:2399–2405
Chang K, Chen W, Ma L, Li H, Li H, Huang F, Xu Z, Zhangd Q, Lee JY (2011) J Mater Chem 21:6251–6257
Zhou X, Wan LJ, Guo YG (2012) Nanoscale 4:5868–5871
Sen UK, Mitra S (2013) ACS Appl Mater Interfaces 5:1240–1247
Li J, Dahn HM, Krause LJ, Le DB, Dahn JR (2008) J Electrochem Soc 155:A812–A816
Liu G, Zheng H, Kim S, Deng Y, Minor AM, Song X, Battaglia VS (2008) J Electrochem Soc 155:A887–A892
Liu G, Zheng H, Simens AS, Minor AM, Song X, Battaglia VS (2007) J Electrochem Soc 154:A1129–A1134
Veluri PS, Mitra S (2013) RSC Adv 3:15132–15138
Wang Z, Madhavi S, Lou XW (2012) J Phys Chem C 116:12508–12513
Gillot F, Boyanov S, Dupont L, Doublet ML, Morcrette M, Monconduit L, Tarascon JM (2005) Chem Mater 17:6327–6337
Gu Y, Xu Y, Wang Y (2013) ACS Appl Mater Interfaces 5:801–806
Wang Y, Wu J, Tang Y, Lü X, Yang C, Qin M, Huang F, Li X, Zhang X (2012) ACS Appl Mater Interfaces 4:4246–4250
Liu H, Su D, Wang G, Qiao SZ (2012) J Mater Chem 22:17437–17440
Bhandavat R, David L, Singh G (2012) J Phys Chem Lett 3:1523–1530
Feldman Y, Wasserman E, Srolovitz DJ, Tenne R (1995) Science 267:222–225
Li XL, Li YD (2003) Chem Euo J 9:2726–2731
Hsu WK, Chang BH, Zhu YQ, Han WQ, Terrones H, Terrones M, Grobert N, Cheetham AK, Kroto HW, Walton DRM (2000) J Am Chem Soc 122:10155–10158
Chen Z, Cummins D, Reinecke BN, Clark E, Sunkara MK, Jaramillo TF (2011) Nano Lett 11:4168–4175
Sen UK, Mitra S (2012) RSC Adv 2:11123–11131
Wang M, Li G, Xu H, Qian Y, Yang J (2013) ACS Appl Mater Interfaces 5:1003–1008
Shi YM, Wang Y, Wong JI, Tan AYS, Hsu CL, Li LJ, Lu YC, Yang HY (2013) Sci Rep 3:2169
Stephenson T, Li Z, Olsen B, Mitlin D (2014) Energy Environ Sci 7:209–231
Ji X, Nazar LF (2010) J Mater Chem 20:9821–9826
Mabuchi A, Tokumitsu K, Fujimoto H, Kasuh T (1995) J Electrochem Soc 142:1041–1046
Andrews R, Jacques D, Qian D, Dickey EC (2001) Carbon 39:1681–1687
Endo M, Kim YA, Hayashi T, Yanagisawa T, Muramatsu H, Ezaka M, Terrones H, Terrones H, Dresselhaus MS (2003) Carbon 41:1941–1947
Ataca C, Şahin H, Aktürk E, Ciraci S (2011) J Phys Chem C 115:3934–3941
Ghorbani-Asl M, Enyashin AN, Kuc A, Seifert G, Heine T (2013) Phys Rev B 88(245440):1–7
Acknowledgments
We thank the “National Centre for Photovoltaic Research and Education (NCPRE),” Ministry of New and Renewable Energy, Govt. of India, and IRCC-IIT Bombay for financial support and infrastructural facilities. The authors are thankful to the members of SAIF, IIT Bombay, for their assistance with electron diffraction and FEG-SEM analysis.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 1687 kb)
Rights and permissions
About this article
Cite this article
Sen, U.K., Mitra, S. Improved electrode fabrication method to enhance performance and stability of MoS2-based lithium-ion battery anode. J Solid State Electrochem 18, 2701–2708 (2014). https://doi.org/10.1007/s10008-014-2518-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-014-2518-8