Abstract
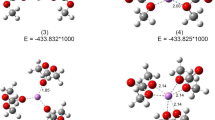
Electrolyte solutions based on the propylene carbonate (PC)-dimethoxyethane (DME) mixtures are of significant importance and urgency due to emergence of lithium-ion batteries. Solvation and coordination of the lithium cation in these systems have been recently attended in detail. However, analogous information concerning anions (tetrafluoroborate, hexafluorophosphate) is still missed. This work reports PM7-MD simulations (electronic-structure level of description) to include finite-temperature effects on the anion solvation regularities in the PC-DME mixture. The reported result evidences that the anions appear weakly solvated. This observation is linked to the absence of suitable coordination sites in the solvent molecules. In the concentrated electrolyte solutions, both BF4 − and PF6 − prefer to exist as neutral ion pairs (LiBF4, LiPF6).






Similar content being viewed by others
References
Barthel J, Gores HJ, Neueder R, Schmid A (1999) Electrolyte solutions for technology — new aspects and approaches. Pure Appl Chem 71:1705–1715
Ganesh P, Jiang DE, Kent PRC (2011) Accurate static and dynamic properties of liquid electrolytes for li-ion batteries from ab initio molecular dynamics. J Phys Chem B 115:3085–3090
Borodin O, Smith GD (2009) Quantum chemistry and molecular dynamics simulation study of dimethyl carbonate: ethylene carbonate electrolytes doped with Lipf6. J Phys Chem B 113:1763–1776
Singh HJ, Mukherjee U (2013) A computational approach to design energetic ionic liquids. J Mol Model 19:2317–2327
Zhang Q, Zhang X, Zhao DX (2013) Ion disturbance and clustering in the NaCl water solutions. J Mol Model 19:661–672
Lai WP, Lian P, Liu YZ, Yu T, Zhu WL, Ge ZX, Lv J (2014) Design and theoretical study of 15 novel high energy density compounds. J Mol Model 20:2479
Zhu XY, Sun H, Zhang DJ, Liu CB (2011) Theoretical study on the interactions between methanol and imidazolium-based ionic liquids. J Mol Model 17:1997–2004
Darvas M, Jorge M, Cordeiro MNDS, Jedlovszky P (2011) Solvation free energy profile of the Scn- ion across the water-1,2-dichloroethane liquid/liquid interface. A computer simulation study. J Phys Chem C 115:11140–11146
Gulaboski R, Pereira CM, Cordeiro MNDS, Bogeski I, Ferreira E, Ribeiro D, Chirea M, Silva AF (2005) Electrochemical study of ion transfer of acetylcholine across the interface of water and a lipid-modified 1,2-dichloroethane. J Phys Chem B 109:12549–12559
Videa M, Xu W, Geil B, Marzke R, Angell CA (2001) High Li+ self-diffusivity and transport number in novel electrolyte solutions. J Electrochem Soc 148:A1352–A1356
Sun X, Lee HS, Yang XQ, McBreen J (2002) A New Additive for Lithium Battery Electrolytes Based on an Alkyl Borate Compound. J Electrochem Soc 149:A355–A359
Soetens JC, Millot C, Maigret B (1998) Molecular new additive for lithium battery electrolytes based on an alkyl borate compound. J Phys Chem A 102:1055–1061
Azeez F, Fedkiw PS (2010) Conductivity of Libob-Based Electrolyte for Lithium-Ion Batteries. J Power Sources 195:7627–7633
Postupna OO, Kolesnik YV, Kalugin ON, Prezhdo OV (2011) Microscopic structure and dynamics of Libf4 solutions in cyclic and linear carbonates. J Phys Chem B 115:14563–14571
Barthel J, Neueder R, Roch H (2000) Density, relative permittivity, and viscosity of propylene carbonate plus dimethoxyethane mixtures from 25 degrees C to 125 degrees C. J Chem Eng Data 45:1007–1011
Kameda Y, Umebayashi Y, Takeuchi M, Wahab MA, Fukuda S, Ishiguro SI, Sasaki M, Amo Y, Usuki T (2007) Solvation structure of Li+ in concentrated Lipf6-propylene carbonate solutions. J Phys Chem B 111:6104–6109
Chaban V (2014) Competitive solvation of (bis)(trifluoromethanesulfonyl)imide anion by acetonitrile and water. Chem Phys Lett 613:90–94
Chaban V (2015) The thiocyanate anion is a primary driver of carbon dioxide capture by ionic liquids. Chem Phys Lett 618:89–93
Chaban V (2015) Annealing relaxation of ultrasmall gold nanostructures. Chem Phys Lett 618:46–50
Andreeva NA, Chaban VV (2015) Global minimum search via annealing: nanoscale gold clusters. Chem Phys Lett 622:75–79
Stewart JJP (2007) Optimization of parameters for semiempirical methods v: modification of Nddo approximations and application to 70 elements. J Mol Model 13:1173–1213
Stewart JJP (2008) Application of the Pm6 method to modeling the solid state. J Mol Model 14:499–535
Stewart JJP (2009) Application of the Pm6 method to modeling proteins. J Mol Model 15:765–805
Stewart JJP (2013) Optimization of parameters for semiempirical methods Vi: more modifications to the Nddo approximations and re-optimization of parameters. J Mol Model 19:1–32
Berendsen HJC, Postma JPM, Vangunsteren WF, Dinola A, Haak JR (1984) Molecular-dynamics with coupling to an external bath. J Chem Phys 81:3684–3690
Nielson KD, van Duin ACT, Oxgaard J, Deng WQ, Goddard WA (2005) Development of the reaxff reactive force field for describing transition metal catalyzed reactions, with application to the initial stages of the catalytic formation of carbon nanotubes. J Phys Chem A 109:493–499
van Duin ACT, Strachan A, Stewman S, Zhang QS, Xu X, Goddard WA (2003) Reaxff(Sio) Reactive force field for silicon and silicon oxide systems. J Phys Chem A 107:3803–3811
van Duin ACT, Dasgupta S, Lorant F, Goddard WA (2001) Reaxff: a reactive force field for hydrocarbons. J Phys Chem A 105:9396–9409
Zhang, Qing and Çağin, Tahir and van Duin, Adri and Goddard, William A, III and Qi, Yue and Hector, Louis G, Jr (2004) Adhesion and nonwetting-wetting transition in the Al/alpha-Al2O3 interface. Phys Rev B 69(4). doi:10.1103/PhysRevB.69.045423
Allouche AR (2011) Gabedit-a graphical user interface for computational chemistry softwares. J Comput Chem 32:174–182
Acknowledgments
V.V.C. is a CAPES fellow in Brazil under the “Science Without Borders” program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chaban, V. Solvation of the fluorine containing anions and their lithium salts in propylene carbonate and dimethoxyethane. J Mol Model 21, 172 (2015). https://doi.org/10.1007/s00894-015-2717-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-015-2717-y