Abstract
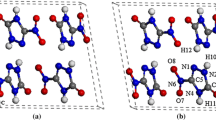
DFT calculations have been performed to study the structural, electronic, absorption, and thermodynamic properties of crystalline 1,1-diamino-2,2-dintroethylene (α-FOX-7) in the pressure range of 0–40 GPa. A comprehensive analysis of the variation trends of the lattice constants, bond lengths, bond angles, and twist angles under compression shows that six structural transformations occur in α-FOX-7 at 2, 5, 11, 19, 29, and 35 GPa, respectively. The C1-N1 and C1-N2 bond lengths decrease much faster than any other bonds under compression, indicating that the C-NO2 cleavage is possible to trigger the decomposition of α-FOX-7. The intra-molecular H-bonding interaction weakens at 2 and 5 GPa, which may be caused by the structural transformations, but it then strengthens with the increasing pressure up to 40 GPa. The inter-molecular H-bonding interaction strengthens with the increasing pressure. The band gap of α-FOX-7 increases at 11 GPa suddenly and decreases obviously at 19, 29, and 35 GPa, which are caused by the structural transformations. α-FOX-7 has relatively high optical activity at high pressure. All the structural transformations are endothermic and not spontaneous at room temperature.










Similar content being viewed by others
References
Bemm U, Östmark H (1998) Acta Crystallogr C: Cryst Struct Commun 54:1997–1999
Kjellstrom A, Latypov N, Eldsater C, Eriksson L (2005) FOI Swedish Defense Research Agency Technical Reprot No. SE-147 25 Tumba
Evers J, Klapötke TM, Mayer P, Oehlinger G, Welch JM (2006) Inorg Chem 45:4996–5007
Crawford MJ, Evers J, Göbel M, Klapötke TM, Mayer P, Oehlinger G, Welch JM (2007) Propell Explos Pyrot 32:478–495
Herve G, Jacob G, Latypov NV (2005) Tetrahedron 61:6743–6748
Peiris S, Wong C, Zerilli F, Russell T (2001) Shock Comp Cond Matter CP620:181–184
Hu A, Larade B (2006) Propell Explos Pyrot 31:355–360
Politzer P, Concha MC, Grice ME, Murray JS, Lane P (1998) J Mol Struct (THEOCHEM) 452:75–83
Gindulyte A, Massa L, Huang L, Karle J (1999) J Phys Chem A 103:11045–11051
Ji GF, Xiao HM, Dong HS, Gong XD, Li JS, Wang ZY (2001) Acta Chim Sin 59:39–47
Sorescu DC, Boatz JA, Thompson DL (2001) J Phys Chem A 105:5010–5021
Ju XH, Xiao HM, Xia QY (2003) J Chem Phys 119:10247–10255
Gilardi R (1999) CCCD 127539, Cambridge Structural Database, Cam-bridge Crystallographic Data Center, Cambridge, UK
Zerilli FJ, Kuklja M (2007) J Phys Chem A 111:1721–1725
Trzciński WA, Cudziło S, Chyłek Z, Szymańczyk L (2008) J Hazard Mater 15:605–612
Zerilli FJ (2006) J Phys Chem A 110:5173–5179
Fabbiani FPA, Pulham CR (2006) Chem Soc Rev 35:932–942
Cady HH, Smith LC (1961) Los Alamos Scientific Laboratory Repot LAMS-2653 TID-4500; Los Almos National Laboratory: Los Alamos, NM
Main P, Cobbledic RE, Small RWH (1985) Acta Crystallogr Sect C 41:1351–1354
Dreger ZA, Gupta YM (2010) J Phys Chem A 114:8099–8105
Dreger ZA, Gupta YM (2007) J Phys Chem B 111:3893–3903
Pravica M, Liu Y, Robinson J, Velisavljevic N, Liu ZX, Galley M (2012) J Appl Phys 111:103524–103529
Peiris S, Wong C, Zerilli F (2004) J Chem Phys 120:8060–8066
Peiris S, Wong C, Kuklja M, Zerilli F (2002) 12th Int Deton Symp Pro, pp 12.0617-12.0624
Brangham J, Pravica M, Galley M (2011) Undergergraduate Research Opportunities Program (UROP). pp 23 http://diaitalscholarship.unlv.edu/cs_urop/2011/aug9/23
Zhao J, Liu H (2008) Comput Mater Sci 42:698–703
Zhu WH, Zhang XW, Wei T, Xiao HM (2009) Theor Chem Acc 124:179–186
Zhu WH, Zhang XW, Zhu W, Xiao HM (2008) Phys Chem Chem Phys 10:7318–7323
Zhu WH, Xiao JJ, Xiao HM (2006) Chem Phys Lett 422:117–121
Zhu WH, Xiao HM (2006) J Phys Chem B 110:18196–18203
Segall MD, Lindan PJD, Probert MJ, Pickard CJ, Hasnip PJ, Clark SJ, Payne MC (2002) J Phys Condens Matter 14:2717–2744
Vanderbilt D (1990) Phys Rev B 41:7892–7895
Kresse G, Furthmüller J (1996) Phys Rev B 54:11169–11186
Fletcher R (1980) Practical methods of optimization, vol 1. Wiley, New York
Ceperley DM, Alder BJ (1980) Phys Rev Lett 45:566–569
Perdew JP, Zunger A (1981) Phys Rev B 23:5048–5079
Yoo C-S, Cynn H (1999) J Chem Phys 111:10229–10235
Gump JC, Peiris SM (2005) J Appl Phys 97:053513
Zhu WH, Xiao HM (2009) J Phys Chem B 113:10315–10321
Zhu WH, Wei T, Zhu W, Xiao HM (2008) J Phys Chem A 112:4688–4693
Perdew JP, Chevary JA, Vosko SH, Jackson KA, Pederson MR, Singh DJ, Fiolhais C (1992) Phys Rev B 46:6671–6687
Zhu WH, Xiao JJ, Ji GF, Zhang F, Xiao HM (2007) J Phys Chem B 111:12715–12722
Xu XJ, Zhu WH, Xiao HM (2007) J Phys Chem B 111:2090–2097
Zhu WH, Xiao HM (2008) J Comput Chem 29:176–184
Zhu WH, Xiao HM (2010) Struct Chem 21:657–665
Saha S, Sinha TP, Mookerjee A (2000) Phys Rev B 62:8828–8834
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant No. 21273115) and A Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, Q., Zhu, W. & Xiao, H. DFT study on crystalline 1,1-diamino-2,2-dintroethylene under high pressures. J Mol Model 19, 4039–4047 (2013). https://doi.org/10.1007/s00894-013-1931-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-013-1931-8