Abstract
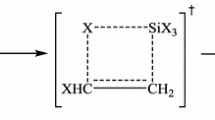
This study uses the Gaussian 03 program and density functional theory B3LYP with three basis set methods—[B3LYP/6-311+G(d,p), B3LYP/6-31+G(2d,p), and B3LYP/6-31G(d,p)]—to model the highly energetic ionic compound diguanidinium 5,5′-azotetrazolate (GZT) to research its decomposition mechanisms and thermodynamic properties. Molecular-type cracking patterns are proposed, which were initiated by heterocyclic ring opening, sequential cracking of the two five-membered rings of GZT, and simultaneous release of N2 molecules; whereas proton transfer, bond-breaking, and atomic rearrangements were performed subsequently. Finally, 15 reaction paths and five transition states were obtained. All possible decomposition species and transition states, including intermediates and products, were identified, and their corresponding enthalpy and Gibbs free energy values were obtained. The results revealed that (1) the maximum activation energy required is 187.8 kJ mol–1, and the enthalpy change (ΔH) and Gibbs free-energy change (ΔG) of the net reaction are −525.1 kJ mol–1 and −935.6 kJ mol–1, respectively; (2) GZT can release large amounts of energy, the main contribution being from the disintegration of the 5,5'-azotetrazolate anion (ZT2−) skeleton (ΔH = −598.3 kJ mol–1); and (3) the final products contained major amounts of N2 gas, but remaining gas molecules such as HCN and NH3 were obtained, which are in agreement with experimental results. The detailed decomposition simulation results demonstrated the feasibility of this method to calculate the energies of the thermodynamic reactions for the highly energetic GZT and predict the most feasible pathways and the final products.








Similar content being viewed by others
References
Bucerius KM (1993) Stable nitrogen-rich composition: U.S. Pat. 5,198,046
Thiele J (1892) Ueber nitro-und amidoguanidin. Justus Liebigs Ann Chem 270:1–63
Hiskey MA, Goldman N, Stine JR (1998) High-nitrogen energetic materials derived from azotetrazolate. J Energ Mater 16:119–127
Hammerl A, Holl G, Kaiser M, Klapötke TM et al (2001) Methylated Ammonium and Hydrazinium Salts of 5,5'-azotetrazolate. Z Naturforsch 56:847–856
Yang SQ, Yue ST (2003) Progress in high-nitrogen energetic materials derived from tetrazine and tetrazole. Chin J Energ Mater 11:231–235
Yang SQ, Xu SL, Lei YP (2006) Development of nitrogen heterocyclic energetic compounds. Chin J Energ Mater 14:475–484
Bucerius KM, Eisenreich N, Schmid H, Engel W (1997) Gas generating mixture containing copper diammine dinitrate. U.S. Pat. 5,663,524
Schmid H, Eisenreich N (2000) Investigation of a two-stage airbag module with azide-free gas generators. Propellants Explos Pyrotech 25:230–235
Khandhadia PS, Burns SP (2001) Thermally stable nonazide automotive airbag propellants. US 6:306,232
Walsh CM, Knott CD, Leveritt CS (2006) Reduced erosion additive for a propelling charge. US 6:984,275
Hudson MK, Wright AM, Luchini C, Wynne PC et al (2004) Guanidinium Azo-Tetrazolate (GAT) as a High Performance Hybrid Rocket Fuel Additive. J Pyrotech 19:37–42
Damse RS, Siker AK (2009) Suitability of nitrogen rich compounds for gun propellant formulations. J Hazard Mater 166:967–971
Peng YL, Wong CW (1999) Preparation guanidinium 5,5'-azotetrazolate. U.S. Pat. 5,877,300
Hammerl A, Holl G, Klapötke TM, Mayer P et al (2002) Salts of 5,5'-azotetrazolate. Eur J Inorg Chem 4:834–845
Bucerius KM (1992) Guanidinium-5,5'-azo-tetrazolate - is stable nitrogen-rich cpd. yielding non-harmful gases upon decomposition. Ger. Pat. DE4,034,645
Bucerius KM, Wasmann FW, Menke K (1991) Pyrotechnical compositions for the generation of environmentally safe, nontoxic gases. Ger. Pat. DE4,108,225
Xu SL, Yang SQ (2006) Synthesis and properties of high-nitrogen energetic compounds based on azotetrazolate nonmetallic salts. Chin J Energ Mater 14:377–340
Xu SL, Yang SQ, Yue ST (2005) Synthesis and characterization of high-nitrogen energetic compounds derived from azotetrazolate. Chin J Explos Propell 28:52–55
Wang HS, Du ZM (2005) Progress in synthesis and properties of nitrogen-rich compounds. Chin J Energ Mater 13:196–199
Hammerl A, Hiskey MA, Holl G, Klapötke TM (2005) Azidoforma-midinium and guanidinium 5,5’-Azotetrazolate Salts. Chem Mater 17:3784–3793
Sivabalan R, Talawar MB, Senthilkumar N, Kavitha B et al (2004) Studies on azotetrazolate based high nitrogen content high energy materials potential additives for rocket propellants. J Them Anal Calorim 78:781–792
Damse RS, Naik NH, Ghosh M, Venugopalan S (2007) Structure-decomposition mechanism relationship for the energetic nitrogen rich compounds. 38th International Annual Conference of ICT 79-1-79-12
Tappan BC, Ali AN, Son SF, Brill TB (2006) Decomposition and Ignition of the High-Nitrogen Compound Triaminoguanidinium Azotetrazolate (TAGzT). Propell Explos Pyrot 31:163–168
Damse RS, Naik NH, Ghosh M, Sikder AK (2009) Thermoanalytical Screening of Nitrogen-Rich Compounds for Ballistic Requirements of Gun Propellant. J Propul Power 25:249–256
Miyata Y, Hasue K (2013) Thermal decomposition of aminoguanidinium 5,5’-azobis-1H-tetrazolate. Thermochim Acta 553:68–77
Alavi S, Reilly LM, Thompson DL (2003) Theoretical predictions of the decomposition mechanism of 1, 3, 3-trinitroazetidine (TNAZ). J Chem Phys 119:8297–8304
Liu MH, Cheng SR, Cheng KF, Chen C (2008) Kinetics of decomposition pathways of an energetic GZT molecule. Int J Quant Chem 108:482–486
Chen C, Liu MH, Cheng SR, Wu LS (2003) Theoretical study of the inter-ionic hydrogen bonding in the GZT molecular system. J Chin Chem Soc 50:765–775
Cheng C, Liu MH, Liu CW (2004) Theoretical study of the inter-ionic and inter-molecular hydrogen bonds constructed by GZT, ZT2- ions and their relative derivatives. J Mol Struct (THEOCHEM) 685:163–174
Storm CB, Ryan RR, Ritchie JP, Hall JH et al (1989) Structural Basis of the Impact Sensitivities of I-Picryl-I,2,3-triazole, 2-Picryl-I,2,3-triazole, 4-Nitro-I-picryl-I,2,3-triazole, and 4-Nitro-2-picryl-I,2,3-triazole. J Phys Chem 93:1000–1007
Politzer P, Grice ME, Seminario JM (1997) Density Functional Analysis of a Decomposition of 4-Nitro-1, 2, 3- Triazole Through the Evolution of N2. Int J Quant Chem 61:389–392
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE (2003) Gaussian 03, Revision B.03. Gaussian, Inc, Pittsburgh
Lee C, Yang W, Parr RG (1988) Development of the Colle-Salvetti Correlation-energy Formula into a Functional of the Electron Density. Phys Rev B37:785–789
Peng C, Schlegel HB (1993) Combining Synchronous Transit and Quasi-Newton Methods to Find Transition States. Israel J Chem 33:449–454
Peng C, Ayala PY, Schlegel HB, Frisch MJ (1998) Using Redundant Internal Coordinates to Optimize Equilibrium Geometries and Transition States. J Comput Chem 17:49–56
Herzberg G (1966) Infrared and Raman Spectra of Polyatomic Molecules. Florida, USA 279–295
Hammerl A, Klapötke TM, Nöth H, Warchhold M (2001) [N2H5]+2[N4C − N = N − CN4]2- : A New High-Nitrogen High-Energetic Material. Inorg Chem 40:3570–3575
Klapötke TM, Sabaté CM (2008) Bistetrazoles: Nitrogen-Rich, High-Performing, Insensitive Energetic Compounds. J Chem Mater 20:3629–3637
Klapötke TM, Sabaté CM (2008) Nitrogen-Rich Tetrazolium Azotetrazolate Salts: A New Family of Insensitive Energetic Materials. J Chem Mater 20:1750–1763
Sivabalan R, Anniyappan M, Pawar SJ, Talawar MB et al (2006) Synthesis, characterization and thermolysis studies on triazole and tetrazole based high nitrogen content high energy materials. J Hazard Mater A137:672–680
Fischer N, Klapötke TM, Scheutzow S, Stierstorfer J (2008) Hydrazinium 5-Aminotetrazolate: an Insensitive Energetic Material Containing 83.72% Nitrogen. Cent Eur J Energ Mater 5:3–18
Warner KF, Granholm HG (2011) Synthesis of Insensitive 5,5’-Azotetrazolate Salts. J Energ Mater 29:1–6
Klapötke TM, Miró Sabaté C (2009) New energetic compounds based on the nitrogen-rich 5,5′-azotetrazolate anion ([C2N10]2−). New J Chem 33:1605–1617
Laus G, Kahlenberg V, Wurst K, Schottenberger H et al (2012) Synthesis and Crystal Structures of New 5,5'-Azotetrazolates Crystals 2:127–136
Lin TP, Chen C (2000) Vibrational Study of Pyrazole Molecule. J Chin Chem Society 58:591–596
Acknowledgments
The authors would like to thank the National Center for High-Performance Computing for performing the calculations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cheng, SR., Cheng, KF., Liu, MH. et al. Computational study of decomposition mechanisms and thermodynamic properties of molecular-type cracking patterns for the highly energetic molecule GZT. J Mol Model 19, 3705–3717 (2013). https://doi.org/10.1007/s00894-013-1903-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-013-1903-z