Abstract
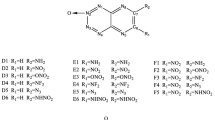
The derivatives of purine are designed through substituting the hydrogen atoms on it for nitro and amino functional groups. Geometries and frequency are analyzed at the B3LYP/6-31 G** level of density functional theory (DFT). Heats of formation (HOF), bond dissociation energy (BDE) and detonation parameters (detonation velocity and detonation pressure) are obtained in detail at the same level. It is found that the BDE values of all derivatives are over 120KJ·mol-1, and have high positive heats of formation. These derivatives possess excellent detonation properties, for B1, B2, and C, the detonation velocity are 9.58, 9.57, and 9.90 km·s-1, and the detonation pressure are 43.40, 46.05, and 46.37 Gpa, respectively, the detonation performances are better than cyclotrimethylenetrinitramine (RDX) and cyclotetramethylenetetranitramine (HMX). Hence, the derivations of purine may be promising well-behaved high energy density materials.

Similar content being viewed by others
References
Sewell TD, Menikoff R, Bedrov D, Smith GD (2003) J Chem Phys 119:7417–7426
Shalashilin DV, Thompson DL (1997) J Phys Chem A 101:961–966
Nielsen AT, Chafin AP, Christian SL, Moore DW, Nadler MP, Nissan RA, Vanderah DJ, Gilardi RD, George CF, Flippen-Anderson JL (1998) Tetrahedron 54:11793–11812
Wang GX, Gong XD, Du HC (2011) LiuY, Xiao HM. J Phys Chem A 115:795–804
Liu Y, Gong XD, Wang L, Wang GX, Xiao HM (2011) J Phys Chem A 115:1754–1762
Jadhav HS, Talawar MB, Sivabalan R, Dhavale DD, Asthana SN, Krishnamurthy VN (2007) J Hazard Mater 143:192–197
Jin RCGC, Kwang JK, Jeong KK (2000) Insensitive Munitions & Engergetic Materials Technology Symposium, p 393
Nurullah S (2007) Tetrahedron 63:4199–4236
Wade LG (2009) Organic Chemistry, 6th edn. Higher Education Press, China
Rosemeyer H (2007) Chem Biodivers 1:361–401
Hirschi JS, Arora K, Brooks CL 3rd, Schramm VL (2010) J Phys Chem B 114:16263–16272
Lecka J, Molski S, Komoszynski M (2010) Nucleos Nucleot Nucl 29:647–657
Sharma S, Das M, Kumar A, Marwaha V, Shankar S, Singh P, Raghu P, Aneja R, Grover R, Arya V, Dhir V, Gupta R, Kumar U, Juyal RC, Thelma BK (2009) Pharmacogn Genom 19:823–828
Zhao HL, Zhou LX (2011) Comput Theor Chem. doi:10.1016/j.comptc.2011.10.010
Verga D, Nadai M, Doria F, Percivalle C, Di Antonio M, Palumbo M, Richter SN, Freccero M (2010) J Am Chem Soc 132:14625–14637
Frisch MJ, Trucks GW, Schlegel BH et al (2004) Gaussian 03, Revision C02. Gaussian Inc, Wallingford, CT
Parr RG, Yang W (1989) Density Functional Theory of Atoms and Molecules[M]. Oxford University Press, New York
Hahre WJ, Ditchfield R, Pople JA (1972) J Chem Phys 56:2257
Woon DE, Dunning TH (1993) J Chem Phys 98:1358–1371
Xu XJ, Xiao HM, Ju XH et al (2006) J Phys Chem A 110:5929–5933
Chen ZX, Xiao JM, Xiao HM et al (1999) J Phys Chem A 103:8062–8066
Wei T, Zhu WH, Zhang JJ, Xiao HM (2010) J Hazard Mater 179:581–590
Nathanael SG, Soojin K, Peter GS (1997) Tetrahedron Lett 38:1161–1164
Owens FJ (1996) J Mol Struct (THEOCHEM) 370:11–16
Blanksby SJ, Ellison CB (2003) Acc Chem Res 36:255–263
Fan JF, Xiao HM (1996) J Mol Struct (THEOCHEM) 365:225–229
Kamlet M, Jocobs SJ (1968) J Chem Phys 48:23–35
Zhang JY, Du HC, Wang F, Gong XD, Huang YS (2011) J Phys Chem A 115:6617–6621
Wang GX, Shi CH, Gong XD, Xiao HM (2009) J Phys Chem A 113:1318–1326
Xiao HM, Xu XJ, Qiu L (2008) Theoretical design of high energy density materials. Science Press, Beijing
Ghule VD, Jadhav PM, Patil RS, Radhakrishanan S, Soman T (2010) J Phys Chem A 114:498–503
Rodenko B, Koch M, van der Burg AM, Wanner MJ, Koomen GJ (2005) J Am Chem Soc 127:5957–5963
Talawar MB, Sivabalan R, Mukundan T, Muthurajan H, Sikder AK, Gandhe BR, Subhananda Rao A (2009) J Hazard Mater 161:589–607
Acknowledgments
This work is supported by the Natural Science Foundation of Shanxi Province (No.2010021009-2), the Natural Science Foundation of China (No.20871077), the Research Project Supported by Shanxi Scholarship Council of China (No.201063) and the Natural Science Foundation of Shanxi Normal University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chi, WJ., Li, LL., Li, BT. et al. Density functional study on the derivatives of purine. J Mol Model 18, 3501–3506 (2012). https://doi.org/10.1007/s00894-012-1359-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-012-1359-6