Abstract
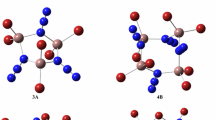
Ab initio methods based on density functional theory at BP86 level were applied to the study of the geometrical structures, relative stabilities, and electronic properties of small bimetallic Be2Au n (n = 1–9) clusters. The optimized geometries reveal that the most stable isomers have 3D structures at n = 3, 5, 7, 8, and 9. Here, the relative stabilities were investigated in terms of the averaged atomic binding energies, fragmentation energies and second-order difference of energies. The results show that the planar Be2Au4 structure is the most stable structure for Be2Au n clusters. The HOMO−LUMO gap, vertical ionization potential, vertical electron affinity and chemical hardness exhibit a pronounced even–odd alternating phenomenon. In addition, charge transfer and natural electron configuration were analyzed and compared.




Similar content being viewed by others
References
Ferrighi L, Hammer B, Madsen GKH (2009) J Am Chem Soc 131:10605–10609
Baruah T, Blundell SA, Zope RR (2001) Phys Rev A 64:043202–043209
Thomas OC, Zheng WJ, Lippa TP, Xu SJ, Lyapustina SA, Bowen KH (2001) J Chem Phys 114:9895–9900
Fantucci P, Bonačić-Koutecký V, Pewestorf W, Koutecký J (1989) J Chem Phys 91:456802
Li X, Kuznetsov AE, Zhang HF, Boldyrev AI, Wang LS (2001) Science 291:859–861
Yoon B, Häkkinen H, Landman U, Wörz AS, Antonietti JM, Abbet S, Judai K, Heiz U (2005) Science 307:403–407
Pal R, Wang LM, Huang W, Zeng XC (2009) J Am Chem Soc 131:3396–3404
Eachus RS, Marchetti AP, Muenter AA (1999) Annu Rev Phys Chem 50:117–123
Li XB, Wang HY, Yang XD, Zhu ZH (2007) J Chem Phys 126:084505
Scaffardi LB, Pellegri N, Sanctis Ode, Tocho JO (2005) Nanotechnology 16:158–163
Häkkinen H, Moseler M, Landman U (2002) Phys Rev Lett 89:033401
Wang F, Liu P, Zhang DJ (2010) J Mol Model. online first. doi:10.1007/s00894-010-0815-4
Torres MB, Fernández EM, Balbás LC (2008) J Phys Chem A 112:6678–6689
Hashmi ASK, Loos A, Littmann A, Braun I, Knight J, Doherty S, Rominger F (2009) Angew Chem 351:576–582
Chrétien SC, Buratto SK, Metiu H (2007) Curr Opin Solid State Mater Sci 11:62–75
Autschbach J, Hess BA, Johansson MP, Neugebauer J, Patzschke M, Pyykkö P, Reiher M, Sundholm D (2004) Phys Chem Chem Phys 6:11–22
Shaw CF III (1999) Chem Rev 99:2589–2600
Albonetti S, Bonelli R, Delaigle R, Femoni C, Gaigneaux EM, Morandi V, Ortolani L, Tiozzo C, Zacchini S, Trifiro F (2010) Appl Catal A 372:138–146
Neumaier M, Weigend F, Hamper O, Kappes MM (2006) J Chem Phys 125:104308
Félix C, Sieber C, Harbich W, Buttet J, Rabin I, Schulze W, Ertl G (2001) Phys Rev Lett 86:2992–2995
Kim SH, Medeiros-Ribeiro G, Ohlberg DAA, Stanley Wlliams R, Heath JR (1999) J Phys Chem B 103:10341–10347
Bishea GA, Arrington CA, Behm JM, Morse MD (1991) J Chem Phys 95:8765–8778
Janssens E, Tanaka H, Neukermans S, Silverans RE, Lievens P (2003) New J Phys 5:46.1–46.10
Negishi Y, Nakamura Y, Nakamura A, Kaya K (2001) J Chem Phys 115:3657–3663
Koyasu K, Naono Y, Akutsu M, Mitsui M, Nakajima A (2006) Chem Phys Lett 422:62–66
Heiz U, Yeretzian C, Stener M, Gisdakis P, Rösch N (1996) J Chem Phys 105:5574–5585
Heinebrodt M, Malinowski N, Tast F, Branz W, Billas IML, Martin TP (1999) J Chem Phys 110:9915–9921
Yuan DW, Wang Y, Zeng Z (2005) J Chem Phys 122:114310
Ghanty TK, Banerjee A, Chakrabarti A (2010) J Phys Chem C 114:20–27
Guo JJ, Yang JX, Die D (2005) Phys B 367:158–164
Guo JJ, Yang JX, Die D (2006) J Mol Struct THEOCHEM 764:117–121
Balducci G, Ciccioli A, Gigli G (2004) J Chem Phys 121:7748–7755
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millan JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2004) Gaussian 03, Revision D.01. Gaussian Inc, Wallingford, CT
Becke AD (1988) Phys Rev A 38:3098–3100
Perdew JP, Yue W (1986) Phys Rev B 33:8800–8802
Perdew JP, Wang Y (1992) Phys Rev B 45:13244–13249
Hay PJ, Wadt WR (1985) J Chem Phys 82:299–310
Ehlers AW, Bohme M, Dapprich S, Gobbi A, Hollwarth A, Jonas V, Kohler KF, Stegmann R, Veldkamp A, Frenking G (1993) Chem Phys Lett 208:111–114
Krishnan R, Binkley JS, Seeger R, Pople JA (1980) J Chem Phys 72:650–654
Barrow RF, Gissane WJG, Travis DN (1965) Proc R Soc London Ser A 287:240–251
Huber KP, Herzberg G (1979) Constants of diatomic molecules. Van Nostrand Reinhold, New York
Pearson RG (1997) Chemical hardness: applications from molecules to solids. Wiley-VCH, Weinheim
Parr RG, Yang W (1989) Density functional theory of atoms and molecules. Oxford University Press, New York
Jackslath C, Rabin I, Schulze W (1992) Ber Bunsenges Phys Chem 96:1200–1204
Acknowledgment
The authors are grateful to the National Natural Science Foundation of China (No. 10974138)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, YR., Kuang, XY., Zheng, BB. et al. Ab initio calculation of the geometries, stabilities, and electronic properties for the bimetallic Be2Au n (n = 1–9) clusters: comparison with pure gold clusters. J Mol Model 18, 275–283 (2012). https://doi.org/10.1007/s00894-011-1051-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-011-1051-2