Abstract
In recent years, it has become clear that the neuronal nicotinic acetylcholine receptor (nAChR) is a valid target in the treatment of a variety of diseases, including Alzheimer’s disease, anxiety, and nicotine addiction. As with most membrane proteins, information on the three-dimensional (3D) structure of nAChR is limited to data from electron microscopy, at a resolution that makes the application of structure-based design approaches to develop specific ligands difficult. Based on a high-resolution crystal structure of AChBP, homology models of the extracellular domain of the neuronal rat and human nAChR subtypes α4β2 and α7 (the subtypes most abundant in brain) were built, and their stability assessed with molecular dynamics (MD). All models built showed conformational stability over time, confirming the quality of the starting 3D model. Lipophilicity and electrostatic potential studies performed on the rat and human α4β2 and α7 nicotinic models were compared to AChBP, revealing the importance of the hydrophobic aromatic pocket and the critical role of the α-subunit Trp—the homolog of AChBP-Trp 143—for ligand binding. The models presented provide a valuable framework for the structure-based design of specific α4β2 nAChR subtype ligands aimed at improving therapeutic and diagnostic applications.

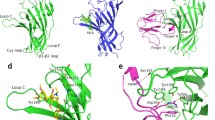
Electrostatic surface potential of the binding site cavity of the neuronal nicotinic acetylcholine receptor (nAChR). Nicotinic models performed with the MOLCAD program: a rat α7, b rat α4β2, c human α7, d human α4β2. All residues labeled are part of the α7 (a,c) or α4 (b,d) subunit with the exception of Phe 117, which belongs to subunit β2 (d). Violet Very negative, blue negative, yellow neutral, red very positive





Similar content being viewed by others
References
Karlin A (2002) Nat Rev Neurosci 3:102–114
Sine SM, Engel AG (2006) Nature 440:448–455
Badio B, Daly JW (1994) Mol Pharmacol 45:563–569
Arneric SP, Holladay M, Williams M (2007) Biochem Pharmacol 74:1092–1101
Itièr V, Bertrand D (2001) FEBS Lett 504:118–125
Hogg R, Bertrand D (2003) Drug News Perspect 16:261–266
Miyazawa A, Fujiyoshi Y, Unwin N (2003) Nature 423:949–955
Unwin NJ (2005) Mol Biol 346:967–989
Brejc K, Van Dijk WJ, Klaassen RV, Schuurmans M, Van der Oost J, Smit AB, Sixma TK (2001) Nature 411:269–276
Sixma TK, Smit AB (2003) Annu Rev Biophys Struct 32:311–334
Celie PH, van Rossum-Fikkert SE, Van Dijk WJ, Brejc K, Smit AB, Sixma TK (2004) Neuron 41:907–914
Celie PH, Kasheverov IE, Mordvintsev DY, Hogg RC, van Nierop P, van Elk R, van Rossum-Fikkert SE, Zhmak MN, Bertrand D, Tselin V, Sixma TK, Smit AB (2005) Nat Struct Mol Biol 12:582–588
Taylor P, Talley TT, Radic Z, Hansen SB, Hibbs RE, Shi J (2007) Biochem Pharmacol 74:1164–1171
Harel M, Kasher R, Nicolas A, Guss JM, Balass M, Fridkin M, Smit AB, Brejc K, Sixma TK, Katchalski-Katzir E, Sussman JL, Fuchs S (2001) Neuron 32:265–275
Hansen SB, Radic Z, Talley TT, Molles BE, Deerinck T, Tsigelny I, Taylor P (2002) J Biol Chem 277:41299–41302
Hansen SB, Radic Z, Talley TT, Taylor P (2003) Faseb J 17:A641–A641
Hansen SB, Talley TT, Radic Z, Taylor P (2004) J Biol Chem 279:24197–24202
Law RJ, Henchman RH, McCammon JA (2005) Proc Natl Acad Sci USA 102:6813–6818
Henchman RH, Wang H-L, Sine SM, Taylor P, McCammon JA (2005) Biophys J 88:2564–2576
Zhang D, Gullingsrud J, McCammon JA (2006) J Am Chem Soc 128:3019–3026
Taly A, Corringer P-J, Grutter T, de Carvalho LP, Karplus M, Changeux JP (2006) Proc Natl Acad Sci USA 103:16965–16970
Szarecka A, Xu Y, Tang P (2007) Proteins 68:948–960
Amiri S, Sansom MSP, Biggin PC (2007) Protein Eng Des Sel 20:353–359
Stoye J (1998) Gene 211:45–56
Guex N, Peitsch MC (1997) Electrophoresis 18:2714–2723
Peitsch MC (1996) Biochem Soc Trans 24:274–279
Peitsch MC (1995) Biotechnology 13:658–660
Le Novère N, Grutter T, Changeux JP (2002) Proc Natl Acad Sci USA 99:3210–3215
Laskowski RA, MacArthur MW, Moss DS, Thornton JM (1993) J Appl Crystallogr 26:283–291
Zhong W, Gallivan JP, Zhang Y, Li L, Lester HA, Dougherty DA (1998) Proc Natl Acad Sci USA 95:12088–12093
Schmitt JD, Sharples CGV, Caldwell WS (1999) J Med Chem 42:3066–3074
Cornell WD, Cieplak P, Bayly CI, Gould IR, Merz KM, Ferguson DM, Spellmeyer DC, Fox T, Caldwell JW, Kollman PA (1995) J Am Chem Soc 117:5179–5197
Berendsen HJC, Postma JPM, van Gunsteren WF, DiNola A, Haak JRJ (1984) Chem Phys 81:3684–3690
Brickmann J, Goetze T, Heiden W, Moeckel G, Reiling S, Vollhardt H, Zachmann C-D (1995) Data visualization in molecular science: tools for insight and innovation. Addison-Wesley, Reading, MA
Lukas RJ (1998) The nicotinic acetylcholine receptor: current views and future trends. Springer, Berlin
Jensen AA, Zlotos DP, Liljefors TJ (2007) J Med Chem 50:4616–4629
Bisson WH, Scapozza L, Westera G, Mu L, Schubiger PA (2005) J Med Chem 48:5123–5130
Huang X, Miller W (1991) Adv Appl Math 12:373–381
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bisson, W.H., Westera, G., Schubiger, P.A. et al. Homology modeling and dynamics of the extracellular domain of rat and human neuronal nicotinic acetylcholine receptor subtypes α4β2 and α7. J Mol Model 14, 891–899 (2008). https://doi.org/10.1007/s00894-008-0340-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-008-0340-x