Abstract
Objectives
Effective disinfection of the root canals is the cornerstone of successful endodontic treatment. Diminishing the microbial load within the root canal system is crucial for healing in endodontically treated teeth. The aim of this study was to evaluate the effect of 2780 nm Er,Cr:YSGG and 940 nm diode lasers on the eradication of microorganisms from single-rooted teeth with asymptomatic apical periodontitis.
Materials and Methods
Thirty participants conforming to the inclusion criteria were randomly divided into 3 groups according to the disinfection protocol used; Conventional group: 2.5% Sodium Hypochlorite (NaOCl) and 17% EDTA solution NaOCl/EDTA, Dual laser group: 2780 nm Erbium, chromium: yttrium scandium-gallium-garnet (Er,Cr:YSGG) laser and 940 nm diode laser Er,CrYSGG/Diode, and Combined group: 17% EDTA and 940 nm diode laser EDTA/Diode. Bacterial samples were collected before and after intervention. The collected data were statistically analyzed using Friedman’s test and Kruskal–Wallis test (P ≤ 0.05).
Results
The results of the study showed that both dual laser Er,CrYSGG/Diode and combined laser EDTA/Diode groups showed significantly less mean Log10 CFU/ml of aerobic and anaerobic bacterial counts than the conventional NaOCl/EDTA group.
Conclusions
In this study we evaluated in vivo the bactericidal efficacy of three disinfection protocols for endodontic treatment of single-rooted teeth with apical periodontitis. The results indicated that both dual laser Er,CrYSGG/Diode and combined laser EDTA/Diode groups provide superior bactericidal effect compared to the conventional NaOCl/EDTA group.
Clinical relevance
The integration of lasers into root canal disinfection protocols has demonstrated significant bacterial reduction which might promote healing and long-term success.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The ultimate goal of endodontic treatment is the creation of a sterile, bacteria-free environment inside the tooth as well as the periapical tissue and the surrounding bone to prevent or treat chronic periapical lesions [1]. Sodium hypochlorite (NaOCl) is the most commonly used irrigating solution in endodontic practice due to its ability to dissolve organic matter as well as its broad antimicrobial action [2]. Ethylenediaminetetraacetic acid (EDTA) is a chelating agent that is employed to remove calcium from dentine leaving a softened matrix. It also removes the smear layer associated with root canal instrumentation. and facilitates the mechanical preparation of the root canal [3]. Combining NaOCl and EDTA can be considered the standard method of endodontic cleaning.
However, conventional endodontic treatment is confronted with many challenges. First, it is limited by the morphological complexity of the root canal system, including lateral canals and accessory foramina especially in the apical area [4]. Second, 5% NaOCl is unable to eliminate bacteria that have penetrated deeper dentin layers, which may lead to recurrent endodontic lesions [5]. Thirdly, NaOCl is irritant to periapical tissues; a concentration of NaOCl over 0.5% is cytotoxic [6, 7]. Hence, prompting the need for new approaches to enhance endodontic disinfection.
Lately, laser therapy for root canal disinfection has gained attention. The goals for a laser-assisted root canal treatment are optimum removal of smear layer, deep penetration of laser energy into dentin, and intricate root canal structures to destroy microorganisms that can reach > 1000 µm inside dentin, as well as effective healing of damaged tissues [8, 9].
Er,Cr:YSGG laser belongs to the infrared Erbium family. It interacts with aqueous solutions and at a power of 3.5 W or more ablates efficiently dental hard tissue. Er,Cr:YSGG is used in endodontic treatment to remove the smear layer from the root canal walls because it exerts a limited penetration depth of 17 µm into the dentin [10]. Its bactericidal property depends on either instant evaporation of intracellular water or bacterial dehydration [11, 12]. In comparison with irrigation with EDTA, Er,Cr:YSGG laser has been consistently providing superior smear layer removal effectiveness [13].
940-nm diode laser is one of the near-infrared lasers that are highly absorbed by melanin and hemoglobin, and little absorbed by hydroxyapatite crystals or water. This property allows diode lasers to penetrate deep into dentine and exhibit its anti-bacterial effect [14, 15]. 940 nm Diode laser is used in endodontic treatment for disinfection of root canals. Diode laser has an intense antibacterial effect by causing changes in the bacterial cell wall and destroying the cell membrane1. Diode laser exerts a photo-thermal effect on the reachable bacteria. It also exerts a photo-disruptive effect on the unreachable bacteria; where immediate cell death might not occur; but rather sublethal damage occurs inhibiting the cell growth through the destruction of cell wall integrity and accumulation of denatured proteins causing the cessation of bacterial growth and consecutive cell lysis. This effect on bacteria occurs with very small doses of heat [16].
Diode laser uses a thin flexible 200 µm optical fiber to deliver its beam to the target area. The optical fiber can easily reach the apical third of the root canal, curved canals, and different anatomical areas that are difficult to access; eventually distributing the light consistently inside the root canal assuring a better photoreaction [1]. An additional advantage of diode laser is its extreme compactness, affordability, and ease of operation.
Well-reported in vitro studies have evaluated the bactericidal effect of Er,Cr:YSGG and diode laser [16,17,18,19,20]. One of these studies investigated the bactericidal potential of 2780-nm ER,CR:YSGG and 940-nm diode lasers. The findings revealed that the combined use of these lasers was more effective than either laser alone and is comparable to needle irrigation with sodium hypochlorite and EDTA [20].
An in vivo study assessed the results of laser-assisted endodontic treatment/ retreatment using 940-nm diode laser and EDTA in teeth with apical periodontitis. They concluded that 940-nm diode laser-assisted endodontic protocol is an effective replacement for conventional treatment that promotes faster healing of periapical lesions, requiring less chemical irrigation and systemic antibiotics [21].
further in vivo research used 2780 and 940 nm wavelengths for smear layer removal and achieved deep dentin disinfection respectively on two cases of relatively high endodontic complexity. Their 1-year follow-up supported the effectiveness of this treatment strategy [22].
Another study examined the safety of the combination of Er,Cr:YSGG and diode laser. They reported no unfavorable thermal variations on the external root surface. Thus, the aim of this study was to assess in vivo the impact of Er,Cr:YSGG/diode laser, and Diode/EDTA on the bacterial count in root canal treatment compared to conventional endodontic treatment in an evidence-based clinical trial. The null hypothesis being tested is that there will be no difference in total bacterial count reduction between the three groups [23].
Methods
This study is a double-blinded, three-arm, randomized, clinical trial that was designed, reported, and written according to Preferred Reporting Items for Randomized Trials in Endodontics (PRIRATE) 2020 guidelines (Nagendrababu et al., 2020). The protocol was approved by the Faculty of Dentistry, Ain Shams University research ethics committee (approval number FDASU-REC ID041908). The trial design was registered at www.clinicaltrials.gov database with identifier number(NCT05964686).(28/07/2023).
https://clinicaltrials.gov/study/NCT05964686?term=NCT05964686&rank=1
Sample size calculation
The sample size was calculated according to the results of the study by Wenzler et al.[24]. The standard deviation within groups was assumed to be 15%. The effect size (f) was 0.73 Using alpha (α) level of (5%) and Beta (β) level of (20%) i.e., power = 80%; the minimum estimated sample size was a total of 24 subjects (8 subjects per group). The sample size was increased to 10 subjects per group to compensate for a drop-out rate of 20%. Sample size calculation was performed using G*Power Version 3.1.9.2.
Randomization
Thirty participants were randomly allocated using www.randomizer.org to 3 groups (n = 10). Randomization for this study was conducted on the Randomizer website on January 9th, 2022, at 4:00 PM. Patients referred to the outpatient endodontic clinic at the Faculty of Dentistry, Ain Shams University, between January and September 2022, were assessed for eligibility and subsequently assigned to groups in accordance with the pre-established randomization scheme.
The inclusion criteria were as follows
Patients between 18 and 35 years old were included in this study if they had one single-rooted maxillary anterior tooth with necrotic pulp, closed apex, and asymptomatic apical periodontitis requiring root canal treatment. For inclusion in this study, the periapical lesion should have a periapical index score of 3 or 4 Ørstavik, et al.[25] The diagnosis of non-vital pulp was based on history-taking, clinical and radiographic examination.
The exclusion criteria were the presence of pain, swelling, a fistulous tract, or the presence of periodontal pockets more than 3 mm in the involved tooth. Patients who had received antibiotics during the last month, had previous root canal treatment in the related tooth, had any systematic disease, or had an allergy to NSAIDs were also excluded. The vulnerable groups, including pregnant females and mentally or physically disabled individuals, were not included. Patients who refused to participate in the study and those who were presented with technical difficulties during root canal treatment, including curved roots or complications during treatment such as broken files, were also excluded. The research goal and procedures were clearly explained to each patient before they signed the informed consent.
Patients were randomly distributed into 3 groups. Each participant was given a number from (1 to 30) using randomization software (www.randomizer.org). The root canal treatments were performed in a single visit by one investigator.
Antisepsis of the oral cavity was performed by rinsing for 1 min with 10 mL of 0.125% chlorhexidine gluconate mouthwash. Lidocaine topical anesthetic gel was used at the site of the injection. The tooth was anesthetized by buccal infiltration using (1:80,000 Arcaine, Aarge Pvt, India) local anesthetic solution using a side-loading cartridge aspirating syringe and a 30-gauge short needle.
Single-tooth isolation was performed using a suitable clamp and rubber dam. An antiseptic solution was used to disinfect the isolated tooth and surrounding rubber dam. All caries and/or coronal restorations were completely removed with sterile bur without exposure of the pulp chamber. The access cavity was prepared using sterile round carbide bur size #4. The patency of the canals was done using a stainless-steel hand K-file size 15. The root canal was irrigated with 1 ml of sterile saline solution. The root canals were scraped with H-file and irrigated with 1 ml of sterile saline solution. The first microbial samples (S1) were collected, the sample collection was adapted from the protocol previously described by Gomes et al. [26] to assess the initial colonizers of the root canals. Three sterile paper points were inserted into the root canal for 1 min each with pumping movements. Care was taken to avoid contact between the paper points and access cavity walls to prevent contamination. They were immediately inserted into sterile tubes containing a transport medium of thioglycolate and transferred to the microbiology lab.
Working length was measured using an apex locator (Root ZX mini, J. Morita, Japan) and then confirmed with an intraoral periapical radiograph to be 0.5–1 mm shorter than the radiographic apex. Root canals were mechanically prepared using ProTaper Next nickel-titanium rotary instruments (Dentsply Maillefer, Ballaigues, Switzerland) according to the manufacturer’s instructions until the X4 (40/06) master apical file. The patients were divided into 3 groups according to the disinfection protocol, as follows:
Conventional group (NaOCl/EDTA): 5 ml of 2.5% sodium hypochlorite for 1 min was used for irrigation between each file and the next using a 30-gauge side-vented needle reaching 2 mm short of the working length, then 5 ml of saline, followed by 1 ml of 5% sodium thiosulfate to neutralize the effect of NaOCl and finally 5 ml of 17% EDTA (Denteck, Zoetermeer, Netherlands) for 1 min was used at the end of the procedure to remove the smear layer. Thus, a total of 20 ml of NaOCl and a total of 5 ml of EDTA were used in the NaOCl-EDTA group.
Dual laser group (Er,Cr:YSGG/Diode): Er,Cr:YSGG intracanal laser irradiation was performed (ʎ = 2780 nm, 1.25 W source power, 20 Hz, 10% Air, 1% water) using a radial emitting tip RFT2 200 µm in diameter. 5 ml of 0.9% sterile saline was used for irrigation between laser applications. The irradiation protocol adopted for this study was 10 s irradiations followed by 10 s intervals, which constituted one lasing cycle. Each lasing cycle was performed 4 times for each root canal using the Er,Cr:YSGG laser to remove the smear layer.
That was followed by diode laser (Epic X™, BIOLASE Tech, Irvine, USA) for the activation process, diode laser (ʎ = 940 nm, 1W, source power) was utilized using the E2-14 tip, an endo 200 µm flexible laser tip with a 14 mm length, 1 mm short of the working length, in continuous mode, using the same irradiation protocol as previously mentioned [27].
Combined group (EDTA/Diode): 5 ml of 17% EDTA was used to remove the inorganic part of the smear layer. 5 ml of 0.9% sterile saline was used for irrigation. Disinfection of the root canal was done using the same diode laser machine and protocol.
Following the disinfection protocol, in all groups, sterile saline was placed in the canal, and then the canal was dried with 3 sterile ProTaper Next absorbent paper points corresponding to the same size as the master file. Each paper point was left for 1 min, and then the second microbial sample (S2) was collected to assess the colonization of bacteria and the status of the root canal just before obturation.
The root canals were obturated using the EQ-V system (Meta Biomed Korea) continuous wave condensation technique with proper selection of gutta-percha master cone corresponding to the same size as the master apical file and ADSEAL (Meta Biomed Korea) resin root canal sealer. Postoperative radiographs were taken to ensure proper obturation. The access cavity was sealed using a resin-modified glass ionomer. Some of the steps of the research are presented in Fig. 1.
Sterile tube containing Thioglycolate, (B) Paper point is inserted inside the root canal, (C) The paper point is then inserted inside the sterile tube, (D) laser is applied inside root canal, (E) Preoperative periapical radiograph is taken, (F) Working length is confirmed with k-file #15, (G) Periapical radiograph is taken with the master apical cone in place, (H) Postoperative periapical radiograph is acquired
Microbiological analysis
Bacterial count
Once the samples arrived at the microbiology lab, the tubes containing the thioglycolate (transport medium) (Thioglycollate broth U.S.P alternative, Oxoid microbiology product, England) with the paper points were placed in a microcentrifuge and vortexed for 30 s. One hundred µl aliquots of the vortexed samples were placed in a new sterile tube containing 1 ml of thioglycolate to obtain 1/10, 1/100, and 1/1000 concentrations to assess the microbial load of common aerobes and anaerobes found in each root canal.
Aerobic bacterial culture
Fifty µl of these diluted samples were transferred to Brain Heart Infusion BHI (Oxoid microbiology product, England) agar plates and cultured under aseptic conditions, followed by incubation at 37 °C for 24 h for the aerobic bacteria. The number of bacterial colonies in each plate was counted and reported as colony-forming units per milliliter (CFU/ml).
Anaerobic bacterial culture
The other 50 µl of these diluted samples were transferred to BHI agar plates under aseptic conditions. The agar plates were placed in an anaerobic sealed jar with Gas-Pak (Gas-Pak system) (Oxoid microbiology product, Basingstoke, Hants, England), and anaerobic indicator (Anaerobic indicator, BR0055B.Oxoid. Basingstoke, Hants, England) were incubated for 48 h at 37 °C. The number of bacterial colonies in each plate was eventually counted in each plate of the most diluted sample and reported as CFU/ml. Visible colonies were enumerated in each petri dish, and the number of colonies/plate was multiplied by the corresponding dilution factor and by 10 to determine the total CFUs/ mL of each specimen.
Statistical Analysis
Numerical data were explored for normality by checking the distribution of data and using tests of normality (Kolmogorov–Smirnov and Shapiro–Wilk tests). Percentage reduction in bacterial counts data showed non-normal distribution. Data were presented as median, range, mean and standard deviation (SD) values. Friedman’s test was used to assess the changes in bacterial counts within each group after disinfection. The Kruskal–Wallis test was used to compare between the three groups. Dunn’s test was used for pair-wise comparisons when Kruskal–Wallis test or Friedman’s test is significant. The significance level was set at P ≤0.05. Statistical analysis was performed with IBM SPSS Statistics for Windows, Version 23.0. Armonk, NY: IBM Corp.
Results
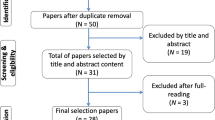
A total of 56 patients were assessed for eligibility, 26 were excluded. The details are presented in a PRIRATE 2020 flow chart in Fig. 2. There were no dropouts in this study because the endodontic treatment was completed in one visit.
Percentage reduction in bacterial counts
Percentage reduction was calculated as follows: [(Counts before application – After application) / Before application × 100]. There was a statistically significant difference between the three groups regarding the percentage reduction in aerobic and anaerobic bacterial counts. Pair-wise comparisons between the groups revealed no significant difference between Er,Cr;YSGG/Diode and EDTA/Diode groups; both showed significantly higher percentage reductions in aerobic and anaerobic bacterial counts than the NaOCl/EDTA group.
Overall percentage reduction in bacterial counts
A statistically significant difference was found between the three groups regarding the overall percentage reduction in bacterial counts. Pair-wise comparisons between the groups revealed that there was no significant difference between Er,Cr;YSGG/Diode, and EDTA/Diode groups; both showed higher percentage reduction in bacterial counts than NaOCl/EDTA group. The percentage reduction in aerobic, anaerobic bacterial counts and overall bacterial counts in the three groups are represented in Table 1, Fig. 3.
Discussion
The trial design of this prospective study was a double-blinded randomized clinical design since both the patients and the medical microbiologist who measured the bacterial count were blinded. The goal of randomization was to produce comparable groups in terms of general participants’ characteristics. Thus, eliminating any bias that may affect the relation between the interventions and the outcomes. The target population for this study included individuals between 18 and 35 years old. This age range was chosen due to the relative consistency of maxillary anterior teeth canal diameter and anatomy exhibited within this group. To further enhance sample homogeneity, patients with specific anatomical variations, such as open apices, were excluded from participation.
Patients under antibiotic therapy in the last month were excluded as this may reduce bacterial counts, inhibit and suppress bacterial growth which directly affect the colony count and impair the microbiological results, additionally antibiotic therapy cause disruption of the microbial flora acquiring a higher risk of developing postoperative infection and selecting for antibiotic resistant bacteria [28].
In the present study, dual laser group and combined laser groups resulted in less aerobic and anaerobic bacterial counts. Thus, the null hypothesis was rejected.
The success of endodontic treatment depends ultimately on the elimination of the microorganism, host response, and coronal seal of root canals [22]. Achieving long-term successful endodontic treatment without toxic chemical irrigants has always been a long-awaited goal of scientists. Hence the need for more sophisticated techniques to enhance disinfection like automated irrigation devices, ultrasonics [29], and lasers [30]. Combining NaOCl and EDTA represents the control group in this research because it is the standard method of endodontic disinfection. The Er,Cr;YSGG/Diode group was chosen to combine the intense antibacterial effect of diode laser together with the smear layer removal ability of Er,Cr:YSGG laser. The EDTA/Diode group represents a more economic merger, where the affordable diode laser with its antimicrobial effect is combined with 17% EDTA as a cost-effective chelating agent to remove the smear layer with minimal demineralizing effect [31] instead of the expensive Er,Cr:YSGG laser.
Root canal treatments for the three groups were completed in a single visit. According to the systematic review by Manfredi et al. [32] there is no evidence to suggest that single-visit or multiple-visit root canal treatment is better than the other. On the other hand, there is a tendency for clinicians to perform single-visit endodontic treatment recently owing to the benefits such as not requiring additional anesthetic injections, no need to replace the rubber dam or intracanal medication, absence of inter-visit leakage, loss of temporary seal, or any of the accidents that can and do occur between the visits [33]. Similarly, Özcan et al.[34] concluded that single-visit root canal treatment may be a strong alternative to multi-visit treatment.
Mechanical preparation was done using the ProTaper Next rotary system and a standardized irrigation protocol for the standardization of the procedure. The (NiTi) rotary instruments were chosen because they result in minimal debris extrusion compared to the stainless-steel hand K-files due to their design and motion [35, 36].
Vertical compaction techniques and ADSEAL were applied for the three groups since the obturation material mass becomes more homogeneous and adapts better to root canal irregularities compared to cold lateral compaction (CLC). Moreover, CLC is time-consuming, causes voids in the obturation mass, and increases the risk of vertical root fracture [37, 38].
In this study, 2780 nm Er,Cr:YSGG was used at a power of 1.25W. Er,Cr:YSGG has been used to clean the root canal system at output powers ranging from 1 to 3W [39, 40]. However, increasing the power above 1.5 W might potentially cause thermal damage. Moreover, high power Er,Cr:YSGG laser has limited usefulness in removing the smear layer because the structural damage that it causes in the dentinal structure in itself might become a source of smear layer formation [40, 41].
Diode Laser at a power of 1W in a continuous mode was used in this study. The continuous mode ensures equal distribution of light along the surface of the root canal and enhances the regeneration of periapical tissue. Continuous mode at a power higher than 1.5W is not advisable in the root canal because of the heat damage that it may cause [1, 9].
Following the methodology of Godbole et al.[42] in vitro study, the root canals were irradiated from the apical to the coronal portion, in a helicoidal movement touching the canal walls. This standardized protocol ensures uniform diffusion of laser light inside the root lumen and reduces heating of dentin, thereby avoiding damage to the surrounding periodontal tissues. This protocol improves the reduction of microbial load as shown also by Garcez et al.[43].
The total time of radiation was 40 s. It was divided into four times of 10 s irradiation and three times of 10 s pause between the lasing cycle. Each irradiation had a rest period of 10 s to avoid the temperature raise above the 7 °C threshold of periodontal tissues. Studies [15, 44] evaluating the rise in temperature on the external root surface during intracanal irradiation using Diode lasers have recorded temperature rise in the range of 1–7 °C. Since the threshold bone necrosis temperature for periapical structures is 47 °C for 1 min [45] this temperature rise is well below the danger threshold. Moreover, Al-Karadaghi et al. [23] evaluated in vitro the effects of dual wavelength (2780 nm Er,Cr:YSGG and 940 nm diode) laser on temperature changes during laser-assisted root canal treatment and concluded that within the parameters studied, the dual-wavelength laser did not lead to unfavorable thermal changes on the external root surface.
In this study, the bacterial count method was used to measure the viable cells in CFU/mL- × 10*(colony-forming units per milliliter). Some studies [46, 47] have shown that the culturing method demands a microbiological facility in close proximity to the dental office to ensure that the bacteria do not die in transit. To avoid this problem, root canal samples were cultured within four hours in the microbiology department after sample collection. Many advanced techniques have been proposed to evaluate the bacterial load of root canals, including flow cytometry, PCR, and others. Esterela et al. [48] compared the results obtained from PCR and culture techniques; the same results were obtained from both techniques despite the higher sensitivity of the PCR technique.
In the current study, we assessed aerobic and anaerobic bacteria. As the periapical infection is a polymicrobial disease, the aerobic culturing technique alone is not sufficient to reflect the microbiologic status of the root canal system [1]. Thus, the total aerobic and anaerobic count of bacteria were counted following the same methodology of Garcez et al. [47].
Several techniques have been described to obtain a representative sample of root canal bacteria. Orstavik et al.[49, 50] used reamers. However, in most of the vivo studies [1, 50, 51] root canal samples were acquired with paper points, as in this study.
In the conventional group, 5 ml of 2.5% sodium hypochlorite for 1 min together with 5 ml of 17% EDTA for 1 min was used in group 1 for disinfection and has resulted in 57.6% mean overall bacterial reduction. Similarly, Abbaszadegan et al. [52] Laukkanen et al. [53] and Rubio et al. [54] concluded that conventional endodontic treatments have proved to be insufficient to lower the detection limits of endodontic pathogenic microorganisms, with success percentage not exceeding 62% to 83%.
Superficial bacteria have shown a total bacterial reduction of 57.6% in the conventional group. However, an inferior rate of bacterial reduction is expected in the deeper layers of dentin because microorganisms can invade dentin up to a depth of 1100 µm [55]. While a 6% solution of NaOCl for 20 min at 45°C cannot penetrate the dentinal tubules more than 300 µm [56].
In the dual laser Er,CrYSGG/Diode group, a total bacterial reduction of 99.97% for the aerobic bacteria and 99.97% for the anaerobic bacteria was found in the S2 samples. Similarly, Aksoy et al. [57] evaluated the smear layer removal efficiency of Er,Cr;YSGG in vitro. They concluded that Er,Cr:YSGG laser application is more efficient than NaOCl irrigation in removing the smear layer. Montero et al. [58] also compared EDTA and Er,Cr:YSGG laser in vitro regarding debris and smear layer removal. They reported that the laser showed greater cleaning efficacy than EDTA and concluded that combining the laser with EDTA improved the cleanliness even better, as the effect was accumulative.
Moreover, Erben et al. [20] evaluated in vitro the bactericidal potential of 2780-nm Er,Cr:YSGG and 940-nm diode lasers. They concluded that the wavelength combination of Er,Cr:YSGG and 940-nm diode laser is safe and highly effective than either laser alone and is comparable to needle irrigation with sodium hypochlorite and EDTA.
Merigo et al. [59] evaluated the bactericidal effect of Er,Cr:YSGG laser irradiation alone and when associated with NaOCl irrigation on endodontic biofilm in freshly extracted single-rooted human teeth. They concluded that Er,Cr:YSGG laser has better decontamination capacity when used in conjunction with NaOCl irrigation. However, the present study demonstrates that optimum root canal disinfection could be achieved without NaOCl.
The combined (Diode/EDTA) group S2 sample resulted in a cumulative total bacterial reduction of 99.93% for aerobic bacteria and 99.94% for anaerobic bacteria when compared to the initial S1 sample. Similarly, Beer et al. [60] reported an average bacterial reduction of 98.66% in vitro using 940-nm diode laser and saline. Moreover, Morsy et al. [1] found that intracanal irradiation in vivo with Diode laser together with the application of NaOCl and EDTA resulted in a bacterial reduction of 99.73% for aerobic and 99.9% for anaerobic bacteria.
Masilionyte et al. [21] compared the outcomes of laser-assisted endodontic treatment/ retreatment using a 940-nm diode laser and EDTA versus the conventional protocol of NaOCl and EDTA in teeth with apical periodontitis. They concluded that the 940-nm diode laser-assisted endodontic protocol is an effective replacement to conventional treatment. The advantages of the Diode/EDTA protocol included decreased consumption of chemical irrigants and systemic antibiotics as well as promoting faster healing of periapical lesions.
In the present study, the conventional NaOCl/EDTA group resulted in a total bacterial reduction of 63.57% for aerobic bacteria and 58.79% for anaerobic bacteria, while the combined Diode/EDTA group S2 sample resulted in a total bacterial reduction of 99.93% for aerobic bacteria and 99.94% for anaerobic bacteria when compared to the initial S1 sample. Opposingly, Sohrabi et al. [17] found that the NaOCl eradicated 99.87% of the bacteria and showed a significantly enhanced antibacterial impact compared to the 980-nm diode laser, which resulted in 96.56% bacterial eradication. This difference from our results may be due to the fact that they tested the pure antibacterial effect of Diode laser not in conjunction with Er,Cr:YSGG laser or EDTA that aids in the removal of the smear layer. The promising results observed using laser-assisted disinfection warrant further investigation against ultrasonic activated irrigation [29] to establish their relative efficacy and identify potential niche applications for each [30]. Future studies are also warranted to evaluate the efficacy of Er,CrYSGG/Diode and EDTA/Diode in multirooted teeth with smaller clinical anatomical diameters. This will address potential limitations identified in the present investigation and broaden the generalizability of the findings.
Conclusions
In this study we evaluated in vivo the bactericidal efficacy of three disinfection protocols for endodontic treatment of single-rooted teeth with apical periodontitis. The results indicated that both dual laser Er,CrYSGG/Diode and combined laser EDTA/Diode groups provide superior bactericidal effect compared to the conventional NaOCl/EDTA group.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Morsy DA, Negm M, Diab A, Ahmed G (2018) Postoperative pain and antibacterial effect of 980 nm diode laser versus conventional endodontic treatment in necrotic teeth with chronic periapical lesions: a randomized control trial. F1000Res 7:1795–1809. https://doi.org/10.12688/f1000research.16794.1
Abuhaimed TS, Abou Neel EA (2017) Sodium hypochlorite irrigation and its effect on bond strength to dentin. Biomed Res Int 2017:1930360. https://doi.org/10.1155/2017/1930360
Iqbal A (2012) Antimicrobial irrigants in the endodontic therapy. Int J Health Sci 6:186–192. https://doi.org/10.12816/0005998
Dioguardi M, Di Gioia G, Illuzzi G, Laneve E, Cocco A, Troiano G (2018) Endodontic irrigants: Different methods to improve efficacy and related problems. Eur J Dent 12:459–466. https://doi.org/10.4103/ejd.ejd_56_18
Kaiwar A, Usha HL, Meena N, Ashwini P, Murthy CS (2013) The efficiency of root canal disinfection using a diode laser: in vitro study. Indian J Dent Res 24:14–18. https://doi.org/10.4103/0970-9290.114916
Chaugule VB, Panse AM, Gawali PN (2015) Adverse reaction of sodium hypochlorite during endodontic treatment of primary teeth. Int J Clin Pediatr Dent 8:153–156. https://doi.org/10.5005/jp-journals-10005-1304
Hülsmann M, Hahn W (2000) Complications during root canal irrigation–literature review and case reports. Int Endod J 33:186–193. https://doi.org/10.1046/j.1365-2591.2000.00303.x
Bytyqi A, Aliu X, Barani M, Stubljar D, Jukic T, Starc A, Krasniqi S (2021) Disinfection of infected artificial dental periapical lesions with diode laser: an in vitro study. Med Sci Monit Basic Res 27:e932492–1–e932492–7. https://doi.org/10.12659/MSMBR.932492
Dragana R, Jelena M, Jovan M, Biljana N, Dejan M (2022) Antibacterial efficiency of adjuvant photodynamic therapy and high-power diode laser in the treatment of young permanent teeth with chronic periapical periodontitis. A prospective clinical study. Photodiagnosis Photodyn Ther 41:103129. https://doi.org/10.1016/j.pdpdt.2022.103129
Diaci J, Gaspirc B (2012) Comparison of Er: YAG and Er, Cr: YSGG lasers used in dentistry. J Laser Health Acad 1:1–13
Blanken J, De Moor RJG, Meire M, Verdaasdonk R (2009) Laser induced explosive vapor and cavitation resulting in effective irrigation of the root canal. Part 1: a visualization study. Lasers in Surgery and Medicine: The Official Journal of the American Society for Laser Medicine and Surgery 41:514–519. https://doi.org/10.1002/lsm.20798
López-Jiménez L, Arnabat-Domínguez J, Viñas M, Vinuesa T (2015) Atomic force microscopy visualization of injuries in Enterococcus faecalis surface caused by Er, Cr: YSGG and diode lasers. Med Oral Patol Oral Cir Bucal 20:e45–e51
George R, Walsh LJ (2010) Thermal effects from modified endodontic laser tips used in the apical third of root canals with erbium-doped yttrium aluminium garnet and erbium, chromium–doped yttrium scandium gallium garnet lasers. Photomed Laser Surg 28:161–165. https://doi.org/10.1089/pho.2008.2423
Galui S, Pal S, Mahata S, Saha S, Sarkar S (2019) Laser and its use in pediatric dentistry: a review of literature and a recent update. International Journal of Pedodontic Rehabilitation 4:1–5. https://doi.org/10.4103/ijpr.ijpr_17_18
Gutknecht N (2008) Lasers in endodontics. J Laser Health Acad 4:1–5
Preethee T, Kandaswamy D, Arathi G, Hannah R (2012) Bactericidal effect of the 908 nm diode laser on Enterococcus faecalis in infected root canals. J Conserv Dent 15:46–50. https://doi.org/10.4103/0972-0707.92606
Sohrabi K, Sooratgar A, Zolfagharnasab K, Kharazifard MJ, Afkhami F (2016) Antibacterial activity of diode laser and sodium hypochlorite in enterococcus faecalis-contaminated root canals. Iran Endod J 11:8–12. https://doi.org/10.7508/iej.2016.01.002
Asnaashari M, Ebad LT, Shojaeian S (2016) Comparison of antibacterial effects of 810 and 980-nanometer diode lasers on enterococcus faecalis in the root canal system-an in vitro study. Laser Ther 25:209–214. https://doi.org/10.5978/islsm.16-OR-17
Cretella G, Lajolo C, Castagnola R, Somma F, Inchingolo M, Marigo L (2017) The effect of diode laser on planktonic Enterococcus faecalis in infected root canals in an ex vivo model. Photomed Laser Surg 35:190–194. https://doi.org/10.1089/pho.2016.4174
Erben P, Chang AM, Darveau RP, Fong H, Johnson JD, Paranjpe A (2019) Evaluation of the bactericidal potential of 2780-nm ER, CR: YSGG and 940-nm diode lasers in the root canal system. Lasers Dent Sci 3:137–146. https://doi.org/10.1007/s41547-019-00060-z
Masilionyte M, Gutknecht N (2018) Outcome of 940-nm diode laser-assisted endodontic treatment of teeth with apical periodontitis: a retrospective study of clinical cases. Lasers Dent Sci 2:169–179. https://doi.org/10.1007/s41547-018-0035-2
Martins MR, Franzen R, Depraet F, Gutknecht N (2018) Rationale for using a double-wavelength (940 nm+ 2780 nm) laser in endodontics: literature overview and proof-of-concept. Lasers Dent Sci 2:29–41. https://doi.org/10.1007/s41547-017-0017-9
Al-Karadaghi TS, Gutknecht N, Jawad HA, Vanweersch L, Franzen R (2015) Evaluation of temperature elevation during root canal treatment with dual wavelength laser: 2780 nm Er, Cr: YSGG and 940 nm diode. Photomed Laser Surg 33:460–466. https://doi.org/10.1089/pho.2015.3907
Wenzler JS, Falk W, Frankenberger R, Braun A (2021) Impact of Adjunctive Laser Irradiation on the Bacterial Load of Dental Root Canals: A Randomized Controlled Clinical Trial. Antibiotics 10:1557. https://doi.org/10.3390/antibiotics10121557
Ørstavik D, Kerekes K, Eriksen HM (1986) The periapical index: a scoring system for radiographic assessment of apical periodontitis. Dent Traumatol 2:20–34
Gomes BPFA, Pinheiro ET, Gadê-Neto CR et al (2004) Microbiological examination of infected dental root canals. Oral Microbiol Immunol 19:71–76
Hendi SS, Shiri M, Poormoradi B, Alikhani MY, Afshar S, Farmani A (2021) Antibacterial Effects of a 940 nm Diode Laser With/Without Silver Nanoparticles Against Enterococcus faecalis. J Lasers Med Sci 12:e73–e80
Segura-Egea JJ, Gould K, Şen BH et al (2017) Antibiotics in Endodontics: a review. Int Endod J 50:1169–1184. https://doi.org/10.1111/iej.12741
Rajamanickam K, Teja KV, Ramesh S et al (2023) Evaluation of Root Canal Cleanliness on Using a Novel Irrigation Device with an Ultrasonic Activation Technique: An Ex Vivo Study. Applied Sciences (Switzerland) 13:796–807. https://doi.org/10.3390/app13020796
Badami V, Akarapu S, Kethineni H et al (2023) Efficacy of Laser-Activated Irrigation Versus Ultrasonic-Activated Irrigation: A Systematic Review. Cureus 15:e36352–e36361. https://doi.org/10.7759/cureus.36352
Gandolfi MG, Taddei P, Pondrelli A et al (2019) Demineralization, collagen modification and remineralization degree of human dentin after EDTA and citric acid treatments. Materials 12:25–44. https://doi.org/10.3390/ma12010025
Manfredi M, Figini L, Gagliani M, Lodi G (2016) Single versus multiple visits for endodontic treatment of permanent teeth. Cochrane Database of Systematic Reviews 12:CD005296. https://doi.org/10.1002/14651858.CD005296.pub3
ÇAKICI E, GÜNAYDIN A, Uysal B, ÇAKICI F (2020) Effect of low level laser therapy on postoperative pain after single visit endodontic treatment: a placebo-controlled randomized clinical trial. Middle Black Sea Journal of Health Science 6:166–171. https://doi.org/10.19127/mbsjohs.703749
Özcan HÖ, Haznedaroğlu F, Gökyay S (2021) Comparison of the incidence of post-operative pain after low-level laser therapy between single-and multi-visit root canal treatments for chronic apical periodontitis: A prospective randomized clinical trial. International Dental Research 11:30–37. https://doi.org/10.5577/intdentres.2021.vol11.no1.6
Nekoofar MH et al (2015) Comparison of the effect of root canal preparation by using Wave One and ProTaper on postoperative pain: a randomized clinical trial. J Endod 41:575–578. https://doi.org/10.1016/j.joen.2014.12.026
Oliveira PS et al (2022) Postoperative Pain Following Root Canal Instrumentation Using ProTaper Next or Reciproc in Asymptomatic Molars: A Randomized Controlled Single-Blind Clinical Trial. Clin Med 11:3816. https://doi.org/10.3390/jcm11133816
Demirci GK, Çalışkan MK (2016) A prospective randomized comparative study of cold lateral condensation versus core/gutta-percha in teeth with periapical lesions. J Endod 42:206–210. https://doi.org/10.1016/j.joen.2015.10.022
Sabadin N, Böttcher DE, Hoppe CB et al (2014) Resin-based sealer penetration into dentinal tubules after the use of 2% chlorhexidine gel and 17% EDTA: in vitro study. Braz J Oral Sci 13:308–313. https://doi.org/10.1590/1677-3225v13n4a13
Peeters HH, Suardita K (2011) Efficacy of smear layer removal at the root tip by using ethylenediaminetetraacetic acid and erbium, chromium: yttrium, scandium, gallium garnet laser. J Endod 37:1585–1589. https://doi.org/10.1016/j.joen.2011.08.022
Bolhari B, Ehsani S, Etemadi A et al (2014) Efficacy of Er, Cr: YSGG laser in removing smear layer and debris with two different output powers. Photomed Laser Surg 32:527–532. https://doi.org/10.1089/pho.2014.3766
Altundasar E, Özçelik B, Cehreli ZC, Matsumoto K (2006) Ultramorphological and histochemical changes after ER, CR: YSGG laser irradiation and two different irrigation regimes. J Endod 32:465–468. https://doi.org/10.1016/j.joen.2005.08.005
Godbole NJ, Dodwad PK, Bhat K, Patil A (2017) Determination of minimum instrumentation size for penetration of diode laser against Enterococcus faecalis in root canals using scanning electron microscope: An in vitro study’. Indian Journal of Health Sciences and Biomedical Research (KLEU) 10:138–144. https://doi.org/10.4103/kleuhsj.ijhs_429_16
Garcez AS, Núñez SC, Hamblin MR, Ribeiro MS (2008) Antimicrobial comparison on effectiveness of endodontic therapy and endodontic therapy combined with photo-disinfection on patients with periapical lesion: a 6 month follow-up. Mechanisms for Low-Light Therapy III 6846:86–92. https://doi.org/10.1117/12.763705
Hmud R, Kahler WA, Walsh LJ (2010) Temperature changes accompanying near infrared diode laser endodontic treatment of wet canals. J Endod 36:908–911. https://doi.org/10.1016/j.joen.2010.01.007
Eriksson AR, Albrektsson T (1983) Temperature threshold levels for heat-induced bone tissue injury: a vital-microscopic study in the rabbit. J Prosthet Dent 50:101–107. https://doi.org/10.1016/0022-3913(83)90174-9
Pirnat S, Lukac M, Ihan A (2011) Study of the direct bactericidal effect of Nd: YAG and diode laser parameters used in endodontics on pigmented and nonpigmented bacteria. Lasers Med Sci 26:755–761. https://doi.org/10.1007/s10103-010-0808-7
Garcez AS, Nuñez SC, Hamblin MR, Ribeiro MS (2008) Antimicrobial effects of photodynamic therapy on patients with necrotic pulps and periapical lesion. J Endod 34:138–142. https://doi.org/10.1016/j.joen.2007.10.020
Estrela C, Silva JA, de Alencar AHG et al (2008) Efficacy of sodium hypochlorite and chlorhexidine against Enterococcus faecalis: a systematic review. J Appl Oral Sci 16:364–368. https://doi.org/10.1590/s1678-77572008000600002
Ørstavik D, Kerekes K, Molven O (1991) Effects of extensive apical reaming and calcium hydroxide dressing on bacterial infection during treatment of apical periodontitis: a pilot study. Int Endod J 24:1–7. https://doi.org/10.1111/j.1365-2591.1991.tb00863.x
Kaplan T, Sezgin GP, Kaplan SS (2020) Effect of Root Canal Disinfection with 980 µm Diode Laser on Post-operative Pain after Endodontic Treatment in Teeth with Apical Periodontitis: A Randomized Clinical Trial. BMC Oral Health 21:41–49. https://doi.org/10.1186/s12903-021-01401-w
Stănuşi AŞ et al (2020) Endodontic bacterial reduction using Diode LASER radiation–short literature review. Romanian Journal of Oral Rehabilitation 12:196–207
Abbaszadegan A, Khayat A, Motamedifar M (2010) Comparison of antimicrobial efficacy of IKI and NaOCl irrigants in infected root canals: An in vivo study. Iran Endod J 5:101–106
Laukkanen E, Vehkalahti MM, Kotiranta AK (2019) Impact of type of tooth on outcome of non-surgical root canal treatment. Clin Oral Investig 23:4011–4018. https://doi.org/10.1007/s00784-019-02832-0
Rubio F, Wienecke F, Arnabat-Domínguez J, Betancourt P (2022) Photobiomodulation therapy and endodontic treatment of teeth with apical periodontitis using 940-nm diode laser. Report of two cases. J Clin Exp Dent 14:e298–e302. https://doi.org/10.4317/jced.59058
Vatkar NA, Hegde V, Sathe S (2016) Vitality of Enterococcus faecalis inside dentinal tubules after five root canal disinfection methods. J Conserv Dent 19:445–449. https://doi.org/10.4103/0972-0707.190019
Zou L, Shen Y, Li W, Haapasalo M (2010) Penetration of sodium hypochlorite into dentin. J Endod 36:793–796. https://doi.org/10.1016/j.joen.2010.02.005
Aksoy F, Aydin U, Alacam T (2019) Smear layer removal efficiency of Er, Cr; YSGG and Er: YAG lasers in root canals prepared with different NiTi File systems. Annals of Medical Research 26:1545–1550. https://doi.org/10.5455/ANNALSMEDRES.2019.05.291
Montero-Miralles P et al (2018) Comparative study of debris and smear layer removal with EDTA and Er, Cr: YSGG laser. J Clin Exp Dent 10:e598–e602. https://doi.org/10.4317/jced.54936
Merigo E et al (2021) Bactericidal effect of Er, Cr: YSGG laser irradiation on endodontic biofilm: An ex vivo study. J Photochem Photobiol B 218:112185. https://doi.org/10.1016/j.jphotobiol.2021.112185
Beer F, Buchmair A, Wernisch J, Georgopoulos A, Moritz A (2012) Comparison of two diode lasers on bactericidity in root canals—an in vitro study. Lasers Med Sci 27:361–364. https://doi.org/10.1007/s10103-011-0884-3
Acknowledgements
This research is self-funded and didn’t receive any funding from public, commercial, or non-profit sectors.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Sara Zakaria Fahim. Formulating the research question; Collecting patients according to the inclusion/exclusion criteria; Endodontic treatment of the three studied groups; Laser application in the 2 test groups; Collecting microbiologic samples and sending them for bacterial count analysis; Data collection and tabulation; Sending the results for statistical analysis; Writing the manuscript; Critical revision of the article.
Rami Maher Ghali. Formulating the research question;Laser application in the 2 test groups; Interpretation of the statistical results; Writing the manuscript; Critical revision of the article.
Ahmed Abdelrahman Hashem. Formulating the research question; Endodontic treatment of the three studied groups; Collecting microbiologic samples; Interpretation of the statistical results; Writing the manuscript; Critical revision of the article.
Mary Medhat Farid. Formulating the research question; Interpretation of the statistical results; Writing the manuscript; Critical revision of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics Approval and Consent to Participants
The present study followed the Declaration of Helsinki for the ethical principles of medical research involving human subjects and was approved by our institute’s research ethics committee (approval number FDASU-REC ID041908).
Informed written consent was obtained from all subjects and/or their legal guardians.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Clinical trial registration: The trial design was registered at www.clinicaltrials.gov database with identifier number NCT05964686.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fahim, S.Z., Ghali, R.M., Hashem, A.A. et al. The efficacy of 2780 nm Er,Cr;YSGG and 940 nm Diode Laser in root canal disinfection: A randomized clinical trial. Clin Oral Invest 28, 175 (2024). https://doi.org/10.1007/s00784-024-05563-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00784-024-05563-z