Abstract.
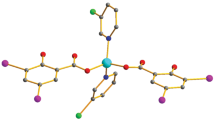
Eight scorpionate-zinc thiolate complexes, [(L1O)ZnSPh], [(L1O)ZnSPhF5], [(L1O)ZnSBz], [(L1O)ZnSPh2,6-Me], [(L1O)ZnSPh2,4-Me], \({\rm [(L1O)ZnSPh}^{{\rm 4 - NO}_{\rm 2} } {\rm ]}\) , [(TpPh,Ph)ZnSPh], and [(L2S)ZnSPh], were reacted with methyl iodide in chloroform, liberating the corresponding methyl thioethers as determined by 1H NMR. Three of these complexes are new and their synthesis and structural characterization are reported here. Weak alkylating agents such as trimethyl phosphate failed to undergo methyl transfer to the zinc thiolates under these conditions. Analysis of kinetic data as a function of concentration, temperature, pK a of the exogenous thiolate, and donor atom of the tripodal ligand are consistent with a mechanism where the zinc-bound thiolate is the active nucleophile in an associative-type methyl transfer reaction. Our model studies also provide experimental evidence to support the hypothesis that some enzymes can use the charge of the metal coordination site to modulate catalytic activity.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Warthen, C., Hammes, B., Carrano, C. et al. Methylation of neutral pseudotetrahedral zinc thiolate complexes: model reactions for alkyl group transfer to sulfur by zinc-containing enzymes. J. Biol. Inorg. Chem. 6, 82–90 (2001). https://doi.org/10.1007/s007750000171
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s007750000171