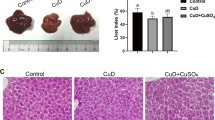
Abstract
Iron [Fe(II)] and copper [Cu(II)] ions produced liver oxidative stress and damage, and as a consequence, changes in the antioxidant protection. The objective of this work is to evaluate whether control of redox homeostasis in chronic overload of Fe(II) and Cu(II) is associated with nitric oxide (NO) and antioxidant enzymes protection in liver. Male Sprague–Dawley rats of 80–90 g received the standard diet ad libitum and drinking water supplemented with either 1.0 g/L of ferrous chloride (0.1% w/v, n = 24) or 0.5 g/L cupric sulfate (0.05% w/v, n = 24) for 42 days. The activities of the enzymes involved in the control of cellular redox homeostasis, nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, superoxide dismutase (SOD), catalase and glutathione peroxidase (GPx), were determined by spectrophotometric methods, and NO production was determined by the determination of nitrite levels in liver. Chronic overload with Fe(II) and Cu(II) led to a significant increase of NO production while hampering the activity of NADPH oxidase. Meanwhile, the animals supplemented with Fe(II) showed a decrease in SOD and Gpx activities in liver homogenates with respect to baseline activity after 7 days of treatment, whereas the rats which received Cu(II) showed an increased SOD and catalase activity after 28 and 7 days of chronic overload. Further research is required to understand whether the modulation of the activity of these enzymes upon Cu and Fe overload is involved in a common toxic pathway or may serve to control the steady state of oxidant species related to redox signaling pathways.
Graphical abstract






Similar content being viewed by others
Abbreviations
- AMP:
-
5′-Adenosine monophosphate
- AMPK:
-
AMP-activated protein kinase
- ATOX1:
-
Antioxidant chaperone for copper 1
- C50 :
-
Metal content necessary to produce the half of the maximal effect
- c-GMP:
-
Cyclic guanosine monophosphate
- CSS:
-
Copper chaperone for superoxide dismutase
- eNOS:
-
Endothelial nitric oxide synthase
- GPx:
-
Glutathione peroxidase
- GSH:
-
Glutathione
- GSSG:
-
Oxidized glutathione
- H2O2 :
-
Hydrogen peroxide
- HO· :
-
Hydroxyl radical
- iNOS:
-
Inducible nitric oxide synthase
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate oxidase
- nNOS:
-
Neuronal nitric oxide synthase
- NO:
-
Nitric oxide
- O2 :
-
Oxygen
- O2 − :
-
Superoxide anion
- 1O2 :
-
Singlet oxygen
- ROOH:
-
Organic hydroperoxides
- ROO. :
-
Hydroperoxyl radical
- SOD:
-
Superoxide dismutase
- SOD1:
-
Cu,Zn-SOD (cytosolic SOD)
- SOD2:
-
Mn-SOD (mitochondrial SOD)
- t 1/2 :
-
Time necessary to produce the half of the maximal effect
References
Theil EC (1987) Ferritin: structure, gene regulation, and cellular function in animals, plants, and microorganisms. Annu Rev Biochem 56:289–315. https://doi.org/10.1146/annurev.bi.56.070187.001445
Liu ZX, Theil EC (2005) Ferritins: dynamic management of biological iron and oxygen chemistry. Acc Chem Res 38:167–175. https://doi.org/10.1021/ar0302336
Lalioti V, Sandoval I, Cassio D, Duclos-Vallée J (2010) Molecular pathology of Wilson’s disease: a brief. J Hepatol 53:151–1153. https://doi.org/10.1016/j.jhep.2010.07.008
Van den Berghe PV, Folmer DE, Malingre HE, van Beurden E, Klomp AEM, van de Sluis B, Merkx M, Berger R, Klomp LWJ (2007) Human copper transporter 2 is localized in late endosomes and lysosomes and facilitates cellular copper uptake. Biochem J 407:49–59. https://doi.org/10.1042/BJ20070705
Harris DIM, Sass-Kortsak A (1967) The influence of amino acids on copper uptake by rat liver slices. J Clin Investig 46:659–667
Musacco-Sebio R, Saporito-Magriñá C, Semprine J, Torti H, Ferrarotti N, Castro-Parodi M, Damiano A, Boveris A, Repetto MG (2014) Rat liver antioxidant response to iron and copper overloads. J Inorg Biochem 137:94–100. https://doi.org/10.1016/j.jinorgbio.2014.04.014
Musacco-Sebio R, Ferrarotti N, Saporito-Magriñá C, Fuda J, Torti H, Lairión F, Boveris A, Repetto MG (2019) Redox dyshomeostasis in the experimental chronic hepatic overloads with iron or copper. J Inorg Biochem 191:119–125. https://doi.org/10.1016/j.jinorgbio.2018.11.014
Boveris A, Cadenas E, Reiter R, Filipkowski M, Nakase Y, Chance B (1980) Organ chemiluminescence: noninvasive assay for oxidative radical reactions. Proc Natl Acad Sci USA 177:347–351. https://doi.org/10.1073/pnas.77.1.347
Vitale AA, Bernatene EA, Vitale MG, Pomilio AB (2016) New insights of the Fenton reaction using glycerol as the experimental model. Effect of O2, inhibition by Mg(2+), and oxidation state of Fe. J Phys Chem A 120:5435–5445. https://doi.org/10.1021/acs.jpca.6b03805
Wiegand HL, Orths CT, Kerpen K, Lutze HV, Schmidt TC (2017) Investigation of the iron-peroxo complex in the fenton reaction: kinetic indication, decay kinetics, and hydroxyl radical yields. Environ Sci Technol 51:14321–14329. https://doi.org/10.1021/acs.est.7b03706
Musacco-Sebio R, Saporito-Magriñá C, Acosta JM, Boveris A, Repetto MG (2017) Iron and copper toxicity in rat liver: a kinetic and holistic overview. Liver Res Open J. https://doi.org/10.17140/LROJ-2-110
Levi S, Corsi B, Bosisio M, Invernizzi R, Volz A, Sanford D, Arosio P, Drysdale J (2001) A human mitochondrial ferritin encoded by an intronless gene. J Biol Chem 276:24437–24440. https://doi.org/10.1074/jbc.C100141200
Walker J, Tsivkovskii R, Lutsenko S (2002) Metallochaperone Atox1 transfers copper to the NH2-terminal domain of the Wilson’s disease protein and regulates its catalytic activity. J Biol Chem 277:27953–27959. https://doi.org/10.1074/jbc.M203845200
Nuttall KL, Palaty J, Lockitch G (2003) Reference limits for copper and iron in liver biopsies. J Ann Clin Lab Sci 33:443–450
Adams PC, Bradley C, Henderson AR (1997) Evaluation of the hepatic iron index as a diagnostic criterion for genetic hemochromatosis. J Lab Clin Med 130:509–514. https://doi.org/10.1016/S0022-2143(97)90127-X
Bacon BR, Adams PC, Kowdley KV, Powell LW, Tavill AS (2011) Diagnosis and management of hemochromatosis: 2011 practice guideline. Hepatology 54:328–343. https://doi.org/10.1002/hep.24330
Ala A, Walker AP, Ashkan K, Dooley JS, Schilsky ML (2007) Wilson’s disease. Lancet 369:397–408. https://doi.org/10.1016/S0140-6736(07)60196-2
Rodriguez-Castro KI, Hevia-Urrutia FJ, Sturniolo G (2015) Wilson’s disease: a review of what we have learned. World J Hepatol 7:2859–2870. https://doi.org/10.4254/wjh.v7.i29.2859
Sengupta P (2013) The laboratory rat: relating its age with human’s. Int J Prev Med 4(6):624–630
Flecknell P (2009) Anaesthesia of common laboratory species: special considerations. Small rodents. In: Laboratory animal anaesthesia, 3rd edn. Academic Press, Elsevier, London
Leary S, Underwood W, Anthony R, Cartner S, Corey D, Grandin T, Greenacre C, Gwaltrey-Brant S, Mc Crackin MA, Meyer R, Miller D, Shearer J, Yanong R (2013) Methods of euthanasia by species and environment. Laboratory animals. In: AVMA guidelines for the euthanasia of animals. American Veterinary Medical Association, Schaumburg
Skoog D, West D, Holler F (2014) Fundamentos de Química Analítica. McGraw-Hill, México
Callaghan J, Gutman S (2006) Counterpoint: food and drug administration guidance for C-reactive protein assays: matching claims with performance data. Clin Chem 52:1256–1257. https://doi.org/10.1373/clinchem.2006.072009
González Flecha B, Llesuy S, Boveris A (1991) Hydroperoxide-initiated chemiluminescence: an assay for oxidative stress in biopsies of heart, liver, and muscle. Free Radic Biol Med 10:93–100. https://doi.org/10.1016/0891-5849(91)90002-k
Boveris A, Oshino N, Chance B (1972) The cellular production of hydrogen peroxide. Biochem J 128:617–627. https://doi.org/10.1042/bj1280617
Boveris A, Musacco-Sebio R, Ferrarotti N, Saporito-Magriñá C, Torti H, Massot F, Repetto MG (2012) The acute toxicity of iron and copper: biomolecule oxidation and oxidative damage in rat liver. J Inorg Biochem 116:63–69. https://doi.org/10.1016/j.jinorgbio.2012.07.004
Ding AH, Nathan CF, Stuehr DJ (1998) Release of reactive nitrogen intermediates and reactive oxygen intermediates from mouse peritoneal macrophages. Comparison of activating cytokines and evidence for independent production. J Immunol 141:2407–2412. https://doi.org/10.1167/iovs.15-16864
Wei Y, Sowers J, Nistala R, Gong H, Uptergrove G, Clark S, Morris M, Szary N, Manrique C, Stump C (2006) Angiotensin II-induced NADPH oxidase activation impairs insulin signaling in skeletal muscle cells. J Biol Chem 281:35137–35146. https://doi.org/10.1074/jbc.m601320200
McCord J, Fridovich I (1969) Superoxide dismutase. An enzymatic function for erythrocuprein hemocuprein. J Biol Chem 244:049–6055
Misra HP, Fridovich I (1972) The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 247:3170–3175
Aebi H (1984) Catalase in vitro. Method Enzymol 10:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Chance B (1954) Determination of catalase activity. In: Glick J (ed) Methods of biochemical analysis. Interscience Publishers, New York
Wendel A (1981) Glutathione peroxidase. Method Enzymol 77:325–333. https://doi.org/10.1016/s0076-6879(81)77046-0
Semprine J, Ferrarotti N, Musacco-Sebio R, Saporito-Magriñá C, Fuda J, Torti H, Castro-Parodi M, Damiano A, Boveris A, Repetto MG (2014) Brain antioxidant responses to acute iron and copper intoxications in rats. Metallomics 6:2083–2089. https://doi.org/10.1039/c4mt00159a
Valko M, Rhodes C, Moncol J, Izakovic M, Mazur M (2006) Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem Biol Interact 160:1–40. https://doi.org/10.1016/j.cbi.2005.12.009
Rao P, Hayon E (1973) Experimental determination of the redox potential of the superoxide radical O2−. Biochem Biophys Res Commun 51:468–473. https://doi.org/10.1016/0006-291X(73)91280-1
Carter D (2005) Environ Health Perspect 103:17–20
Boveris A, Cadenas E (1997) Cellular sources and steady-state levels of reactive oxygen species. In: Biadasz-Clerch L, Massaro DJ (eds) Oxygen, gene expression and cellular function. Marcel Dekker, New York
Saporito Magriñá CM, Musacco Sebio RN, Andrieux G, Kook L, Orrego MT, Tuttolomondo MV, Desimone MF, Boerries M, Borner C, Repetto MG (2018) Copper-induced cell death and the protective role of glutathione: the implication of impaired protein folding rather than oxidative stress. Metallomics 10(12):1743–1754. https://doi.org/10.1039/c8mt00182k
Nordberg J, Arner E (2001) Reactive oxygen species, antioxidants, and the mammalian thioredoxin system. Free Radic Biol Med 31:1287–1312. https://doi.org/10.1016/s0891-5849(01)00724-9
Boveris A, Repetto MG, Bustamante J, Boveris AD, Valdez L (2008) The concept of oxidative stress in pathology. In: Alvarez S, Evelson P, Boveris A (eds) Free radical pathophysiology. Research Signpost, Kerala
Sies H (2016) The concept of oxidative stress after 30 years. In: Gelpi RJ, Boveris A, Poderoso JJ (eds) Biochemistry of oxidative stress. Springer, Switzerland. https://doi.org/10.1007/978-3-319-45865-6_1
Sies H (1991) Oxidative stress: from basic research to clinical application. Am J Med 91:31S-38S. https://doi.org/10.1016/0002-9343(91)90281-2
Sies H (2015) Oxidative stress: a concept in redox biology and medicine. Redox Biol 4:180–183. https://doi.org/10.1016/j.redox.2015.01.002
Boveris A, Poderoso JJ (2000) In: Ignaro (ed) Nitric oxide. Biology and pathobiology. Academic Press, Los Angeles
Finkel T (2000) Redox dependent signal transduction. FEBS Lett 476:52–54. https://doi.org/10.1016/S0014-5793(00)01669-0
Bedard K, Krause K (2007) The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol Rev 87:245–313. https://doi.org/10.1152/physrev.00044.2005
Bastian N, Hibbs J (1994) Assambly and regulation of NADPH oxidase and nitric oxidase synthase. Curr Opin Immunol 6:131–139
Ansari H, Roberts K, Scheff S (2014) A time course of NADPH-oxidase up-regulation and endothelial nitric oxide synthase activation in the hippocampus following neurotrauma. Free Radic Biol Med 77:21–29. https://doi.org/10.1016/J.freeradbiomed.2014.08.025
Qian J, Chen F, Kovalenkou Y, Pandey D, Moseleley M, Foster M, Black S, Venema R, Stopp D, Fultn D (2012) Nitric oxide reduces NADPH oxidase 5 (NOX 5) activity by reversible S-nitrosylation. Free Radic Biol Med 52:1806–1819. https://doi.org/10.1016/J.freeradbiomed.2012.02.029
Cordero-Herrera I, Kozyra M, Zhuge Z, McCann Z, Moretti C, Peleli M, Caldeira-Dias M, Jahandideh A, Huirong H, de Campos CJ, Kleschyov A, Montenegro M, Ingelman-Sundberg M, Weitzberg E, Lundberg J, Carlstroma M (2019) AMP-activated protein kinase activation and NADPH oxidase inhibition by inorganic nitrate and nitrite prevent liver steatosis. Proc Natl Acad Sci U S A 116(1):217–226. https://doi.org/10.1073/pnas.1809406115
Fridovich I (1974) Superoxide dismutases. Adv Enzymol 41:35–97. https://doi.org/10.1002/9780470122860.ch2
Brown N, Torres A, Doan P, O´Halloran T (2004) Oxygen and the copper chaperone CCS regulate posttranslational activation of Cu, Zn superoxide dismutase. PNAS 101:5518–5523. https://doi.org/10.1073/pnas.0401175101
Ozcelik D, Ozares R, Gurel Z, Uzun H, Aydin S (2003) Oxygen and the copper chaperone CCS regulate posttranslational activation of Cu, Zn superoxide dismutase. Biol Trace Elem Res 96:209–215. https://doi.org/10.1073/pnas.0401175101
Sansinanea AS, Cerone SI, Streitenberger SA, García C, Auza N (1998) Oxidative effect of hepatic copper overload. Acta Physiol Pharmacol Ther Latinoam 48:25–31
Zhang SS, Noordin MM, Rahman SO, Haron J (2000) Effects of copper overload on hepatic lipid peroxidation and antioxidant defense in rats. Vet Hum Toxicol 42:261–264
Repetto M, Ferrarotti N, Boveris A (2010) The involvement of transition metal ions on iron depended lipid peroxidation. Arch Toxicol 84:255–262
Russanov EM, Kassabova TA, Konstantinova SG, Balevska PS (1986) Effect of chronic copper loading on the activity of rat liver antioxidative enzymes. Acta Physiol Pharmacol Bulg 12:51–56
Ossola JO, Groppa MD, Tomaro ML (1997) Heme oxygenase induction by menadione bisulfite adduct-generated oxidative stress in rat liver. Arch Biochem Biophys 337:332–337. https://doi.org/10.1016/s0742-8413(00)00133-x
Acknowledgements
Authors thank Professor Alberto Boveris for his teachings and scientific contributions at the beginning of this research work, guiding the development of this experimental line. This study was supported by grants from the University of Buenos Aires (UBACyT 20020170100197BA); the National Research Council of Argentina (CONICET) and the National Agency of Science and Technology of Argentina (ANPCYT) (PICT-2016-002077).
Author information
Authors and Affiliations
Contributions
FL: validation, formal analysis, investigation, and writing—original–draft; CS-M: methodology, validation, investigation, and visualization; RM-S: methodology and investigation; JF: investigation; HT: investigation and resources; MGR: conceptualization, formal analysis, and writing—original–draft, writing, review and editing, project administration, and funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
None of the authors has anything to disclose regarding the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lairion, F., Saporito-Magriñá, C., Musacco-Sebio, R. et al. Nitric oxide, chronic iron and copper overloads and regulation of redox homeostasis in rat liver. J Biol Inorg Chem 27, 23–36 (2022). https://doi.org/10.1007/s00775-021-01908-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-021-01908-1