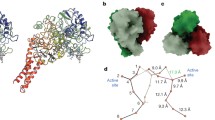
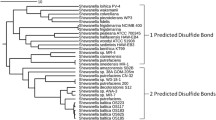
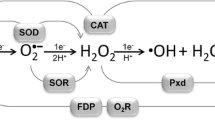
Abstract
Multicopper oxidases (MCOs) are unique among copper proteins in that they contain at least one each of the three types of biologic copper sites, type 1, type 2, and the binuclear type 3. MCOs are descended from the family of small blue copper proteins (cupredoxins) that likely arose as a complement to the heme-iron-based cytochromes involved in electron transport; this event corresponded to the aerobiosis of the biosphere that resulted in the conversion of Fe(II) to Fe(III) as the predominant redox state of this essential metal and the solubilization of copper from Cu2S to Cu(H2O) n 2+. MCOs are encoded in genomes in all three kingdoms and play essential roles in the physiology of essentially all aerobes. With four redox-active copper centers, MCOs share with terminal copper-heme oxidases the ability to catalyze the four-electron reduction of O2 to two molecules of water. The electron transfers associated with this reaction are both outer and inner sphere in nature and their mechanisms have been fairly well established. A subset of MCO proteins exhibit specificity for Fe2+, Cu+, and/or Mn2+ as reducing substrates and have been designated as metallooxidases. These enzymes, in particular the ferroxidases found in all fungi and metazoans, play critical roles in the metal metabolism of the expressing organism.













Similar content being viewed by others
Abbreviations
- Cp:
-
Ceruloplasmin
- hCp:
-
Human ceruloplasmin
- Hp:
-
Hephaestin
- LUMO:
-
Lowest unoccupied molecular orbital
- MCO:
-
Multicopper oxidase
- NiR:
-
Nitrite reductase
- SLAC:
-
Small laccase
- T1:
-
Type 1
- T2:
-
Type 2
- T3:
-
Type 3
- TNC:
-
Trinuclear cluster
References
Murphy ME, Lindley PF, Adman ET (1997) Protein Sci 6:761–770
Nakamura K, Kawabata T, Yura K, Go N (2003) FEBS Lett 553:239–244
Nakamura K, Go N (2005) Cell Mol Life Sci 62:2050–2066
Hoegger PJ, Kilaru S, James TY, Thacker JR, Kues U (2006) FEBS J 273:2308–2326
Ellis MJ, Grossmann JG, Eady RR, Hasnain SS (2007) J Biol Inorg Chem 12:1119–1127
Solomon EI, Penfield KW, Gewirth AA, Lowery MD, Shadle SE, Guckert JA, LaCroix LB (1996) Inorg Chim Acta 243:67–78
Palmer AE, Randall DW, Feng X, Solomon EI (1999) J Am Chem Soc 121:7138–7149
Szilagyi RK, Solomon EI (2002) Curr Opin Chem Biol 6:250–258
Solomon EI, Szilagyi RK, DeBeer George S, Basumallick L (2004) Chem Rev 104:419–458
Sakurai T, Kataoka K (2007) Cell Mol Life Sci 64:2642–2656. doi:10.1007/s00018-007-7183-y
Malmstrom BG, Reinhammar B, Vanngard T (1968) Biochim Biophys Acta 156:67–76
Malkin R, Malmstrom BG (1970) Adv Enzymol Relat Areas Mol Biol 33:177–244
Marcus RA, Sutin N (1985) Biochim Biophys Acta 811:265–322
Stoj CS, Augustine AJ, Zeigler L, Solomon EI, Kosman DJ (2006) Biochemistry 45:12741–12749
Quintanar L, Stoj C, Taylor AB, Hart PJ, Kosman DJ, Solomon EI (2007) Acc Chem Res 40:445–452. doi:10.1021/ar600051a
Kosman DJ (2008) Inorg Chim Acta 361:844–849. doi:10.1016/j.ica.2007.10.013
Solomon EI, Augustine AJ, Yoon J (2008) Dalton Trans 3921–3932. doi:10.1039/b800799c
Tadesse MA, D’Annibale A, Galli C, Gentili P, Sergi F (2008) Org Biomol Chem 6:868–878. doi:10.1039/b716002j
Terzulli AJ, Kosman DJ (2009) J Biol Inorg Chem 14:315–325. doi:10.1007/s00775-008-0450-z
Rosenzweig AC, Sazinsky MH (2006) Curr Opin Struct Biol 16:729–735. doi:10.1016/j.sbi.2006.09.005
Yoon J, Solomon EI (2007) J Am Chem Soc 129:13127–13136. doi:10.1021/ja073947a
Quintanar L, Stoj C, Wang TP, Kosman DJ, Solomon EI (2005) Biochemistry 44:6081–6091
Yoon J, Solomon EI (2005) Inorg Chem 44:8076–8086. doi:10.1021/ic0507870
Augustine AJ, Quintanar L, Stoj CS, Kosman DJ, Solomon EI (2007) J Am Chem Soc 129:13118–13126. doi:10.1021/ja073905m
Kataoka K, Sugiyama R, Hirota S, Inoue M, Urata K, Minagawa Y, Seo D, Sakurai T (2009) J Biol Chem 284:14405–14413. doi:10.1074/jbc.M808468200
Ghosh S, Dey A, Sun Y, Scholes CP, Solomon EI (2009) J Am Chem Soc 131:277–288. doi:10.1021/ja806873e
Stoj CS, Kosman DJ (2005) In: King RB (ed) Encyclopedia of inorganic chemistry. Wiley, New York, pp 1134–1159
Taylor AB, Stoj CS, Ziegler L, Kosman DJ, Hart PJ (2005) Proc Natl Acad Sci USA 102:15459–15464
Thompson IA, Huber DM, Schulze DG (2006) Phytopathology 96:130–136. doi:10.1094/PHYTO-96-0130
Miyata N, Tani Y, Sakata M, Iwahori K (2007) J Biosci Bioeng 104:1–8. doi:10.1263/jbb.104.1
Ridge JP, Lin M, Larsen EI, Fegan M, McEwan AG, Sly LI (2007) Environ Microbiol 9:944–953
Dick GJ, Torpey JW, Beveridge TJ, Tebo BM (2008) Appl Environ Microbiol 74:1527–1534. doi:10.1128/AEM.01240-07
Djoko KY, Xiao Z, Wedd AG (2008) Chembiochem 9:1579–1582. doi:10.1002/cbic.200800100
Hall SJ, Hitchcock A, Butler CS, Kelly DJ (2008) J Bacteriol 190:8075–8085. doi:10.1128/JB.00821-08
Hellman NE, Gitlin JD (2002) Annu Rev Nutr 22:439–458
Miyajima H (2002) Intern Med 41:762–769
Petrak J, Vyoral D (2005) Int J Biochem Cell Biol 37:1173–1178. doi:10.1016/j.biocel.2004.12.007
Xu X, Pin S, Gathinji M, Fuchs R, Harris ZL (2004) Ann N Y Acad Sci 1012:299–305
Torsdottir G, Sveinbjornsdottir S, Kristinsson J, Snaedal J, Johannesson T (2006) J Neurol Sci 241:53–58. doi:10.1016/j.jns.2005.10.015
Madsen E, Gitlin JD (2007) Annu Rev Neurosci 30:317–337. doi:10.1146/annurev.neuro.30.051606.094232
Barnham KJ, Bush AI (2008) Curr Opin Chem Biol 12:222–228. doi:10.1016/j.cbpa.2008.02.019
di Patti MC, Maio N, Rizzo G, De Francesco G, Persichini T, Colasanti M, Polticelli F, Musci G (2009) J Biol Chem 284:4545–4554. doi:10.1074/jbc.M805688200
Jeong SY, Rathore KI, Schulz K, Ponka P, Arosio P, David S (2009) J Neurosci 29:610–619. doi:10.1523/JNEUROSCI.5443-08.2009
Adman ET (1991) Adv Protein Chem 42:145–197
DiSpirito AA, Taaffe LR, Lipscombe JD, Hooper AB (1985) Biochim Biophys Acta 827:320–326
Boulanger MJ, Murphy ME (2002) J Mol Biol 315:1111–1127. doi:10.1006/jmbi.2001.5251S0022283601952519
Machczynski MC, Vijgenboom E, Samyn B, Canters GW (2004) Protein Sci 13:2388–2397
Lawton TJ, Sayavedra-Soto LA, Arp DJ, Rosenzweig AC (2009) J Biol Chem 284:10174–10180. doi:10.1074/jbc.M900179200
Skalova T, Dohnalek J, Ostergaard LH, Ostergaard PR, Kolenko P, Duskova J, Stepankova A, Hasek J (2009) J Mol Biol 385:1165–1178. doi:10.1016/j.jmb.2008.11.024
Lindley PF, Card G, Zaitseva I, Zaitsev V, Reinhammar B, Selin-Lindgren E, Yoshida K (1997) J Biol Inorg Chem 2:454–463
Zaitsev VN, Zaitseva I, Papiz M, Lindley PF (1999) J Biol Inorg Chem 4:579–587
Machonkin TE, Solomon EI (2000) J Am Chem Soc 122:12547–12560
La Fontaine S, Quinn JM, Nakamoto SS, Page MD, Gohre V, Moseley JL, Kropat J, Merchant S (2002) Eukaryot Cell 1:736–757
Barrett ML, Harvey I, Sundararajan M, Surendran R, Hall JF, Ellis MJ, Hough MA, Strange RW, Hillier IH, Hasnain SS (2006) Biochemistry 45:2927–2939. doi:10.1021/bi052372w
Piontek K, Antorini M, Choinowski T (2002) J Biol Chem 277:37663–37669
Zaitseva I, Zaitsev V, Card G, Moshkov K, Bax B, Ralph A, Lindley P (1996) J Biol Inorg Chem 1:15–23
Solomon EI (2006) Inorg Chem 45:8012–8025. doi:10.1021/ic060450d
Vallee BL, Williams RJ (1968) Proc Natl Acad Sci USA 59:498–505
Gray HB, Malmstrom BG, Williams RJ (2000) J Biol Inorg Chem 5:551–559
Betts JN, Beratan DN, Onuchic JN (1992) J Am Chem Soc 114:4043–4046
Balaban IA, Onuchic JN (1996) J Chem Phys 100:11573–11580
Bertrand T, Jolivalt C, Briozzo P, Caminade E, Joly N, Madzak C, Mougin C (2002) Biochemistry 41:7325–7333
Quintanar L, Gebhard M, Wang TP, Kosman DJ, Solomon EI (2004) J Am Chem Soc 126:6579–6589
Onuchic JN, Beratan DN, Winkler JR, Gray HB (1992) Annu Rev Biophys Biomol Struct 21:349–377
Augustine AJ, Kragh ME, Sarangi R, Fujii S, Liboiron BD, Stoj CS, Kosman DJ, Hodgson KO, Hedman B, Solomon EI (2008) Biochemistry 47:2036–2045. doi:10.1021/bi7020052
Rulisek L, Solomon EI, Ryde U (2005) Inorg Chem 44:5612–5628
Solomon EI, Chen P, Metz M, Lee SK, Palmer AE (2001) Angew Chem Int Ed 40:4570–4590
Yoon J, Liboiron BD, Sarangi R, Hodgson KO, Hedman B, Solomon EI (2007) Proc Natl Acad Sci USA 104:13609–13614. doi:10.1073/pnas.0705137104
Francis CA, Tebo BM (2002) Appl Environ Microbiol 68:874–880
Grass G, Rensing C (2001) Biochem Biophys Res Commun 286:902–908
Lee SM, Grass G, Rensing C, Barrett SR, Yates CJ, Stoyanov JV, Brown NL (2002) Biochem Biophys Res Commun 295:616–620
Shi X, Stoj C, Romeo A, Kosman DJ, Zhu Z (2003) J Biol Chem 278:50309–50315
Stoj C, Kosman DJ (2003) FEBS Lett 554:422–426
Stoj CS, Augustine AJ, Solomon EI, Kosman DJ (2007) J Biol Chem 282:7862–7868. doi:10.1074/jbc.M609766200
Roberts SA, Weichsel A, Grass G, Thakali K, Hazzard JT, Tollin G, Rensing C, Montfort WR (2002) Proc Natl Acad Sci USA 99:2766–2771
Roberts SA, Wildner GF, Grass G, Weichsel A, Ambrus A, Rensing C, Montfort WR (2003) J Biol Chem 278:31958–31963
Singh SK, Grass G, Rensing C, Montfort WR (2004) J Bacteriol 186:7815–7817. doi:10.1128/JB.186.22.7815-7817.2004
Kataoka K, Komori H, Ueki Y, Konno Y, Kamitaka Y, Kurose S, Tsujimura S, Higuchi Y, Kano K, Seo D, Sakurai T (2007) J Mol Biol 373:141–152. doi:10.1016/j.jmb.2007.07.041
Jung WH, Sham A, White R, Kronstad JW (2006) PLoS Biol 4:e410. doi:10.1371/journal.pbio.0040410
Jung WH, Kronstad JW (2008) Cell Microbiol 10:277–284. doi:10.1111/j.1462-5822.2007.01077.x
Jung WH, Sham A, Lian T, Singh A, Kosman DJ, Kronstad JW (2008) PLoS Pathog 4:e45. doi:10.1371/journal.ppat.0040045
Kwok EY, Kosman DJ (2006) Top Curr Genet 14:59–99
Kwok EY, Severance S, Kosman DJ (2006) Biochemistry 45:6317–6327. doi:10.1021/bi052173c
Singh A, Severance S, Kaur N, Wiltsie W, Kosman DJ (2006) J Biol Chem 281:13355–13564
Tree JJ, Kidd SP, Jennings MP, McEwan AG (2005) Biochem Biophys Res Commun 328:1205–1210. doi:10.1016/j.bbrc.2005.01.084
Chidambaram MV, Barnes G, Frieden E (1983) FEBS Lett 159:137–140
Griffiths TA, Mauk AG, MacGillivray RT (2005) Biochemistry 44:14725–14731. doi:10.1021/bi051559k
Paz Y, Katz A, Pick U (2007) J Biol Chem 282:8658–8666. doi:10.1074/jbc.M609756200
Fisher M, Zamir A, Pick U (1998) J Biol Chem 273:17553–17558
Ganz T (2005) Cell Metab 1:155–157. doi:10.1016/j.cmet.2005.02.005
De Domenico I, Ward DM, di Patti MC, Jeong SY, David S, Musci G, Kaplan J (2007) EMBO J 26:2823–2831. doi:10.1038/sj.emboj.7601735
Anderson GJ, Frazer DM, McKie AT, Vulpe CD (2002) Blood Cells Mol Dis 29:367–375
Chen H, Attieh ZK, Su T, Syed BA, Gao H, Alaeddine RM, Fox TC, Usta J, Naylor CE, Evans RW, McKie AT, Anderson GJ, Vulpe CD (2004) Blood 103:3933–3939. doi:10.1182/blood-2003-09-31392003-09-3139
Crichton RR, Pierre JL (2001) Biometals 14:99–112
Pierre JL, Fontecave M, Crichton RR (2002) Biometals 15:341–346
Acknowledgments
The author gratefully acknowledges the several members of his laboratory and his collaborators Edward Solomon and P. John Hart who have together assembled the experimental data that form the basis of our understanding of MCO proteins and their role in metal metabolism. The work in the Kosman laboratory was supported by NIH RO1 DK053820.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article will be printed in the upcoming Journal of Biological Inorganic Chemistry special issue Cell Biology of Copper.
Rights and permissions
About this article
Cite this article
Kosman, D.J. Multicopper oxidases: a workshop on copper coordination chemistry, electron transfer, and metallophysiology. J Biol Inorg Chem 15, 15–28 (2010). https://doi.org/10.1007/s00775-009-0590-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-009-0590-9