Abstract
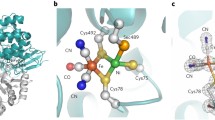
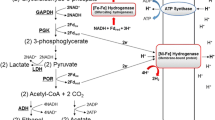
The genome of Desulfovibrio vulgaris Hildenborough (DvH) encodes for six hydrogenases (Hases), making it an interesting organism to study the role of these proteins in sulphate respiration. In this work we address the role of the [NiFeSe] Hase, found to be the major Hase associated with the cytoplasmic membrane. The purified enzyme displays interesting catalytic properties, such as a very high H2 production activity, which is dependent on the presence of phospholipids or detergent, and resistance to oxygen inactivation since it is isolated aerobically in a Ni(II) oxidation state. Evidence was obtained that the [NiFeSe] Hase is post-translationally modified to include a hydrophobic group bound to the N-terminal, which is responsible for its membrane association. Cleavage of this group originates a soluble, less active form of the enzyme. Sequence analysis shows that [NiFeSe] Hases from Desulfovibrionacae form a separate family from the [NiFe] enzymes of these organisms, and are more closely related to [NiFe] Hases from more distant bacterial species that have a medial [4Fe4S]2+/1+ cluster, but not a selenocysteine. The interaction of the [NiFeSe] Hase with periplasmic cytochromes was investigated and is similar to the [NiFe]1 Hase, with the Type I cytochrome c 3 as the preferred electron acceptor. A model of the DvH [NiFeSe] Hase was generated based on the structure of the Desulfomicrobium baculatum enzyme. The structures of the two [NiFeSe] Hases are compared with the structures of [NiFe] Hases, to evaluate the consensual structural differences between the two families. Several conserved residues close to the redox centres were identified, which may be relevant to the higher activity displayed by [NiFeSe] Hases.








Similar content being viewed by others
Abbreviations
- DvH:
-
D. vulgaris Hildenborough
- Dm :
-
Desulfomicrobium
- Hase:
-
Hydrogenase
- [NiFeSe]m :
-
Membrane-bound form of the [NiFeSe] Hase
- [NiFeSe]s :
-
Soluble form of the [NiFeSe] Hase
- TpIc 3 :
-
Type I cytochrome c 3
- TpIIc 3 :
-
Type II cytochrome c 3
- HmcA:
-
16-haem high molecular weight cytochrome c
References
Woodward J, Orr M, Cordray K, Greenbaum E (2000) Nature 405: 1014–1015
Hallenbeck PC, Benemann JR (2002) Int J Hydrogen Energ 27: 1185–1193
Kalia VC, Lal S, Ghai R, Mandal M, Chauhan A (2003) Trends Biotechnol 21:152–156
Nandi R, Sengupta S (1998) Crit Rev Microbiol 24:61–84
Armstrong FA (2004) Curr Opin Chem Biol 8:133–140
Jones AK, Sillery E, Albracht SPJ, Armstrong FA (2002) Chem Commun 8:866–867
Mertens R, Liese A (2004) Curr Opin Biotechnol 15:343–348
Schwartz E, Friedrich B (2003) In: Dworkin M et al. (ed) The prokaryotes: an evolving electronic resource for the microbiological community. Springer, Berlin Heidelberg New York. http://link.springer-ny.com/link/service/books/10125/. New York,
Odom JM, Peck HD Jr (1981) FEMS Microbiol Lett 12:47–50
Vignais PM, Billoud B, Meyer J (2001) FEMS Microbiol Rev 25: 455–501
Vignais PM, Colbeau A (2004) Curr Issues Mol Biol 6:159–188
Wu LF, Mandrand MA (1993) FEMS Microbiol Rev 10:243–269
Matias PM, Pereira IAC, Soares CM, Carrondo MA (2005) Prog Biophys Mol Biol 89:292–329
Fauque G, Peck HD Jr, Moura JJ, Huynh BH, Berlier Y, DerVartanian DV, Teixeira M, Przybyla AE, Lespinat PA, Moura I, LeGall J (1988) FEMS Microbiol Rev 4:299–344
Frey M, Fontecilla-Camps JC, Volbeda A (2001) In: Messerschmidt A, Huber R, Wieghardt K, Poulos T (eds) Handbook of metalloproteins, Wiley, New York, pp 880–896
Lemon BJ, Peters JW (2001) In: Messerschmidt A, Huber R, Wieghardt K, Poulos T (eds) Handbook of metalloproteins, Wiley, New York, pp 738–751
Voordouw G, Niviere V, Ferris FG, Fedorak PM, Westlake DWS (1990) Appl Environ Microbiol 56:3748–3754
Rossi M, Pollock WBR, Reij MW, Keon RG, Fu R, Voordouw G (1993) J Bacteriol 175:4699–4711
Pereira IAC, Romão CV, Xavier AV, LeGall J, Teixeira M (1998) J Biol Inorg Chem 3:494–498
Valente FMA, Saraiva LM, LeGall J, Xavier AV, Teixeira M, Pereira IAC (2001) ChemBioChem 2:895–905
Heidelberg J F, Seshadri R, Haveman SA, Hemme CL, Paulsen IT, Kolonay JF, Eisen JA, Ward N, Methe B, Brinkac LM, Daugherty SC, Deboy RT, Dodson RJ, Durkin AS, Madupu R, Nelson WC, Sullivan SA, Fouts D, Haft DH, Selengut J, Peterson JD, Davidsen TM, Zafar N, Zhou LW, Radune D, Dimitrov G, Hance M, Tran K, Khouri H, Gill J, Utterback TR, Feldblyum TV, Wall JD, Voordouw G, Fraser CM (2004) Nat Biotechnol 22: 554–559
Rodrigues R, Valente FM, Pereira IA, Oliveira S, Rodrigues-Pousada C (2003) Biochem Biophys Res Commun 306:366–375
Hedderich R (2004) J Bioenerg Biomembr 36:65–75
Casalot L, Valette O, De Luca G, Dermoun Z, Rousset M, de Philip P (2002) FEMS Microbiol Lett 214:107–112
Casalot L, De Luca G, Dermoun Z, Rousset M, de Philip P (2002) J Bacteriol 184:853–856
Pohorelic BK, Voordouw JK, Lojou E, Dolla A, Harder J, Voordouw G (2002) J Bacteriol 184:679–686
Romao CV, Pereira IA, Xavier AV, LeGall J, Teixeira M (1997) Biochem Biophys Res Commun 240:75–79
Lissolo T, Choi ES, LeGall J, Peck HD Jr (1986) Biochem Biophys Res Commun 139:701–708
Yagi T (1970) J Biochem (Tokyo) 68:649–657
Rieder R, Cammack R, Hall DO (1984) Eur J Biochem 145:637–643
Teixeira M, Fauque G, Moura I, Lespinat PA, Berlier Y, Prickril B, Peck HD, Xavier AV, Legall J, Moura JJG (1987) Eur J Biochem 167:47–58
Voordouw G, Menon NK, LeGall J, Choi ES, Peck HD Jr, Przybyla A E (1989) J Bacteriol 171:2894–2899
He SH, Teixeira M, LeGall J, Patil DS, Moura I, Moura JJ, DerVartanian DV, Huynh BH, Peck HD Jr (1989) J Biol Chem 264: 2678–2682
Pereira AS, Franco R, Feio MJ, Pinto C, Lampreia J, Reis MA, Calvete J, Moura I, Beech I, Lino AR, Moura JJ (1996) Biochem Biophys Res Commun 221:414–421
Eidsness MK, Scott RA, Prickril BC, DerVartanian DV, Legall J, Moura I, Moura JJ, Peck HD Jr (1989) Proc Natl Acad Sci USA 86: 147–151
Garcin E, Vernede X, Hatchikian EC, Volbeda A, Frey M, Fontecilla-Camps JC (1999) Structure 7:557–566
Sorgenfrei O, Duin EC, Klein A, Albracht SP (1996) J Biol Chem 271: 23799–23806
Sorgenfrei O, Duin EC, Klein A, Albracht SP (1997) Eur J Biochem 247:681–687
Legall J, Payne WJ, Chen L, Liu MY, Xavier AV (1994) Biochimie 76: 655–665
Ackrell BAC, Asato RN, Mower HF (1966) J Bacteriol 92:828–838
Wardi AH, Michos GA (1972) Anal Biochem 49:607–609
Teixeira M, Campos AP, Aguiar AP, Costa HS, Santos H, Turner DL, Xavier AV (1993) FEBS Lett 317:233–236
Pandey A, Andersen JS, Mann M (2000) Science’s Stke 37:1–12
Gonnet F, Lemaître G, Waksman G, Tortajada J (2003) Proteome Sci 1:2
Peck HD Jr, Gest H (1956) J Bacteriol 71:70–80
Lallamaharajh WV, Hall DO, Cammack R, Rao KK, Legall J (1983) Biochem J 209:445–454
Bjorklof K, Zickermann V, Finel M (2000) FEBS Lett 467:105–110
Sanchez R, Sali A (1997) Curr Opin Struct Biol 7:206–214
Marti-Renom MA, Stuart AC, Fiser A, Sanchez R, Melo F, Sali A (2000) Annu Rev Biophys Biomol Struct 29:291–325
Sali A, Blundell TL (1993) J Mol Biol 234:779–815
Laskowski RA, Macarthur MW, Moss DS, Thornton JM (1993) J Appl Crystallogr 26:283–291
Meuer J, Kuettner HC, Zhang JK, Hedderich R, Metcalf WW (2002) Proc Natl Acad Sci USA 99:5632–5637
Fox JD, Kerby RL, Roberts GP, Ludden PW (1996) J Bacteriol 178: 1515–1524
Haveman SA, Brunelle V, Voordouw JK, Voordouw G, Heidelberg JF, Rabus R (2003) J Bacteriol 185:4345–4353
Hayashi S, Wu HC (1990) J Bioenerg Biomembr 22:451–471
Makula RA, Finnerty WR (1974) J Bacteriol 120:1279–1283
Volbeda A, Martin L, Cavazza C, Matho M, Faber BW, Roseboom W, Albracht SP, Garcin E, Rousset M, Fontecilla-Camps JC (2005) J Biol Inorg Chem 10:239–249
Sebban C, Blanchard L, Bruschi M, Guerlesquin F (1995) FEMS Microbiol Lett 133:143–149
Costa C, Teixeira M, LeGall J, Moura JJG, Moura I (1997) J Biol Inorg Chem 2:198–208
Pieulle L, Haladjian J, Bonicel J, Hatchikian EC (1996) Biochem Biophys Acta 1273:51–61
Matias PM, Coelho R, Pereira IAC, Coelho AV, Thompson AW, Sieker LC, LeGall J, Carrondo MA (1999) Structure 7:119–130
Aubert C, Brugna M, Dolla A, Bruschi M, Giudici-Orticoni MT (2000) Biochim Biophys Acta 1476:85–92
Matias PM, Coelho AV, Valente FMA, Ptacido D, LeGall J, Xavier AV, Pereira IAC, Carrondo MA (2002) J Biol Chem 277:47907–47916
Frazão C, Sieker L, Sheldrick G, Lamzin V, LeGall J, Carrondo MA (1999) J Biol Inorg Chem 4:162–165
Czjzek M, Guerlesquin F, Bruschi M, Haser R (1996) Structure 4: 395–404
Peck HDJ, Lissolo T (1988) In: Cole JA, Ferguson SJ (eds) The nitrogen and sulphur cycles. Cambridge University Press, Cambridge, pp 99–132
Rohde M, Furstenau U, Mayer F, Przybyla AE, Peck HD Jr, Le Gall J, Choi ES, Menon NK (1990) Eur J Biochem 191:389–396
Casalot L, Rousset M (2001) Trends Microbiol 9:228–237
Price MN, Huang KH, Alm EJ, Arkin AP (2005) Nucleic Acids Res 33: 880–892
Akoh CC, Lee GC, Liaw YC, Huang TH, Shaw JF (2004) Prog Lipid Res 43:534–552
Page CC, Moser CC, Chen X, Dutton PL (1999) Nature (London) 402: 47–52
Bingemann R, Klein A (2000) Eur J Biochem 267:6612–6618
Rousset M, Montet Y, Guigliarelli B, Forget N, Asso M, Bertrand P, Fontecilla-Camps JC, Hatchikian EC (1998) Proc Natl Acad Sci USA 95:11625–11630
Higuchi Y, Yagi T, Yasuoka N (1997) Structure 5:1671–1680
Volbeda A, Charon MH, Piras C, Hatchikian EC, Frey M, Fontecilla-Camps JC (1995) Nature 373:580–587
De Gioia L, Fantucci P, Guigliarelli B, Bertrand P (1999) Int J Quantum Chem 73:187–195
Matias PM, Soares CM, Saraiva LM, Coelho R, Morais J, Le Gall J, Carrondo MA (2001) J Biol Inorg Chem 6:63–81
Delano W (2003) Delano Scientific LLC, San Carlos
Acknowledgements
We would like to thank Mr. João Carita and the staff of the IBET Fermentation Plant for growing the bacterial cells, Mrs. M.Regalla for N-terminal sequence determinations, and Ms. Elisabete Pires for mass spectrometry analyses. We would also like to thank Dr. John Heidelberg from The Institute of Genomic Research for allowing us access to the DvH list of annotated genes prior to publication. This work was supported by FCT grants POCTI/ESP/44782/02 to I.A.C.P., POCTI/BME/32789/99 to C.M.S. and A.S.F.O., and POCTI/QUI/47866/02 to A.V.X. F.M.A.V. is supported by a FCT PhD grant (SFRH/BD/9187/2002).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Valente, F.M.A., Oliveira, A.S.F., Gnadt, N. et al. Hydrogenases in Desulfovibrio vulgaris Hildenborough: structural and physiologic characterisation of the membrane-bound [NiFeSe] hydrogenase. J Biol Inorg Chem 10, 667–682 (2005). https://doi.org/10.1007/s00775-005-0022-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-005-0022-4