Abstract
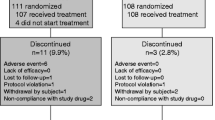
Concurrent treatments with bisphosphonates and vitamin K are promising given that bisphosphonates possibly interfere with vitamin K activation. This is a prospective, multi-center, open-labeled, randomized trial of the efficacy of concurrent treatment with vitamin K2 and risedronate compared with risedronate alone and to explore subsets of patients for which concurrent treatment is particularly efficacious (trial identification number UMIN000000991). Inclusion criteria are women who meet the criteria for pharmacological therapy for osteoporosis, aged ≥65 years, have any of pre-specified risk factors, can walk unassisted, and are able to answer questionnaires. Exclusion criteria are prior warfarin use, secondary osteoporosis or non-osteoporotic metabolic bone diseases, contraindication for vitamin K2 and risedronate, hyper- or hypoparathyroidism, mental disorders, prevalent vertebral fracture at ≥6 sites, severe degenerative spinal deformation between T4 and L4, serious heart, liver, or kidney disease, or bisphosphonate use within the previous 6 months. Patients were recruited from 123 institutes between January 2008 and February 2010, and allocated to vitamin K2 (45 mg/day) and risedronate (2.5 mg/day or 17.5 mg/week) or risedronate alone (2.5 mg/day or 17.5 mg/week) groups. Primary endpoint is a vertebral or non-vertebral fracture. Secondary endpoints are bone mineral density, height, undercarboxylated osteocalcin, JOQOL, EQ-5D and safety. A sample size of 910 subjects per group and 2-year follow-up will provide 80 % power to detect 35 % risk reduction for fracture, with a two-sided significance level of 5 %. Subgroup analysis stratified to adjustment factors for random allocation, body mass index, 25-hydroxyvitamin D, estimated glomerular filtration rate, grade of vertebral fracture, JOQOL, EQ-5D, and co-morbidity is pre-specified.

Similar content being viewed by others
References
Orimo H (2007) The Japanese guidelines for the prevention and treatment of osteoporosis (2006 Edition). Osteoporos Jpn 15:351–355
Luckman SP, Hughes DE, Coxon FP, Russell RGG, Rogers MJ (1998) Nitrogen-containing bisphosphonates inhibit the mevalonate pathway and prevent post-translational prenylation of GTP binding proteins, including rats. J Bone Miner Res 13:581–589
Liberman UA, Weiss SR, Broll J, Minne HW, Quan H, Bell NH, Rodriguez-Portales J, Downs RW, Dequeker J, Favus M, Seeman E, Recker R, Capizzi T, Santora AC, Lombardi A, Shah RV, Hirsch LJ, Karpf DB, For the Alendronate Phase III Osteoporosis Treatment Study Group (1995) Effects of oral alendronate on bone mineral density and the incidence of fracture in postmenopausal osteoporosis. New Engl J Med 333:1437–1443
Black DM, Cummings SR, Karpf DB, Cauley JA, Thompson DE, Nevitt MC, Bauer DC, Genant HK, Haskell WL, Marcus R, Ott SM, Torner JC, Quandt SA, Reiss TF, Ensrud KE, For the Fracture Intervention Trial Group (1996) Randomized trial of effect of alendronate on risk of fracture in women with existing vertebral fracture. Lancet 348:1535–1541
Harris ST, Watts NB, Genant HK, McKeever EF, Hoseyni MS, Axelrod DW, Miller PD, Vertebral Efficacy with Risedronate Therapy (VERT) study group (1999) Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: randomized controlled trial. JAMA 282:1344–1352
Matsumoto T, Hagino H, Shiraki M, Fukunaga M, Nakano T, Takaoka K, Morii H, Ohashi Y, Nakamura T (2009) Effect of daily oral minodronate on vertebral fractures in Japanese postmenopausal women with established osteoporosis: a randomized placebo-controlled double-blind study. Osteoporos Int 20:1429–1437
Tsugawa N, Shiraki M, Suhara Y, Kamao M, Ozaki R, Tanaka K, Okano T (2008) Low plasma phylloquinone concentration is associated with high incidence of vertebral fracture in Japanese women. J Bone Miner Metab 26:79–85
Kaneki M, Hodges SJ, Hosoi T, Fujiwara S, Lyons A, Crean SJ, Ishida N, Nakagawa M, Takeuchi M, Sano Y, Mizuno Y, Hoshino S, Miyao M, Inoue S, Horiki K, Shiraki M, Ouchi Y, Orimo H (2001) Japanese fermented soybean food as the major determinant of the large geographic difference in circulating levels of vitamin K2: possible implications for hip fracture risk. Nutrition 17:315–321
Cheung AM, Tile L, Lee Y, Tomlinson G, Hawker G, Scher J, Hu H, Vieth R, Thompson L, Jamal S, Josse R (2008) Vitamin K supplementation in postmenopausal women with osteopenia (ECKO trial): a randomized controlled trial. PLoS Med 14:e196
Iwamoto J, Takeda T, Ichimura S (2001) Effect of menatetrenone on bone mineral density and incidence of vertebral fractures in postmenopausal women with osteoporosis: a comparison with the effect of etidronate. J Orthop Sci 6:487–492
Shiraki M, Shiraki Y, Aoki C, Miura M (2000) Vitamin K2 (menatetrenone) effectively prevents fractures and sustains lumbar bone mineral density in osteoporosis. J Bone Miner Res 15:515–521
Inoue T, Fujita T, Kishimoto H, Makino T, Nakamura T, Nakamura T, Sato T, Yamazaki K (2009) Randomized controlled study on the prevention of osteoporotic fractures (OF study): a phase IV clinical study of 15-mg menatetrenone capsules. J Bone Miner Metab 27:66–75
Ishida Y, Kawai S (2004) Comparative efficacy of hormone replacement therapy, etidronate, calcitonin, alfacalcidol, and vitamin K in postmenopausal women with osteoporosis: The Yamaguchi Osteoporosis Prevention Study. Am J Med 117:549–555
Stevenson M, Lloyd-Jones M, Papaioannou D (2009) Vitamin K to prevent fractures in older women: systematic review and economic evaluation. Health Technol Assess 13:1–134
Fang Y, Hu C, Tao X, Wan Y, Tao F (2012) Effect of vitamin K on bone mineral density: a meta-analysis of randomized controlled trials. J Bone Miner Metab 30(1):60–68
Fukunaga M, Nakamura T, Shiraki M, Kuroda T, Ohta H, Hosoi T, Orimo H (2004) Absolute height reduction and percent height ratio of the vertebral body in incident fracture in Japanese women. J Bone Miner Metab 22:104–110
Kumamoto K, Nakamura T, Suzuki T, Gorai I, Fujinawa O, Ohta H, Shiraki M, Yoh K, Fujiwara S, Endo N, Matsumoto T (2010) Validation of the Japanese osteoporosis quality of life questionnaire. J Bone Miner Metab 28:1–7
Meurs JB, Dhonukshe-Rutten RA, Pluijm SM, van der Klift M, de Jonge R, Lindemans J, de Groot LC, Hofman A, Witteman JC, van Leeuwen JP, Breteler MM, Lips P, Pols HA, Uitterlinden AG (2004) Homocysteine levels and the risk of osteoporotic fracture. N Engl J Med 350:2033–2041
McLean RR, Jacques PF, Selhub J, Tucker KL, Samelson EJ, Broe KE, Hannan MT, Cupples LA, Kiel DP (2004) Homocysteine as a predictive factor for hip fracture in older persons. N Engl J Med 350:2042–2049
Shiraki M, Kuroda T, Shiraki Y, Tanaka S, Higuchi T, Saito M (2011) Urinary pentosidine and plasma homocysteine levels at baseline predict future fractures in osteoporosis patients under bisphosphonate treatment. J Bone Miner Metab 29:62–70
The Japanese version of National Cancer Institute–Common Toxicity Criteria version 2.0. Available at http://www.jcog.jp/doctor/tool/C_150_0011.pdf. Accessed 31 Jan 2012
Kushida K, Fukunaga M, Kishimoto H, Shiraki M, Itabashi A, Inoue T, Kaneda K, Morii H, Nawata H, Yamamoto K, Ohashi Y, Orimo H (2004) A comparison of incidences of vertebral fracture in Japanese patients with involutional osteoporosis treated with risedronate and etidronate: a randomized, double-masked trial. J Bone Miner Metab 22:469–478
Iwamoto J, Sato Y, Takeda T, Matsumoto H (2009) High-dose vitamin K supplementation reduces fracture incidence in postmenopausal women: a review of the literature. Nutr Res 29:221–228
Kushima Y, Ishii K, Shimizu K, Sasaki A, Yamamoto K, et al. (2007) Effect of combination therapy with risedronate and vitamin K2 for osteoporosis: Multicentre collaborative study. Preliminary report of a 1-year study. Osteoporos Jpn 15:234–237 (written in Japanese)
Dowd R, Recker RR, Heaney RP (2000) Study subjects and ordinary patients. Osteoporos Int 11:533–536
Okano T, Shimomura Y, Yamane M, Suhara Y, Kamao M, Sugiura M, Nakagawa K (2008) Conversion of phylloquinone (Vitamin K1) into menaquinone-4 (Vitamin K2) in mice: two possible routes for menaquinone-4 accumulation in cerebra of mice. J Biol Chem 283:11270–11279
Tabb MM, Sun A, Zhou C, Grün F, Errandi J, Romero K, Pham H, Inoue S, Mallick S, Lin M, Forman BM, Blumberg B (2003) Vitamin K2 regulation of bone homeostasis is mediated by the steroid and xenobiotic receptor SXR. J Biol Chem 278:43919–43927
Ichikawa T, Horie-Inoue K, Ikeda K, Blumberg B, Inoue S (2006) Steroid and xenobiotic receptor SXR mediates vitamin K2-activated transcription of extracellular matrix-related genes and collagen accumulation in osteoblastic cells. J Biol Chem 281:16927–16934
Tsugawa N, Shiraki M, Suhara Y, Kamao M, Tanaka K, Okano T (2006) Vitamin K status of healthy Japanese women: age-related vitamin K requirement for gamma-carboxylation of osteocalcin. Am J Clin Nutr 83:380–386
Shiraki M, Itabashi A (2009) Short-term menatetrenone therapy increases gamma-carboxylation of osteocalcin with a moderate increase of bone turnover in postmenopausal osteoporosis: a randomized prospective study. J Bone Miner Metab 27:333–340
Szulc P, Chapuy MC, Meunier PJ, Delmas PD (1993) Serum undercarboxylated osteocalcin is a marker of the risk of hip fracture in elderly women. J Clin Invest 91:1769–1774
Seibel MJ, Robins SP, Bilezikian JP (1997) Serum undercarboxylated osteocalcin and the risk of hip fracture. J Clin Endocrinol Metab 82:717–718
Vergnaud P, Garnero P, Meunier PJ, Bréart G, Kamihagi K, Delmas PD (1997) Undercarboxylated osteocalcin measured with a specific immunoassay predicts hip fracture in elderly women: the EPIDOS Study. J Clin Endocrinol Metab 82:719–724
Luukinen H, Käkönen SM, Pettersson K, Koski K, Laippala P, Lövgren T, Kivelä SL, Väänänen HK (2000) Strong prediction of fractures among older adults by the ratio of carboxylated to total serum osteocalcin. J Bone Miner Res 15:2473–2478
Tanaka S, Narusawa K, Onishi H, Miura M, Hijioka A, Kanazawa Y, Nishida S, Ikeda S, Nakamura T (2011) Lower osteocalcin and osteopontin contents of the femoral head in hip fracture patients than osteoarthritis patients. Osteoporos Int 22:587–597
Shiraki M, Yamazaki Y, Shiraki Y, Hosoi T, Tsugawa N, Okano T (2010) High level of serum undercarboxylated osteocalcin in patients with incident fractures during bisphosphonate treatment. J Bone Miner Metab 28:578–584
Matsumoto Y, Mikuni-Takagaki Y, Kozai Y, Miyagawa K, Naruse K, Wakao H, Kawamata R, Kashima I, Sakurai T (2009) Prior treatment with vitamin K(2) significantly improves the efficacy of risedronate. Osteoporos Int 20:1863–1872
Iwamoto J, Matsumoto H, Takeda T, Sato Y, Liu X, Yeh JK (2008) Effects of vitamin K(2) and risedronate on bone formation and resorption, osteocyte lacunar system, and porosity in the cortical bone of glucocorticoid-treated rats. Calcif Tissue Int 83:121–128
Kuroda T, Shiraki M, Tanaka S, Ohta H (2009) Contributions of 25-hydroxyvitamin D, co-morbidities and bone mass to mortality in Japanese postmenopausal women. Bone 44:168–172
Nevitt MC, Thompson DE, Black DM, Rubin SR, Ensrud K, Yates AJ, Cummings SR (2000) Effect of alendronate on limited-activity days and bed-disability days caused by back pain in postmenopausal women with existing vertebral fractures. Fracture Intervention Trial Research Group. Arch Intern Med 160:77–85
Orimo H, Nakamura T, Fukunaga M, Ohta H, Hosoi T, Uemura Y, Kuroda T, Miyakawa N, Ohashi Y, Shiraki M, A-TOP (Adequate Treatment of Osteoporosis) research group (2011) Effects of alendronate plus alfacalcidol in osteoporosis patients with a high risk of fracture: the Japanese Osteoporosis Intervention Trial (JOINT)-02. Curr Med Res Opin 27:1273–1284
Acknowledgments
The authors express thanks to the chairman (Dr. Rikushi Morita) and the members of the ethics committee—Ms. Mamiko Matsumura, Dr. Tetsuro Inoue, Dr. Isao Yoshimura, Dr. Mitsuyoshi Nakajima, Tooru Ebihara, R.Ph., Mr. Shinya Hattori and Mr. Kousaku Uchida. This JOINT study was sponsored by the Public Health Research Foundation. The authors would like to thank those who participated as clinical investigators in JOINT-03. The authors also thank the Japan Arteriosclerosis Prevention Fund, Teijin Pharma Limited, Asahi Kasei Corporation, Takeda Pharmaceutical Company Limited, GE Healthcare Japan, Toyo Medic Co., Ltd., MSD Co., Ltd., and Dainippon Sumitomo Pharma Co., Ltd., for their generous donations.
Conflict of interest
T.M. is an employee of the Public Health Research Foundation. The other authors declare that there is no conflict of interest associated with this manuscript.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Tanaka, S., Miyazaki, T., Uemura, Y. et al. Design of a randomized clinical trial of concurrent treatment with vitamin K2 and risedronate compared to risedronate alone in osteoporotic patients: Japanese Osteoporosis Intervention Trial-03 (JOINT-03). J Bone Miner Metab 32, 298–304 (2014). https://doi.org/10.1007/s00774-013-0491-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-013-0491-4