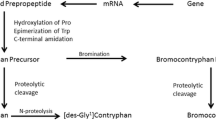
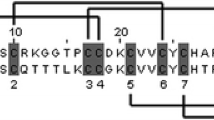
Abstract
In the venom of eusocial bee Lasioglossum laticeps, we identified a novel unique antimicrobial peptide named lasiocepsin consisting of 27 amino acid residues and two disulfide bridges. After identifying its primary structure, we synthesized lasiocepsin by solid-phase peptide synthesis using two different approaches for oxidative folding. The oxidative folding of fully deprotected linear peptide resulted in a mixture of three products differing in the pattern of disulfide bridges. Regioselective disulfide bond formation significantly improved the yield of desired product. The synthetic lasiocepsin possessed antimicrobial activity against both Gram-positive and -negative bacteria, antifungal activity against Candida albicans, and no hemolytic activity against human erythrocytes. We synthesized two lasiocepsin analogs cyclized through one native disulfide bridge in different positions and having the remaining two cysteines substituted by alanines. The analog cyclized through a Cys8–Cys25 disulfide bridge showed reduced antimicrobial activity compared to the native peptide while the second one (Cys17–Cys27) was almost inactive. Linear lasiocepsin having all four cysteine residues substituted by alanines or alkylated was also inactive. That was in contrast to the linear lasiocepsin with all four cysteine residues non-paired, which exhibited remarkable antimicrobial activity. The shortening of lasiocepsin by several amino acid residues either from the N- or C-terminal resulted in significant loss of antimicrobial activity. Study of Bacillus subtilis cells treated by lasiocepsin using transmission electron microscopy showed leakage of bacterial content mainly from the holes localized at the ends of the bacterial cells.






Similar content being viewed by others
References
Amiche M, Galanth C (2011) Dermaseptins as models for the elucidation of membrane-acting helical amphipathic antimicrobial peptides. Curr Pharm Biotechno 12:1184–1193
Čeřovský V, Slaninová J, Fučík V, Hulačová H, Borovičková L, Ježek R, Bednárová L (2008a) New potent antimicrobial peptides from the venom of Polistinae wasps and their analogs. Peptides 29:992–1003
Čeřovský V, Hovorka O, Cvačka J, Voburka Z, Bednárová L, Borovičková L, Slaninová J, Fučík V (2008b) Melectin: a novel antimicrobial peptide from the venom of the cleptoparasitic bee Melecta albifrons. Chem Bio Chem 9:2815–2821
Čeřovský V, Buděšínský M, Hovorka O, Cvačka J, Voburka Z, Slaninová J, Borovičková L, Fučík V, Bednárová L, Votruba I, Straka J (2009) Lasioglossins: three novel antimicrobial peptides from the venom of the eusocial bee Lasioglossum laticeps (Hymenoptera: Halictidae). Chem Bio Chem 10:2089–2099
Čeřovský V, Slaninová J, Fučík V, Monincová L, Bednárová L, Maloň P, Štokrová J (2011) Lucifensin, a novel insect defensin of medicinal maggots: synthesis and structural study. Chem Bio Chem 12:1352–1361
Chen Y, Mant CT, Farmer SW, Hancock REW, Vasil ML, Hodges RS (2005) Rational design of α-helical antimicrobial peptides with enhanced activities and specificity/therapeutic index. J Biol Chem 280:12316–12329
Dawson RM, Liu C-Q (2010) Disulphide bonds of the peptide protegrin-1 are not essential for antimicrobial activity and haemolytic activity. Int J Antimicrob Agents 36:579–580
Dennison SR, Whittaker M, Hartus F, Phoenix DA (2006) Anticancer α-helical peptides and structure/function relationships underpinning their interactions with tumor cell membranes. Curr Protein Peptide Sci 7:487–499
Epand RM, Epand RF (2009) Lipid domains in bacterial membranes and the action of antimicrobial agents. Biochem Biophys Acta 1788:289–294
Epand RM, Epand RF (2011) Bacterial membrane lipids in the action of antimicrobial targets. J Pept Sci 17:298–305
Fázio MA, Iliveira VX Jr, Bulet P, Miranda MTM, Daffre S, Miranda A (2006) Structure-activity relationship studies of gomesin: importance of the disulfide bridges for conformation, bioactivities, and serum stability. Biopolymers (Peptide Scince) 84:205–218
Giuliani A, Pirri G, Nicoletto SF (2007) Antimicrobial peptides: an overview of a promising class of therapeutics. Centr Eur J Biol 2:1–33
Huang Y, Huang J, Chen Y (2010) Alpha-helical cationic antimicrobial peptides: relationships of structure and function. Protein Cell 1:143–152
Jones DT (1999) Protein secondary structure prediction based on position-specific scoring matrices. J Mol Biol 292:195–202
Kim J-Y, Park S-C, Yoon M-Y, Hahm K-S, Park Y (2011) C-terminal amidation of PMAP-23: translocation to the inner membrane of gram-negative bacteria. Amino Acids 40:183–195
Klüver E, Schulz-Maronde S, Scheid S, Meyer B, Forssmann WG, Adermann K (2005) Structure–activity relation of human β-defensin 3: influence of disulfide bonds and cysteine substitution on antimicrobial activity and cytotoxicity. Biochemistry 44:9804–9816
Konno K, Rangel M, Oliveira JS, dos Santos Cabrera MP, Fontana R, Hirata IY et al (2007) Decoralin, a novel linear cationic α-helical peptide from the venom of the solitary eumenine wasp oreumenes decoratus. Peptides 28:2320–2327
Kuhn-Nentwig L (2003) Antimicrobial and cytolytic peptides of venomous arthropods. Cell Mol Life Sci 60:2651–2668
Kuzuhara T, Nakajima Y, Matsuyama K, Natori S (1990) Determination of the disulfide array in sapecin, and antibacterial peptide of Sarcophaga peprigrina (Flesh fly). J Biochem 107:514–518
Kwon M-Y, Hong S-Y, Lee K-H (1998) Structure–activity analysis of brevinin 1E amide, an antimicrobial peptide from Rana esculenta. Biochim Biophys Acta 1387:239–248
Labbé-Julié C, Granier C, Albericio F, Defendini M-L, Ceard B, Rochat H, Van Rietschoten J (1991) Binding and toxicity of apamin. Characterization of the active site. Eur J Biochem 196:639–645
Liu S, Zhou L, Li J, Suresh A, Verma C, Foo YH, Yap EPH, Tan DTH, Beuerman RW (2008) Linear analogues of human β-defensin 3: concepts for design of antimicrobial peptides with reduced cytotoxicity to mammalian cells. Chem Bio Chem 9:964–973
Mandal M, Jagannadham MV, Nagaraj R (2002) Antimicrobial activities and conformations of bovine β-defensin BNBD-12 and analogs: structural and disulfide bridge requirements for activity. Peptides 23:413–418
Monincová L, Buděšínský M, Slaninová J, Hovorka O, Cvačka J, Voburka Z, Fučík V, Borovičková L, Bednárová L, Straka J, Čeřovský V (2010) Novel antimicrobial peptides from the venom of the eusocial bee Halictus sexcinctus (Hymenoptera: Halictidae) and their analogs. Amino Acids 39:763–775
Oren Z, Shai Y (1998) Mode of action of linear amphipathic α-helical antimicrobial peptides. Biopolymers (Peptide Science) 47:451–463
Oyston PCF, Fox MA, Richards SJ, Clark GC (2009) Novel peptide therapeutics for treatment of infections. J Med Microb 58:977–987
Rivas L, Luque-Ortega JR, Andreu D (2009) Amphibian antimicrobial peptides and protozoa: lessons from parasites. Biochim Biophys Acta 1788:1570–1581
Schroeder BO, Wu Z, Nuding S, Groscurth S, Marcinowski M, Beisner J, Buchner J, Schaller M, Stange EF, Wehkamp J (2011) Reduction of disulphide bonds unmasks potent antimicrobial activity of human β-defensin 1. Nature 469:419–423
Slaninová J, Putnová H, Borovičková L, Šácha P, Čeřovská V, Monincová L, Fučík V (2011) The antifungal effect of peptides from the hymenoptera venom and their analogs. Centr Eur J Biol 6:150–159
Toke O (2005) Antimicrobial peptides: new candidates in the fight against bacterial infections. Biopolymers (Peptide Science) 80:717–735
Tossi A, Sandri L, Giangaspero A (2000) Amphipathic, α-helical antimicrobial peptides. Biopolymers (Peptide Science) 55:4–30
Varkey J, Nagaraj R (2005) Antibacterial activity of human neutrophil defensin HNP-1 analogs without cysteines. Antimicrob Agents Chemother 49:4561–4566
Vasileiou Z, Barlos KK, Gatos D, Adermann K, Deraison C, Barlos K (2010) Synthesis of the proteinase inhibitor LEKTI domain 6 by the fragment condensation method and regioselective disulfide bond formation. Biopolymers 94:339–349
Wang G, Li X, Wang Z (2009) APD2: the updated antimicrobial peptide database and its application in peptide design. Nucleic Acids Res 37:D933–D937
Whitmore L, Wallace BA (2008) Protein secondary structure analyses from circular dichroism spectroscopy: methods and reference databases. Biopolymers 89:392–400
Wimley WC, Hristova K (2011) Antimicrobial peptides: successes, challenges and unanswered questions. J Membrane Biol 239:27–34
Yeaman MR, Yount NY (2003) Mechanisms of antimicrobial peptide action and resistance. Pharm Rev 55:27–55
Yeung ATY, Gellatly SL, Hancock REW (2011) Multifunctional cationic host defence peptides and their clinical applications. Cell Mol Life Sci 68:2161–2176
Zaiou M (2007) Multifunctional antimicrobial peptides: therapeutic targets in several human diseases. J Mol Med 85:317–329
Acknowledgments
This work was supported by the Czech Science Foundation, Grants Nos. 203/08/0536 and P205/10/1276, and by Research Project No. Z40550506 of the Institute of Organic Chemistry and Biochemistry, Academy of Sciences of the Czech Republic. We thank our technical assistant Mrs. Hana Hulačová for the help with peptide synthesis. Lenka Monincová thanks the Ministry of Education of the Czech Republic for a stipend and for additional financial support from Specific University Research Project No. 33779266 awarded by Charles University Prague. We also thank Gale A. Kirking at English Editorial Services, s.r.o. for assistance with the English.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Monincová, L., Slaninová, J., Fučík, V. et al. Lasiocepsin, a novel cyclic antimicrobial peptide from the venom of eusocial bee Lasioglossum laticeps (Hymenoptera: Halictidae). Amino Acids 43, 751–761 (2012). https://doi.org/10.1007/s00726-011-1125-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-011-1125-6