Abstract
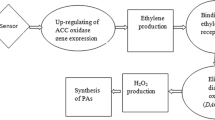
Pollination of pummelo (Citrus grandis L. Osbeck) pistils has been studied in planta by adding compatible and self-incompatible (SI) pollen to the stigma surface. The pollen germination has been monitored inside the pistil by fluorescent microscopy showing SI altered morphologies with irregular depositions of callose in the tube walls, and heavy callose depositions in enlarged tips. The polyamine (PA) content as free, perchloric acid (PCA)-soluble and -insoluble fractions and transglutaminase (TGase) activity have been analyzed in order to deepen their possible involvement in the progamic phase of plant reproduction. The conjugated PAs in PCA-soluble fraction were definitely higher than the free and the PCA-insoluble forms, in both compatible and SI pollinated pistils. In pistils, pollination caused an early decrease of free PAs and increase of the bound forms. The SI pollination, showed highest values of PCA-soluble and -insoluble PAs with a maximum in concomitance with the pollen tube arrest. As TGase mediates some of the effects of PAs by covalently binding them to proteins, its activity, never checked before in Citrus, was examined with two different assays. In addition, the presence of glutamyl-PAs confirmed the enzyme assay data and excluded the possibility of a misinterpretation. The SI pollination caused an increase in TGase activity, whereas the compatible pollination caused its decrease. Similarly to bound PAs, the glutamyl-PAs and the enzyme activity peaked in the SI pollinated pistils in concomitance with the observed block of the pollen tube growth, suggesting an involvement of TGase in SI response.





Similar content being viewed by others
Abbreviations
- BCA:
-
Bicinchoninic acid
- BSA:
-
Bovine serum albumin
- DMC:
-
N′,N′-dimethyl casein
- DTT:
-
Dithiothreitol
- EDTA:
-
Ethylene diamine tetraacetic acid
- EGTA:
-
Ethylene glycol tetraacetic acid
- GSI:
-
Gametophytic self-incompatible
- HPLC:
-
High-performance liquid chromatography
- PAs:
-
Polyamines
- PBS:
-
Phosphate-buffered saline
- PMSF:
-
Phenyl methyl sulfonyl fluoride
- PCA:
-
Perchloric acid
- PCD:
-
Programmed cell death
- PU:
-
Putrescine
- SI:
-
Self-incompatible
- SD:
-
Spermidine
- SM:
-
Spermine
- SSI:
-
Sporophytic self-incompatible
- TCA:
-
Trichloroacetic acid
- TGase:
-
Transglutaminase
- tTGase:
-
Tissue transglutaminase
- UP:
-
Unpollinated
References
Antognoni F, Bagni N (2008) Bis(guanylhydrazones) negatively affect in vitro germination of kiwifruit pollen and alter the endogenous polyamine pool. Plant Biol 10:334–341
Bagni N, Adamo P, Serafini-Fracassini D, Villanueva VR (1981) RNA, Proteins and polyamines during tube growth in germinating apple pollen. Plant Physiol 68:727–730
Beninati S, Bergamini CM, Piacentini M (2009) An overview of the first 50 years of transglutaminase research. Amino Acids 36:591–598
Biasi R, Falasca G, Speranza A, De Stradis A, Scoccianti V, Franceschetti M, Bagni N, Altamura MM (2001) Biochemical and ultrastructural features related to male sterility in the dioecious species Actinidia deliciosa. Plant Physiol Biochem 39:395–406
Bokern M, Witte L, Wray V, Nimtz M, Meurer-Grimes B (1995) Trisubstituted hydroxycinnamic acid Spds from Quercus dentata pollen. Phytochem 39:1371–1375
Brown RE, Jarvis KL, Hyland KJ (1989) Protein measurement using bicinchoninic acid: elimination of interfering substances. Anal Biochem 180:136–139
Cai G, Romagnoli S, Moscatelli A, Ovidi E, Gambellini G, Tiezzi A, Cresti M (1997) Identification and characterization of a novel microtubule-based motor associated with membranous organelles in tobacco pollen tubes. Plant Cell 12:1719–1736
Capell T, Claparols I, Del Duca S, Bassie L, Miro B, Rodriguez-Montesinos J, Christou P, Serafini-Fracassini D (2004) Producing transglutaminases by molecular farming in plants: minireview article. Amino Acids 26:419–423
Cheung A, Wu H (2008) Structural and signaling networks for the polar cell growth machinery in pollen tubes. Annu Rev Plant Biol 59:547–572
Chibi F, Matilla A, Angosto T, Garrido D (1994) Changes in polyamine synthesis during anther development and pollen germination in tobacco (Nicotiana tabacum). Physiol Plant 92:61–68
Del Duca S, Bregoli AM, Bergamini C, Serafini-Fracassini D (1997) Transglutaminase-catalyzed modification of cytoskeletal proteins by polyamines during the germination of Malus domestica pollen. Sex Plant Reprod 10:89–95
Del Duca S, Serafini-Fracassini D, Bonner PLR, Cresti M, Cai G (2009) Effects of post-translational modifications catalyzed by pollen transglutaminase on the functional properties of microtubules and actin filaments. Biochem J 418:651–664
Del Duca S, Cai G, Di Sandro A, Serafini-Fracassini D (2010) Compatible and self-incompatible pollination in Pyrus communis displays different polyamine levels and transglutaminase activity. Amino Acids 38:659–667
Della Mea M, Serafini-Fracassini D, Del Duca S (2007a) Programmed cell death: similarities and differences in animals and plants. A flower paradigm. Amino Acids 33:395–404
Della Mea M, De Filippis F, Genovesi V, Serafini-Fracassini D, Del Duca S (2007b) The acropetal wave of developmental cell death (DCD) of Nicotiana tabacum corolla is preceded by activation of transglutaminase in different cell compartments. Plant Physiol 144:1–13
Deng ZN, La Malfa S, Xie YM, Xiong XX, Gentile A (2007) Identification and evaluation of chloroplast uni- and trinucleotide sequence repeats in citrus. Scientia Hort 111:186–192
Di Sandro A, Del Duca S, Verderio E, Hargreaves A, Scarpellini A, Cai G, Cresti M, Faleri C, Iorio RA, Hirose S, Furutani Y, Coutts IGC, Griffin M, Bonner PLR, Serafini-Fracassini D (2010) An extracellular transglutaminase is required for apple pollen tube growth. Biochem J 429:261–271
Distefano G, Caruso M, La Malfa S, Gentile A, Tribulato E (2009) Histological and molecular analysis of pollen–pistil interaction in Clementine. Plant Cell Rep 28:1439–1451
Fellenberg C, Böttcher C, Vogt T (2009) Phenylpropanoid polyamine conjugate biosynthesis in Arabidopsis thaliana flower buds. Phytochem 70:1392–1400
Folk JE, Park MH, Chung SI, Schrode J, Lester EP, Cooper HL (1980) Polyamines as physiological substrates for transglutaminases. J Biol Chem 255:3695–3700
Foreman J, Demidchik V, Bothwell JH, Mylona P, Miedema H, Torres MA, Linstead P, Costa S, Brownlee C, Jones JD et al (2003) Reactive oxygen species produced by NADPH oxidase regulate plant cell growth. Nature 422:442–446
Galston AW, Kaur-Sawhney R (1990) Polyamines in plant physiology. Plant Physiol 94:406–410
Grienenberger E, Besseau S, Geoffroy P, Debayle D, Heintz D, Lapierre C, Pollet B, Heitz T, Legrand M (2009) A BAHD acyltransferase is expressed in the tapetum of Arabidopsis anthers and is involved in the synthesis of hydroxycinnamoyl spermidines. Plant J 58:246–259
Griffin M, Casadio R, Bergamini CM (2002) Transglutaminases: nature’s biological glues. Biochem J 368:377–396
Ha HC, Sirisoma NS, Kuppusamy P, Zweier JL, Woster PM, Casero RAJ (1998) The natural polyamine spermine functions directly as a free radical scavenger. Proc Natl Acad Sci USA 95:11140–11145
Lam TBT, Iiyama K, Stone B (1992) Cinnamic acid bridges between cell wall polymers in wheat and Phalaris internodes. Phytochem 31:1179–1183
Lilley GR, Skill J, Griffin M, Bonner PL (1998) Detection of Ca2+-dependent transglutaminase activity in root and leaf tissue of monocotyledonous and dicotyledonous plants. Plant Physiol 117:1115–1123
Lorand L, Graham RM (2003) Transglutaminases: crosslinking enzymes with pleiotropic functions. Nat Rev Mol Cell Biol 4:140–156
Martin-Tanguy J, Perdrizet E, Prevost J, Martin C (1982) The distribution of hydroxycinnamic acid amides in fertile and cytoplasmic male sterile lines of maize. Phytochem 21:1939–1945
McClure BA, Franklin-Tong V (2006) Gametophytic self-incompatibility: understanding the cellular mechanisms involved in “self” pollen tube inhibition. Planta 224:233–245
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410
Neill S, Desikan R, Hancock J (2002) Hydrogen peroxide signalling. Curr Opin Plant Biol 5:388–395
Pohjanpelto P, Virtanen I, Holtta E (1981) Polyamine starvation causes disappearance of actin filaments and microtubules in polyamine-auxotrophic cells. Nature 293:475–477
Potocký M, Jones MA, Bezvoda R, Smirnoff N, Žárský V (2007) Reactive oxygen species produced by NADPH oxidase are involved in pollen tube growth. New Phytol 174:742–751
Poulter NS, Vatovec S, Franklin-Tong VE (2008) Microtubules are a target for self-incompatibility signaling in Papaver pollen. Plant Physiol 146:1358–1367
Scaramagli S, Bueno M, Torrigiani P, Altamura MM, Capitani F, Bagni N (1995) Morphogenesis in cultured thin layers and pith explants of tobacco. II. Early hormone modulated polyamine biosynthesis. J Plant Physiol 147:113–117
Serafini-Fracassini D, Della Mea M, Tasco G, Casadio R, Del Duca S (2009) Plant and animal transglutaminases: do similar functions imply similar structures? Amino Acids 36:643–657
Shi J, Fu XZ, Peng T, Huang XS, Fan QJ, Liu JH (2010) Spermine pretreatment confers dehydration tolerance of citrus in vitro plants via modulation of antioxidative capacity and stomatal response. Tree Physiol 30:914–922
Siepaio MP, Meunier JF (1995) Diamine oxidase and transglutaminase activities in white lupin seedlings with respect to crosslinking of proteins. J Agric Food Chem 43:1151–1156
Soost RK (1965) Incompatibility alleles in the genus Citrus. Proc Am Soc Hortic Sci 87:176–180
Speranza A, Calzoni GL, Bagni N (1984) Evidence for a polyamine-mediated control of ribonuclease activity in germinating pollen. Physiol Veg 22:323–331
Thomas SG, Franklin-Tong VE (2004) Self-incompatibility triggers programmed cell death in Papaver pollen. Nature 429:305–309
Thomas SG, Huang S, Li S, Staiger CJ, Franklin-Tong VE (2006) Actin depolymerization is sufficient to induce programmed cell death in self-incompatible pollen. J Cell Biol 174:221–229
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants. Protective role of exogenous polyamines. Plant Sci 151:59–66
Waffenschmidt S, Kusch T, Woessner JP (1999) A transglutaminase immunologically related to tissue transglutaminase catalyses cross-linking of cell wall proteins in Chlamydomonas reinhardtii. Plant Physiol 121:1003–1015
Wang CL, Wu J, Xu GH, Gao YB, Chen G, Wu JY, Wu HQ, Zhang SL (2010) S-RNase disrupts tip-localized reactive oxygen species and induces nuclear DNA degradation in incompatible pollen tubes of Pyrus pyrifolia. J Cell Sci 123:4301–4309
Wheeler MJ, de Graaf BH, Hadjiosif N, Perry RM, Poulter NS, Osman K, Vatovec S, Harper A, Franklin FC, Franklin-Tong VE (2009) Identification of the pollen self-incompatibility determinant in Papaver rhoeas. Nature 459:992–995
Acknowledgments
Thanks are due to the Italian Ministero dell’Università that supported this work by a grant PRIN 2007 to Dipartimento Biologia Evoluzionistica Sperimentale, Università di Bologna (http://prin.miur.it/) and Dipartimento di Scienze delle Produzioni Agrarie e Alimentari, Università di Catania. Thanks to I. Menghetti for work she made during the training in the Dip. Biologia Evoluzionistica Sperimentale and to Nicodemo Mele for his help in figure preparation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gentile, A., Antognoni, F., Iorio, R.A. et al. Polyamines and transglutaminase activity are involved in compatible and self-incompatible pollination of Citrus grandis . Amino Acids 42, 1025–1035 (2012). https://doi.org/10.1007/s00726-011-1017-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-011-1017-9