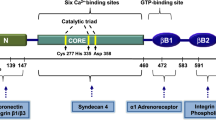
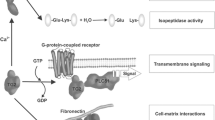
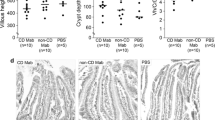
Abstract
Coeliac disease is an autoimmune-mediated disorder with both innate and adaptive immune components. The disease is triggered by dietary gluten, which provokes the development of a massive immune reaction leading to the destruction of the small-intestinal mucosal morphology and intestinal dysfunction. Besides the typical small-bowel symptoms extraintestinal manifestations may also arise in a subset of coeliac disease patients. In addition, gluten evokes the production of antibodies mainly targeting deamidated gluten peptides or transglutaminase 2. Although coeliac disease has traditionally been regarded as a T cell-mediated disorder, this review discusses the role of the gluten-induced disease-specific anti-transglutaminase 2-autoantibodies in the pathogenesis of the disease.

Similar content being viewed by others
References
Aeschlimann D, Aeschlimann P, Strigun A, Woodroofe N, Hadjivassiliou M (2007) TG6-specific antibodies in sera from patients with celiac disease: implications for extraintestinal disease manifestations. In: Abstract book of ninth international conference on transglutaminases and protein cross linking
Akimov SS, Krylov D, Fleischman LF, Belkin AM (2000) Tissue transglutaminase is an integrin-binding adhesion coreceptor for fibronectin. J Cell Biol 148:825–838
Ankelo M, Kleimola V, Simell S, Simell O, Knip M, Jokisalo E, Tarkia M, Westerlund A, He Q, Viander M, Ilonen J, Hinkkanen AE (2007) Antibody responses to deamidated gliadin peptide show high specificity and parallel antibodies to tissue transglutaminase in developing coeliac disease. Clin Exp Immunol 150:285–293
Aoki-Ota M, Tsunoda K, Ota T, Iwasaki T, Koyasu S, Amagai M, Nishikawa T (2004) A mouse model of pemphigus vulgaris by adoptive transfer of naïve splenocytes from desmoglein 3 knockout mice. Brit J Dermatol 151:346–354
Balklava Z, Verderio E, Collighan R, Gross S, Adams J, Griffin M (2002) Analysis of tissue transglutaminase function in the migration of swiss 3T3 fibroblasts: The active-state conformation of the enzyme does not affect cell motility but is important for its secretion. J Biol Chem 277:16567–16575
Barone MV, Caputo I, Ribecco MT, Maglio M, Marzari R, Sblattero D, Troncone R, Auricchio S, Esposito C (2007) Humoral immune response to tissue transglutaminase is related to epithelial cell proliferation in celiac disease. Gastroenterology 132:1245–1253
Beissbarth T, Tye-Din JA, Smyth GK, Speed TP, Anderson RP (2005) A systematic approach for comprehensive T-cell epitope discovery using peptide libraries. Bioinformatics 21:29–37
Bister V, Kolho KL, Karikoski R, Westerholm-Ormio M, Savilahti E, Saarialho-Kere U (2005) Metalloelastase (MMP-12) is upregulated in the gut of pediatric patients with potential celiac disease and in type 1 diabetes. Scand J Gastroenterol 40:1413–1422
Boscolo S, Sarich A, Lorenzon A, Passoni M, Rui V, Stebel M, Sblattero D, Marzari R, Hadjivassiliou M, Tongiorgi E (2007) Gluten ataxia: passive transfer in a mouse model. Ann N Y Acad Sci 1107:319–328
Byrne G, Ryan F, Jackson J, Feighery C, Kelly J (2007) Mutagenesis of the catalytic triad of tissue transglutaminase abrogates coeliac disease serum IgA autoantibody binding. Gut 56:336–341
Cervio E, Volta U, Verri M, Boschi F, Pastoris O, Granito A, Barbara G, Parisi C, Felicani C, Tonini M, De Giorgio R (2007) Sera of patients with celiac disease and neurologic disorders evoke a mitochondrial-dependent apoptosis in vitro. Gastroenterology 133:195–206
Ciccocioppo R, Di Sabatino A, Bauer M, Della Riccia DN, Bizzini F, Biagi F, Cifone MG, Corazza GR, Schuppan D (2005) Matrix metalloproteinase pattern in celiac duodenal mucosa. Lab Invest 85:397–407
Collin P, Pirttilä T, Nurmikko T, Somer H, Erilä T, Keyriläinen O (1991) Celiac disease, brain atrophy, and dementia. Neurology 41:372–375
Collin P, Huhtala H, Virta L, Kekkonen L, Reunala T (2007) Diagnosis of celiac disease in clinical practice: physician’s alertness to the condition essential. J Clin Gastroenterol 41:152–156
Cooke WT, Holmes GKT (1984) Coeliac Disease. Churchill Livingstone, New York
Dieterich W, Ehnis T, Bauer M, Donner P, Volta U, Riecken EO, Schuppan D (1997) Identification of tissue transglutaminase as the autoantigen of celiac disease. Nat Med 3:797–801
Dieterich W, Trapp D, Esslinger B, Leidenberger M, Piper J, Hahn E, Schuppan D (2003) Autoantibodies of patients with coeliac disease are insufficient to block tissue transglutaminase activity. Gut 52:1562–1566
Dieterich W, Esslinger B, Trapp D, Hahn E, Huff T, Seilmeier W, Wieser H, Schuppan D (2006) Cross linking to tissue transglutaminase and collagen favours gliadin toxicity in coeliac disease. Gut 55:478–484
Esposito C, Paparo F, Caputo I, Rossi M, Maglio M, Sblattero D, Not T, Porta R, Auricchio S, Marzari R, Troncone R (2002) Anti-tissue transglutaminase antibodies from coeliac patients inhibit transglutaminase activity both in vitro and in situ. Gut 51:177–181
Fesus L, Piacentini M (2002) Transglutaminase 2: an enigmatic enzyme with diverse functions. Trends Biochem Sci 27:534–539
Freeman HJ (2004) Adult celiac disease and the severe “flat” small bowel biopsy lesion. Dig Dis Sci 49:535–545
Gianfrani C, Auricchio S, Troncone R (2005) Adaptive and innate immune responses in celiac disease. Immunol Lett 99:141–145
Hadjivassiliou M, Gibson A, Davies-Jones GA, Lobo AJ, Stephenson TJ, Milford-Ward A (1996) Does cryptic gluten sensitivity play a part in neurological illness? Lancet 347:369–371
Hadjivassiliou M, Mäki M, Sanders DS, Williamson CA, Grünewald RA, Woodroofe NM, Korponay-Szabó IR (2006) Autoantibody targeting of brain and intestinal transglutaminase in gluten ataxia. Neurology 66:373–377
Halttunen T (2000) Biological functions of coeliac disease autoantibodies. Tampere University Press, Tampere
Halttunen T, Mäki M (1999) Serum immunoglobulin A from patients with celiac disease inhibits human T84 intestinal crypt epithelial cell differentiation. Gastroenterology 116:566–572
Haroon ZA, Hettasch JM, Lai TS, Dewhirst MW, Greenberg CS (1999) Tissue transglutaminase is expressed, active, and directly involved in rat dermal wound healing and angiogenesis. FASEB J 13:1787–1795
Hausch F, Shan L, Santiago NA, Gray GM, Khosla C (2002) Intestinal digestive resistance of immunodominant gliadin peptides. Am J Physiol Gastrointest Liver Physiol 283:G996–G1003
Hue S, Mention JJ, Monteiro RC, Zhang S, Cellier C, Schmitz J, Verkarre V, Fodil N, Bahram S, Cerf-Bensussan N, Caillat-Zucman S (2004) A direct role for NKG2D/MICA interaction in villous atrophy during celiac disease. Immunity 21:367–377
Jabri B, Sollid LM (2006) Mechanisms of disease: Immunopathogenesis of celiac disease. Nat Clin Pract Gastroenterol Hepatol 3:516–525
Jelinkova L, Tuckova L, Cinova J, Flegelova Z, Tlaskalova-Hogenova H (2004) Gliadin stimulates human monocytes to production of IL-8 and TNF-alpha through a mechanism involving NF-kappaB. FEBS Lett 571:81–85
Jones RA, Kotsakis P, Johnson TS, Chau DY, Ali S, Melino G, Griffin M (2006) Matrix changes induced by transglutaminase 2 lead to inhibition of angiogenesis and tumor growth. Cell Death Differ 13:1442–1453
Juuti-Uusitalo K, Mäki M, Kainulainen H, Isola J, Kaukinen K (2007) Gluten affects epithelial differentiation-associated genes in small intestinal mucosa of coeliac patients. Clin Exp Immunol 150:294–305
Kaukinen K, Peräaho M, Collin P, Partanen J, Woolley N, Kaartinen T, Nuutinen T, Halttunen T, Mäki M, Korponay-Szabo I (2005) Small-bowel mucosal transglutaminase 2-specific IgA deposits in coeliac disease without villous atrophy: a prospective and randomized clinical study. Scand J Gastroenterol 40:564–572
Kaukinen K, Collin P, Laurila K, Kaartinen T, Partanen J, Mäki M (2007) Resurrection of gliadin antibodies in coeliac disease. Deamidated gliadin peptide antibody test provides additional diagnostic benefit. Scand J Gastroenterol 42:1428–1433
Kiraly R, Vecsei Z, Demenyi T, Korponay-Szabo IR, Fesus L (2006) Coeliac autoantibodies can enhance transamidating and inhibit GTPase activity of tissue transglutaminase: Dependence on reaction environment and enzyme fitness. J Autoimmun 26:278–287
Korponay-Szabo IR, Halttunen T, Szalai Z, Laurila K, Király R, Kovács JB, Fésüs L, Mäki M (2004) In vivo targeting of intestinal and extraintestinal transglutaminase 2 by coeliac autoantibodies. Gut 53:641–648
Korponay-Szabo IR, Vecsei Z, Kiraly R, Dahlbom I, Chirdo F, Nemes E, Fesus L, Mäki M (2008) Deamidated gliadin peptides form epitopes that transglutaminase antibodies recognize. J Ped Gastroenterol Nutr 46:253–261
Lundin KE, Scott H, Hansen T, Paulsen G, Halstensen TS, Fausa O, Thorsby E, Sollid LM (1993) Gliadin-specific, HLA-DQ(alpha 1*0501, beta 1*0201) restricted T cells isolated from the small intestinal mucosa of celiac disease patients. J Exp Med 178:187–196
Luostarinen L, Pirttilä T, Collin P (1999) Coeliac disease presenting with neurological disorders. Eur Neurol 42:132–135
Maiuri L, Ciacci C, Auricchio S, Brown V, Quaratino S, Londei M (2000) Interleukin 15 mediates epithelial changes in celiac disease. Gastroenterology 119:996–1006
Maiuri L, Ciacci C, Ricciardelli I, Vacca L, Raia V, Auricchio S, Picard J, Osman M, Quaratino S, Londei M (2003) Association between innate response to gliadin and activation of pathogenic T cells in coeliac disease. Lancet 362:30–37
Marzari R, Sblattero D, Florian F, Tongiorgi E, Not T, Tommasini A, Ventura A, Bradbury A (2001) Molecular dissection of the tissue transglutaminase autoantibody response in celiac disease. J Immunol 166:4170–4176
Molberg O, Kett K, Scott H, Thorsby E, Sollid LM, Lundin KE (1997) Gliadin specific, HLA DQ2-restricted T cells are commonly found in small intestinal biopsies from coeliac disease patients, but not from controls. Scand J Immunol 46:103–109
Molberg O, Mcadam SN, Korner R, Quarsten H, Kristiansen C, Madsen L, Fugger L, Scott H, Norén O, Roepstorff P, Lundin KE, Sjöström H, Sollid LM (1998) Tissue transglutaminase selectively modifies gliadin peptides that are recognized by gut-derived T cells in celiac disease. Nat Med 4:713–717
Mustalahti K, Collin P, Sievanen H, Salmi J, Mäki M (1999) Osteopenia in patients with clinically silent coeliac disease warrants screening. Lancet 354:744–745
Myrsky E, Kaukinen K, Syrjänen M, Korponay-Szabo IR, Mäki M, Lindfors K (2008) Coeliac disease-specific autoantibodies targeted against transglutaminase 2 disturb angiogenesis. Clin Exp Immunol 152:111–119
Mäki M (1994) Autoantibodies as markers of autoimmunity in coeliac disease pathogenesis. In: Feighery C, O’Farrelly C (eds) Gastrointestinal immunology and gluten-sensitive disease. Proceedings of the Sixth International Symposium on coeliac disease held at Trinity College, Dublin in July 1992. Oak Tree Press, Dublin
Niveloni S, Sugai E, Cabanne A, Vazquez H, Argonz J, Smecuol E, Moreno ML, Nachman F, Mazure R, Kogan Z, Gomez JC, Mauriño E, Bai JC (2007) Antibodies against synthetic deamidated gliadin peptides as predictors of celiac disease: Prospective assessment in an adult population with a high pretest probability of disease. Clin Chem 53:2186–2192
Pinkas DM, Strop P, Brunger AT, Khosla C (2007) Transglutaminase 2 undergoes a large conformational change upon activation. PLoS Biol 5:e327
Saalman R, Wold AE, Dahlgren UI, Fallstrom SP, Hanson LA, Ahlstedt S (1998) Antibody-dependent cell-mediated cytotoxicity to gliadin-coated cells with sera from children with coeliac disease. Scand J Immunol 47:37–42
Salmi TT, Collin P, Korponay-Szabo IR, Laurila K, Partanen J, Huhtala H, Király R, Lorand L, Reunala T, Mäki M, Kaukinen K (2006) Endomysial antibody-negative coeliac disease: clinical characteristics and intestinal autoantibody deposits. Gut 55:1746–1753
Sardy M, Karpati S, Merkl B, Paulsson M, Smyth N (2002) Epidermal transglutaminase (TGase 3) is the autoantigen of dermatitis herpetiformis. J Exp Med 195:747–757
Shan L, Molberg O, Parrot I, Hausch F, Filiz F, Gray GM, Sollid LM, Khosla C (2002) Structural basis for gluten intolerance in celiac sprue. Science 297:2275–2279
Shaoul R, Lerner A (2007) Associated autoantibodies in celiac disease. Autoimmun Rev 6:559–565
Skovbjerg H, Koch C, Anthonsen D, Sjöström H (2004) Deamidation and cross-linking of gliadin peptides by transglutaminases and the relation to celiac disease. Biochim Biophys Acta 1690:220–230
Smecuol E, Bai JC, Vazquez H, Kogan Z, Cabanne A, Niveloni S, Pedreira S, Boerr L, Mauriño E, Meddings JB (1997) Gastrointestinal permeability in celiac disease. Gastroenterology 112:1129–1136
Sollid LM (2002) Coeliac disease: dissecting a complex inflammatory disorder. Nat Rev Immunol 2:647–655
Sollid LM, Molberg O, McAdam S, Lundin KE (1997) Autoantibodies in coeliac disease: tissue transglutaminase-guilt by association? Gut 41:851–852
Vincent A, Willcox N, Hill M, Curnow J, MacLennan C, Beeson D (1998) Determinant spreading and immune responses to acetylcholine receptors in myasthenia gravis. Immunol Rev 164:157–168
Visakorpi JK, Mäki M (1994) Changing clinical features of coeliac disease. Acta Paediatr Suppl 83:10–13
Volta U, De Giorgio R, Petrolini N, Stangbellini V, Barbara G, Granito A, De Ponti F, Corinaldesi R, Bianchi FB (2002) Clinical findings and anti-neuronal antibodies in coeliac disease with neurological disorders. Scand J Gastroenterol 37:1276–1281
Zanoni G, Navone R, Lunardi C, Tridente G, Bason C, Sivori S, Beri R, Dolcino M, Valletta E, Corrocher R, Puccetti A (2006) In celiac disease, a subset of autoantibodies against transglutaminase binds toll-like receptor 4 and induces activation of monocytes. PLoS Med 3:e358
Acknowledgments
The Coeliac Disease Study Group has been financially supported by the Research Council for Health, The Academy of Finland, The Pediatric Research Foundation, the Competitive Research Funding of the Pirkanmaa Hospital District, the Yrjö Jahnsson Foundation, the Finnish Medical Foundation, the Research Fund of the Finnish Coeliac Society and the European Commission (contract number MRTN-CT-2006-036032).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lindfors, K., Kaukinen, K. & Mäki, M. A role for anti-transglutaminase 2 autoantibodies in the pathogenesis of coeliac disease?. Amino Acids 36, 685–691 (2009). https://doi.org/10.1007/s00726-008-0127-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-008-0127-5