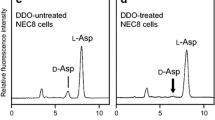
Summary.
The cDNA encoding D-aspartate oxidase (DASPO) was cloned from mouse kidney RNA by RT–PCR. Sequence analysis showed that it contained a 1023-bp open reading frame encoding a protein of 341 amino acid residues. The protein was expressed in Escherichia coli with or without an N-terminal His-tag and had functional DASPO activity that was highly specific for D-aspartate and N-methyl-D-aspartate. To investigate the roles of the Arg-216 and Arg-237 residues of the mouse DASPO (mDASPO), we generated clones with several single amino acid substitutions of these residues in an N-terminally His-tagged mDASPO. These substitutions significantly reduced the activity of the recombinant enzyme against acidic D-amino acids and did not confer any additional specificity to other amino acids. These results suggest that the Arg-216 and Arg-237 residues of mDASPO are catalytically important for full enzyme activity.
Similar content being viewed by others
Abbreviations
- D-Ala:
-
D-alanine
- D-Asn:
-
D-asparagine
- D-Asp:
-
D-aspartate
- BSA:
-
bovine serum albumin
- DAAO:
-
D-amino acid oxidase
- DASPO:
-
D-aspartate oxidase
- E. coli :
-
Escherichia coli
- FAD:
-
flavin adenine dinucleotide
- FMN:
-
flavin mononucleotide
- D-Glu:
-
D-glutamate
- His–mDASPO:
-
N-terminally His-tagged mouse D-aspartate oxidase
- IPTG:
-
isopropyl-β-D-thiogalactopyranoside
- mDASPO:
-
mouse D-aspartate oxidase
- D-Met:
-
D-methionine
- NMDA:
-
N-methyl-D-aspartate
- NMLA:
-
N-methyl-L-aspartate
- D-Pro:
-
D-proline
- SDS-PAGE:
-
SDS-polyacrylamide gel electrophoresis
References
L Amery C Brees M Baes C Setoyama R Miura GP Mannaerts PP Van Veldhoven (1998) ArticleTitleC-terminal tripeptide Ser-Asn-Leu (SNL) of human D-aspartate oxidase is a functional peroxisome-targeting signal Biochem J 336 367–371 Occurrence Handle9820813 Occurrence Handle1:CAS:528:DyaK1MXhtFWjug%3D%3D
MM Bradford (1976) ArticleTitleA rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding Anal Biochem 72 248–254 Occurrence Handle942051 Occurrence Handle1:CAS:528:DyaE28XksVehtrY%3D Occurrence Handle10.1016/0003-2697(76)90527-3
A D’Aniello G D’Onofrio M Pischetola G D’Aniello A Vetere L Petrucelli GH Fisher (1993) ArticleTitleBiological role of D-amino acid oxidase and D-aspartate oxidase. Effects of D-amino acids J Biol Chem 268 26941–26949 Occurrence Handle7903300 Occurrence Handle1:CAS:528:DyaK3sXmsVKnu7o%3D
N Fujii (2002) ArticleTitleD-Amino acids in living higher organisms Orig Life Evol Biosphere 32 103–127 Occurrence Handle1:CAS:528:DC%2BD38XkvFCgtbc%3D Occurrence Handle10.1023/A:1016031014871
SJ Gould GA Keller N Hosken J Wilkinson S Subramani (1989) ArticleTitleA conserved tripeptide sorts proteins to peroxisomes J Cell Biol 108 1657–1664 Occurrence Handle2654139 Occurrence Handle1:CAS:528:DyaL1MXhvVyntLY%3D Occurrence Handle10.1083/jcb.108.5.1657
N Guex MC Peitsch (1997) ArticleTitleSWISS-MODEL and the Swiss-PdbViewer: an environment for comparative protein modeling Electrophoresis 18 2714–2723 Occurrence Handle9504803 Occurrence Handle1:CAS:528:DyaK1cXhvFaks70%3D Occurrence Handle10.1002/elps.1150181505
A Hashimoto T Oka (1997) ArticleTitleFree D-aspartate and D-serine in the mammalian brain and periphery Prog Neurobiol 52 325–353 Occurrence Handle9247969 Occurrence Handle1:CAS:528:DyaK2sXlt1Ghs7w%3D Occurrence Handle10.1016/S0301-0082(97)00019-1
Homma H (2002) D-Aspartate in the mammalian body. Viva Origino 30: 204–215 (available online at http://www.origin-life.gr.jp/3004/3004204/3004204.pdf)
K Imai T Fukushima T Santa H Homma K Hamase K Sakai M Kato (1996) ArticleTitleAnalytical chemistry and biochemistry of D-amino acids Biomed Chromatogr 10 303–312 Occurrence Handle8949912 Occurrence Handle1:CAS:528:DyaK28XntlKmt7s%3D Occurrence Handle10.1002/(SICI)1099-0801(199611)10:6<303::AID-BMC624>3.0.CO;2-B
Y Kera H Aoyama N Watanabe R Yamada (1996) ArticleTitleDistribution of D-aspartate oxidase and free D-glutamate and D-aspartate in chicken and pigeon tissues Comp Biochem Physiol B Biochem Mol Biol 115 121–126 Occurrence Handle8896337 Occurrence Handle1:STN:280:ByiD2M7mslI%3D Occurrence Handle10.1016/0305-0491(96)00089-2
Y Kera H Nagasaki A Iwashima R Yamada (1992) ArticleTitlePresence of D-aspartate oxidase and free D-aspartate in amphibian (Xenopus laevis, Cynops pyrrhogaster) tissues Comp Biochem Physiol B 103 345–348 Occurrence Handle1424565 Occurrence Handle1:STN:280:ByyD2cjosV0%3D Occurrence Handle10.1016/0305-0491(92)90302-8
R Konno (1998) ArticleTitleRat D-amino-acid oxidase cDNA: rat D-amino-acid oxidase as an intermediate form between mouse and other mammalian D-amino-acid oxidases Biochim Biophys Acta 1395 165–170 Occurrence Handle9473656 Occurrence Handle1:CAS:528:DyaK1cXnt12itg%3D%3D
G Kreil (1997) ArticleTitleD-Amino acids in animal peptides Annu Rev Biochem 66 337–345 Occurrence Handle9242910 Occurrence Handle1:CAS:528:DyaK2sXktFOhsrw%3D Occurrence Handle10.1146/annurev.biochem.66.1.337
A Mattevi MA Vanoni F Todone M Rizzi A Teplyakov A Coda M Bolognesi B Curti (1996) ArticleTitleCrystal structure of D-amino acid oxidase: a case of active site mirror-image convergent evolution with flavocytochrome b 2 Proc Natl Acad Sci USA 93 7496–7501 Occurrence Handle8755502 Occurrence Handle1:CAS:528:DyaK28XksFSku7k%3D Occurrence Handle10.1073/pnas.93.15.7496
D Mengin-Lecreulx (1982) ArticleTitleCytoplasmic steps of peptidoglycan synthesis in Escherichia coli J Bacteriol 151 1109–1117 Occurrence Handle6125497 Occurrence Handle1:CAS:528:DyaL38XlsF2hu7s%3D
H Mizutani I Miyahara K Hirotsu Y Nishina K Shiga C Setoyama R Miura (1996) ArticleTitleThree-dimensional structure of porcine kidney D-amino acid oxidase at 3.0 Å resolution J Biochem (Tokyo) 120 14–17 Occurrence Handle1:CAS:528:DyaK28XltFGltr8%3D
A Negri G Tedeschi F Ceciliani S Ronchi (1999) ArticleTitlePurification of beef kidney D-aspartate oxidase overexpressed in Escherichia coli and characterization of its redox potentials and oxidative activity towards agonists and antagonists of excitatory amino acid receptors Biochim Biophys Acta 1431 212–222 Occurrence Handle10209293 Occurrence Handle1:CAS:528:DyaK1MXivFeitb8%3D
MJ Osborn (1969) ArticleTitleStructure and biosynthesis of the bacterial cell wall Annu Rev Biochem 38 501–538 Occurrence Handle4896244 Occurrence Handle1:CAS:528:DyaF1MXkslyht7k%3D Occurrence Handle10.1146/annurev.bi.38.070169.002441
MC Peitsch (1995) ArticleTitleProtein modeling by E-mail Bio/Technology 13 658–660 Occurrence Handle1:CAS:528:DyaK2MXms1CnsLo%3D Occurrence Handle10.1038/nbt0795-658
MS Pilone (2000) ArticleTitleD-Amino acid oxidase: new findings Cell Mol Life Sci 57 1732–1747 Occurrence Handle11130179 Occurrence Handle1:CAS:528:DC%2BD3MXhsFKgsA%3D%3D Occurrence Handle10.1007/PL00000655
S Sacchi S Lorenzi G Molla MS Pilone C Rossetti L Pollegioni (2002) ArticleTitleEngineering the substrate specificity of D-amino-acid oxidase J Biol Chem 277 27510–27516 Occurrence Handle12021281 Occurrence Handle1:CAS:528:DC%2BD38XlvV2hsrw%3D Occurrence Handle10.1074/jbc.M203946200
MG Sarower T Matsui H Abe (2003) ArticleTitleDistribution and characteristics of D-amino acid and D-aspartate oxidases in fish tissues J Exp Zoolog A Comp Exp Biol 295 151–159 Occurrence Handle12541299 Occurrence Handle10.1002/jez.a.10217
T Schwede J Kopp N Guex MC Peitsch (2003) ArticleTitleSWISS-MODEL: An automated protein homology-modeling server Nucleic Acids Res 31 3381–3385 Occurrence Handle12824332 Occurrence Handle1:CAS:528:DC%2BD3sXltVWjsLg%3D Occurrence Handle10.1093/nar/gkg520
C Setoyama R Miura (1997) ArticleTitleStructural and functional characterization of the human brain D-aspartate oxidase J Biochem (Tokyo) 121 798–803 Occurrence Handle1:CAS:528:DyaK2sXktVOit70%3D
T Simonic S Duga A Negri G Tedeschi M Malcovati ML Tenchini S Ronchi (1997) ArticleTitlecDNA cloning and expression of the flavoprotein D-aspartate oxidase from bovine kidney cortex Biochem J 322 729–735 Occurrence Handle9148742 Occurrence Handle1:CAS:528:DyaK2sXit1Ojsbo%3D
M Tada K Fukui K Momoi Y Miyake (1990) ArticleTitleCloning and expression of a cDNA encoding mouse kidney D-amino acid oxidase Gene 90 293–297 Occurrence Handle1976103 Occurrence Handle1:CAS:528:DyaK3MXjs1yqsA%3D%3D Occurrence Handle10.1016/0378-1119(90)90193-U
S Takahashi T Kakuichi K Fujii Y Kera R Yamada (2005) ArticleTitlePhysiological role of D-aspartate oxidase in the assimilation and detoxification of D-aspartate in the yeast Cryptococcus humicola Yeast 22 1203–1212 Occurrence Handle16278929 Occurrence Handle1:CAS:528:DC%2BD2MXhtlSqs7%2FE Occurrence Handle10.1002/yea.1303
S Takahashi T Takahashi Y Kera R Matsunaga H Shibuya R Yamada (2004) ArticleTitleCloning and expression in Escherichia coli of the D-aspartate oxidase gene from the yeast Cryptococcus humicola and characterization of the recombinant enzyme J Biochem 135 533–540 Occurrence Handle15115779 Occurrence Handle1:CAS:528:DC%2BD2cXltFCgtLk%3D Occurrence Handle10.1093/jb/mvh068
VI Tishkov SV Khoronenkova (2005) ArticleTitleD-amino Acid oxidase: structure, catalytic mechanism, and practical application Biochemistry (Mosc) 70 40–54 Occurrence Handle1:CAS:528:DC%2BD2MXhs1Gis7w%3D
S Umhau L Pollegioni G Molla K Diederichs W Welte MS Pilone S Ghisla (2000) ArticleTitleThe x-ray structure of D-amino acid oxidase at very high resolution identifies the chemical mechanism of flavin-dependent substrate dehydrogenation Proc Natl Acad Sci USA 97 12463–12468 Occurrence Handle11070076 Occurrence Handle1:CAS:528:DC%2BD3cXotFyhsr4%3D Occurrence Handle10.1073/pnas.97.23.12463
R Yamada H Nagasaki Y Nagata Y Wakabayashi A Iwashima (1989) ArticleTitleAdministration of D-aspartate increases D-aspartate oxidase activity in mouse liver Biochim Biophys Acta 990 325–328 Occurrence Handle2923913 Occurrence Handle1:CAS:528:DyaL1MXitFSjtrk%3D
R Yamada H Nagasaki Y Wakabayashi A Iwashima (1988) ArticleTitlePresence of D-aspartate oxidase in rat liver and mouse tissues Biochim Biophys Acta 965 202–205 Occurrence Handle2896518 Occurrence Handle1:CAS:528:DyaL1cXktVChu7k%3D
K Zaar (1996) ArticleTitleLight and electron microscopic localization of D-aspartate oxidase in peroxisomes of bovine kidney and liver: an immunocytochemical study J Histochem Cytochem 44 1013–1019 Occurrence Handle8773567 Occurrence Handle1:CAS:528:DyaK28XlsVOisLk%3D
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Katane, M., Furuchi, T., Sekine, M. et al. Molecular cloning of a cDNA encoding mouse D-aspartate oxidase and functional characterization of its recombinant proteins by site-directed mutagenesis. Amino Acids 32, 69–78 (2007). https://doi.org/10.1007/s00726-006-0350-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-006-0350-x