Abstract
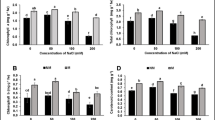
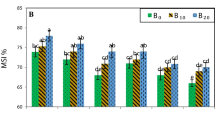
Waterlogging occurs due to poor soil drainage or excessive rainfall. It is a serious abiotic stress factor that negatively affects crop growth. Waterlogging often causes plants to shed leaves, fruits, and, ultimately, to die. Peach (Prunus persica) trees are generally intolerant to waterlogging, and the primary peach rootstock used in Chinais “Maotao,” which has very poor resistance to sensitivity. Therefore, waterlogging has become a restriction on the development of the peach industry in many regions. In this experiment, we tested the waterlogging resistance of “Maotao (Prunus persica (L.) Batsch)” (MT), “Shannong1 (GF677 × Cadaman)” (SN1), and “Mirabolano 29C (Prunus cerasifera)” (M29C) rootstocks. Using a simulated waterlogging method, the effects of waterlogging on the photosynthetic system, leaf pigments, osmotic adjustment, lipid membrane peroxidation, and antioxidant system of these three peach rootstocks were studied, and the changes of chlorophyll fluorescence parameters and fluorescence imaging were observed. The results showed that, with prolonged waterlogging, the photosynthetic pigment content and photosynthesis of the three peach rootstocks decreased rapidly, but the decomposition rate of SN1 and M29C chlorophyll was slower, and it still had high light energy absorption and energy transfer capabilities under waterlogging stress, which reduced the damage caused by waterlogging stress; under the stress of flooding, the osmoregulatory substances of the three rootstocks increased to varying degrees compared with normal conditions. At the same time, the enzyme activity of superoxide dismutase (SOD) activity, peroxidase (POD) activity, and catalase (CAT) activity in the leaves of the three rootstocks under flooding stress all increased and then decreased; during this period, malondialdehyde (MDA) continued to increase, and SN1 and M29C were significantly lower than MT; and chlorophyll fluorescence parameters, including the maximum photochemical efficiency (Fv/Fm), actual photochemical efficiency (ΦPSII), photochemical quenching coefficient (qP), non-photochemical quenching (NPQ), and electron transfer rate (ETR) decreased significantly. The tolerance of SN1 and M29C to waterlogging was significantly better than that of MT rootstocks. The rootstock and grafted seedlings of SN1 have good waterlogging tolerance.








Similar content being viewed by others
Data Availability
The data presented in this study are available on request from the corresponding author.
Change history
16 April 2024
A Correction to this paper has been published: https://doi.org/10.1007/s00709-024-01949-8
References
Ahmed S, Nawata E, Hosokawa M, Domae Y, Sakuratani T (2002) Alterations in photosynthesis and some antioxidant enzymatic activities of mungbean subjected to waterlogging. Plant Sci 163:117–123
Amador ML, Sancho S, Bielsa B, Gomez-Aparisi J, Rubio-Cabetas MJ (2012) Physiological and biochemical parameters controlling waterlogging stress tolerance in Prunus before and after drainage. Physiol Plant 144:357–368
Asada K (1992) Ascorbate peroxidase–a hydrogen peroxide-scavenging enzyme in plants. Physiol Plant 85:235–241
Baciu A, Cosmulescu S, Botu I, Gruia M, Visanu F (2009) Research on new generative rootstocks for peach trees (Prunus persica L. Batsch). Bull UASVM Hortic 66:115–119
Baker NR (2008) Chlorophyll fluorescence: a probe of photosynthesis in vivo. Annu Rev Plant Biol 59:89–113
Bennicelli R, Stępniewski W, Zakrzhevsky D, Balakhnina T, Stępniewska Z, Lipiec J (1998) The effect of soil aeration on superoxide dismutase activity, malondialdehyde level, pigment content and stomatal diffusive resistance in maize seedlings. Environ Exp Bot 39:203–211
Bhusal N, Kim HS, Han S-G, Yoon T-M (2020) Photosynthetic traits and plant–water relations of two apple cultivars grown as bi-leader trees under long-term waterlogging conditions. Environ Exp Bot 176:104111
Boonkorn P (2016) Impact of hot water soaking on antioxidant enzyme activities and some qualities of storage tomato fruits. Int Food Res J 23:934
Bowler C, Montagu MV, Inze D (1992) Superoxide dismutase and stress tolerance. Ann Rev Plant Biol 43:83–116
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chen H, Qualls RG, Blank RR (2005) Effect of soil flooding on photosynthesis, carbohydrate partitioning and nutrient uptake in the invasive exotic Lepidium latifolium. Aquat Bot 82:250–268
Colmer TD, Pedersen O (2008) Underwater photosynthesis and respiration in leaves of submerged wetland plants: gas films improve CO2 and O2 exchange. New Phytol 177:918–926
de Oliveira VC, Joly CA (2010) Flooding tolerance of Calophyllum brasiliense Camb. (Clusiaceae): morphological, physiological and growth responses. Trees 24:185–193
Dickin E, Wright D (2008) The effects of winter waterlogging and summer drought on the growth and yield of winter wheat (Triticum aestivum L.). Eur J Agron 28:234–244
Elstner EF, Heupel A (1976) Inhibition of nitrite formation from hydroxylammoniumchloride: a simple assay for superoxide dismutase. Anal Biochem 70:616–620
Faust M, Timon B (2010) Origin and dissemination of peach. Hortic Rev 17:331–379
Flagella Z, Pastore D, Campanile R, Di Fonzo N (1994) Photochemical quenching of chlorophyll fluorescence and drought tolerance in different durum wheat (Triticum durum) cultivars. J Agric Sci 122:183–192
Ge Y, He X, Wang J, Jiang B, Ye R, Lin X (2014) Physiological and biochemical responses of Phoebe bournei seedlings to water stress and recovery. Acta Physiol Plant 36:1241–1250
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930
Gorbe E, Calatayud A (2012) Applications of chlorophyll fluorescence imaging technique in horticultural research: a review. Sci Hortic 138:24–35
Gu X, Xue L, Lu L, Xiao J, Song G, Xie M, Zhang H (2021) Melatonin enhances the waterlogging tolerance of prunus persica by modulating antioxidant metabolism and anaerobic respiration. J Plant Growth Regul 40:2178–2190
Habibi G, Hajiboland R (2012) Comparison of photosynthesis and antioxidative protection in Sedum album and Sedum stoloniferum (Crassulaceae) under water stress. Photosynthetica 50:508–518
Haddadi BS, Hassanpour H, Niknam V (2016) Effect of salinity and waterlogging on growth, anatomical and antioxidative responses in Mentha aquatica L. Acta Physiol Plant 38:1–11
Han Q, Kang G, Guo T (2013) Proteomic analysis of spring freeze-stress responsive proteins in leaves of bread wheat (Triticum aestivum L.). Plant Physiol Biochem 63:236–244
He L, Yu L, Li B, Du N, Guo S (2018) The effect of exogenous calcium on cucumber fruit quality, photosynthesis, chlorophyll fluorescence, and fast chlorophyll fluorescence during the fruiting period under hypoxic stress. BMC Plant Biol 18:1–10
Hirabayashi Y, Mahendran R, Koirala S, Konoshima L, Yamazaki D, Watanabe S, Kim H, Kanae S (2013) Global flood risk under climate change. Nat Clim Chang 3:816–821
Hossain Z, López-Climent MF, Arbona V, Pérez-Clemente RM, Gómez-Cadenas A (2009) Modulation of the antioxidant system in citrus under waterlogging and subsequent drainage. J Plant Physiol 166:1391–1404
Hu DG, Ma QJ, Sun CH, Sun MH, You CX, Hao YJ (2016) Overexpression of MdSOS2L1, a CIPK protein kinase, increases the antioxidant metabolites to enhance salt tolerance in apple and tomato. Physiol Plant 156:201–214
Irfan M, Hayat S, Hayat Q, Afroz S, Ahmad A (2010) Physiological and biochemical changes in plants under waterlogging. Protoplasma 241:3–17
Jones DT, Sah JP, Ross MS, Oberbauer SF, Hwang B, Jayachandran K (2006) Responses of twelve tree species common in Everglades tree islands to simulated hydrologic regimes. Wetlands 26:830–844
Karuppanapandian T, Moon J-C, Kim C, Manoharan K, Kim W (2011) Reactive oxygen species in plants: their generation, signal transduction, and scavenging mechanisms. Aust J Crop Sci 5:709–725
Kebbas S, Lutts S, Aid F (2015) Effect of drought stress on the photosynthesis of Acacia tortilis subsp. raddiana at the young seedling stage. Photosynthetica 53:288–298
Kołton A, Kęska K, Czernicka M (2020) Selection of tomato and cucumber accessions for waterlogging sensitivity through morpho-physiological assessment at an early vegetative stage. Agronomy 10:1490
Kozlowski T (1982) Water supply and tree growth. Part II. Flooding. For Abstr (United Kingdom) 43:57–95
Kozlowski T (1997) Responses of woody plants to flooding and salinity. Tree Physiol 17:490–490
Kreuzwieser J, Gessler A (2010) Global climate change and tree nutrition: influence of water availability. Tree Physiol 30:1221–1234
Larson KD, Schaffer B, Davies FS (1991) Flooding, leaf gas exchange, and growth of mango in containers. J Am Soc Hortic Sci 116:156–160
Li Z, Fan D, Chen F, Yuan Q, Chow W, Xie Z (2015) Physiological integration enhanced the tolerance of Cynodon dactylon to flooding. Plant Biol 17:459–465
Li Y, Lv Y, Lian M, Peng F, Xiao Y (2021) Effects of combined glycine and urea fertilizer application on the photosynthesis, sucrose metabolism, and fruit development of peach. Sci Hortic 289:110504
Lichtenthaler HK, Wellburn AR (1983) Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Portland Press Ltd
Malik AI, Colmer TD, Lambers H, Setter TL, Schortemeyer M (2002) Short-term waterlogging has long-term effects on the growth and physiology of wheat. New Phytol 153:225–236
Mahajan S, Tuteja N (2005) Cold, salinity and drought stresses: an overview. Arch Biochem Biophys 444(2):139–158
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence—a practical guide. J Exp Bot 51:659–668
Mishra S, Patro L, Mohapatra P, Biswal B (2008) Response of senescing rice leaves to flooding stress. Photosynthetica 46:315–317
Nasef IN (2018) Short hot water as safe treatment induces chilling tolerance and antioxidant enzymes, prevents decay and maintains quality of cold-stored cucumbers. Postharvest Biol Technol 138:1–10
Naumann JC, Young DR, Anderson JE (2008) Leaf chlorophyll fluorescence, reflectance, and physiological response to freshwater and saltwater flooding in the evergreen shrub, Myrica cerifera. Environ Exp Bot 63:402–409
Nedbal L, Soukupová J, Kaftan D, Whitmarsh J, Trtílek M (2000) Kinetic imaging of chlorophyll fluorescence using modulated light. Photosynth Res 66:3–12
Ou L, Dai X, Zhang Z, Zou X (2011) Responses of pepper to waterlogging stress. Photosynthetica 49:339–345
Oxborough K (2004) Imaging of chlorophyll a fluorescence: theoretical and practical aspects of an emerging technique for the monitoring of photosynthetic performance. J Exp Bot 55:1195–1205. https://doi.org/10.1093/jxb/erh145
Parent C, Capelli N, Berger A, Crèvecoeur M, Dat JF (2008) An overview of plant responses to soil waterlogging. Plant Stress 2:20–27
Parolin P (2001) Morphological and physiological adjustments to waterlogging and drought in seedlings of Amazonian floodplain trees. Oecologia 128:326–335
Patterson BD, MacRae EA, Ferguson IB (1984) Estimation of hydrogen peroxide in plant extracts using titanium (IV). Anal Biochem 139:487–492
Pimentel P, Almada RD, Salvatierra A, Toro G, Arismendi MJ, Pino MT, Sagredo B, Pinto M (2014) Physiological and morphological responses of Prunus species with different degree of tolerance to long-term root hypoxia. Sci Hortic 180:14–23
Pompeiano A, Huarancca Reyes T, Moles TM, Guglielminetti L, Scartazza A (2019) Photosynthetic and growth responses of Arundo donax L. plantlets under different oxygen deficiency stresses and reoxygenation. Front Plant Sci 10:408
Rao L, Li S, Cui X (2021) Leaf morphology and chlorophyll fluorescence characteristics of mulberry seedlings under waterlogging stress. Sci Rep 11:1–11
Rolfe SA, Scholes JD (2002) Extended depth-of-focus imaging of chlorophyll fluorescence from intact leaves. Photosynth Res 72:107–115
Schaffer B, Andersen PC, Ploetz R (1992) Responses of fruit crops to flooding. Hortic Rev 13:257–313
Schreiber U, Quayle P, Schmidt S, Escher BI, Mueller JF (2007) Methodology and evaluation of a highly sensitive algae toxicity test based on multiwell chlorophyll fluorescence imaging. Biosens Bioelectron 22:2554–2563
Simova-Stoilova L, Demirevska K, Kingston-Smith A, Feller U (2012) Involvement of the leaf antioxidant system in the response to soil flooding in two Trifolium genotypes differing in their tolerance to waterlogging. Plant Sci 183:43–49
Smethurst CF, Shabala S (2003) Screening methods for waterlogging tolerance in lucerne: comparative analysis of waterlogging effects on chlorophyll fluorescence, photosynthesis, biomass and chlorophyll content. Funct Plant Biol 30:335–343
Steffens B, Sauter M (2009) Epidermal cell death in rice is confined to cells with a distinct molecular identity and is mediated by ethylene and H2O2 through an autoamplified signal pathway. Plant Cell 21:184–196
Szymajda M, Pruski K, Zurawicz E, Sitarek M (2020) The nursery value of new Prunus persica seedling rootstocks for peach cultivars. Sci Hortic 266:109282. https://doi.org/10.1016/j.scienta.2020.109282
Tamang BG, Fukao T (2015) Plant adaptation to multiple stresses during submergence and following desubmergence. Int J Mol Sci 16:30164–30180
Tattini M, Loreto F, Fini A, Guidi L, Brunetti C, Velikova V, Gori A, Ferrini F (2015) Isoprenoids and phenylpropanoids are part of the antioxidant defense orchestrated daily by drought‐stressed P latanus× acerifolia plants during Mediterranean summers. New Phytol 207(3):613–626
Tian L, Bi W, Liu X, Sun L, Li J (2019) Effects of waterlogging stress on the physiological response and grain-filling characteristics of spring maize (Zea mays L.) under field conditions. Acta Physiol Plant 41:1–14
Tuo X-Q, Li S, Wu Q-S, Zou Y-N (2015) Alleviation of waterlogged stress in peach seedlings inoculated with Funneliformis mosseae: changes in chlorophyll and proline metabolism. Sci Hortic 197:130–134
Wei W, Li D, Wang L, Ding X, Zhang Y, Gao Y, Zhang X (2013) Morpho-anatomical and physiological responses to waterlogging of sesame (Sesamum indicum L.). Plant Sci 208:102–111
Woo NS, Badger MR, Pogson BJ (2008) A rapid, non-invasive procedure for quantitative assessment of drought survival using chlorophyll fluorescence. Plant Methods 4:1–14
Wu W-M, Li J-C, Chen H-J, Wang S-J, Wei F-Z, Wang C-Y, Wang Y-H, Wu J-D, Zhang Y (2013) Effects of nitrogen fertilization on chlorophyll fluorescence change in maize (Zea mays L.) under waterlogging at seedling stage. J Food Agric Environ 11:545–552
Xiao Y, Wu X, Sun M, Peng F (2020) Hydrogen sulfide alleviates waterlogging-induced damage in peach seedlings via enhancing antioxidative system and inhibiting ethylene synthesis. Front Plant Sci 11:696
Xu F, Cai H, Zhang X, Su M, Zhou H, Li X, Zhang M, Hu Y, Gu C, Du J, Ye Z (2022) Comparison of Waterlogging Tolerance of Three Peach Rootstock Seedlings Based on Physiological, Anatomical and Ultra-Structural Changes. Horticulturae 8(8):720
Yamauchi T, Colmer TD, Pedersen O, Nakazono M (2018) Regulation of root traits for internal aeration and tolerance to soil waterlogging-flooding stress. Plant Physiol 176:1118–1130
Yin D, Chen S, Chen F, Guan Z, Fang W (2009) Morphological and physiological responses of two chrysanthemum cultivars differing in their tolerance to waterlogging. Environ Exp Bot 67:87–93
Yin D, Chen S, Chen F, Guan Z, Fang W (2010) Morpho-anatomical and physiological responses of two Dendranthema species to waterlogging. Environ Exp Bot 68:122–130
Zhang Y, Jin P, Huang Y, Shan T, Wang L, Li Y, Zheng Y (2016) Effect of hot water combined with glycine betaine alleviates chilling injury in cold-stored loquat fruit. Postharvest Biol Technol 118:141–147
Zhang F, Zhu K, Wang Y, Zhang Z, Lu F, Yu H, Zou J (2019) Changes in photosynthetic and chlorophyll fluorescence characteristics of sorghum under drought and waterlogging stress. Photosynthetica 57:1156–1164
Zhongcheng Z, Gang L, Wei C, Feng X, Xiaomei S, Zexiong C (2019) Physiological responses and tolerance evaluation of five poplar varieties to waterlogging. Notulae Botanicae Horti Agrobotanici Cluj-Napoca 47:658–667
Zhou C, Bai T, Wang Y, Wu T, Zhang X, Xu X, Han Z (2017) Morpholoical and enzymatic responses to waterlogging in three Prunus species. Sci Hortic 221:62–67
Ziegler VH, Ploschuk E, Weibel A, Insausti P (2017) Short-term responses to flooding stress of three Prunus rootstocks. Sci Hortic 224:135–141
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2019YFD1000103) and the National Modern Agro-industry Technology Research System Fund (Grant No. CARS-30-2-02).
Author information
Authors and Affiliations
Contributions
Binbin Zhang: conceptualization, methodology, formal analysis, investigation, writing—original draft, review, editing, and supervision. Maoxiang Sun: writing, review, editing, and supervision. Wenxin Liu: formal analysis and investigation. Min Lian: investigation. Sankui Yang: writing, review, editing, and supervision. Futian Peng: supervision, resources, and funding acquisition. Yuansong Xiao: formal analysis and investigation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Hanns H. Kassemeyer.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Waterlogging had a significant negative effect on the photosynthetic and gas exchange parameters of both rootstocks and grafted seedlings.
• Waterlogging had a significant negative effect on membrane lipid peroxidation and ROS accumulation in leaves of both rootstocks and grafted seedlings.
• Waterlogging induced antioxidant enzymes and osmotic adjustment in both rootstocks and grafted seedlings.
• The tolerance of SN1 and M29C to waterlogging was significantly better than that of MT rootstocks.
The original online version of this article was revised: Figures 5, 6 and 7 were incorrectly renumbered in the published proof.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, B., Sun, M., Liu, W. et al. Waterlogging resistance and evaluation of physiological mechanism of three peach (Prunus persica) rootstocks. Protoplasma 260, 1375–1388 (2023). https://doi.org/10.1007/s00709-023-01850-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-023-01850-w