Abstract
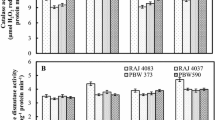
Wheat crop may experience heat stress during post-anthesis phase associated with oxidative stress, enhanced senescence, and reduced productivity. Stay green is a desirable character for the selection for heat tolerance in wheat. In the present study, antioxidant metabolism was studied under post-anthesis heat stress in field during monocarpic senescence by comparing two wheat genotypes, namely Hindi62 (heat tolerant and delayed senescent) and PBW343 (heat susceptible and early senescent). Hindi62 exhibited lesser oxidative stress, membrane damage, and coordinated antioxidant defense as compared to PBW343 under heat stress during post-anthesis stage. Higher activity of SOD, CAT, APX, GR, and MDHAR under heat stress contributed towards delayed senescence in Hindi62 compared to PBW343. GSH/GSSG ratio was also maintained at higher level in Hindi62 under heat stress compared to PBW343 during senescence. Hence, the present study clearly shows that upregulated level of the total antioxidant capacity during grain development contributed towards delayed senescence and heat tolerance in Hindi62 compared to the heat-susceptible PBW343.






Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Almeselmani M, Deshmukh PS, Sairam RK, Kushwaha SR, Singh TP (2006) Protective role of antioxidant enzymes under high temperature stress. Plant Sci 171:382–388
Asada K (1984) Chloroplasts: formation of active oxygen and its scavenging. Methods Enzymol 105:422–429
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assay and an assay applicable to acrylamide gels. Anal Biochem 44:276–287
Bita CE, Gerats T (2013) Plant tolerance to high temperature in a changing environment: scientific fundamentals and production of heat stress-tolerant crops. Front Plant Sci 4:273
Brisson LF, Tenhaken R, Lamb C (1994) Function of oxidative cross-linking of cell wall structural proteins in plant disease resistance. Plant Cell 6:1703–1712
Chance B, Maehly AC (1955) Assay of catalase and peroxidases. Methods Enzymol 2:764–775
Chauhan S, Srivalli S, Nautiyal AR, Khanna-Chopra R (2009) Wheat cultivars differing in heat tolerance show a differential response to monocarpic senescence under high-temperature stress and the involvement of serine proteases. Photosynthetica 47:536–547
Chinnusamy V, Khanna-Chopra R (1996) Heat shock proteins: role in themotolerance in crop plants. Curr Sci 71:275–284
CIMMYT (1995) CIMMYT/NARS consultancy on ME1 bread wheat breeding. Wheat special report no. 30. Mexico, DF
Cossani CM, Reynolds MP (2012) Physiological traits for improving heat tolerance in wheat. Plant Physiol 160:1710–1718
Dat JF, Lopez‐Delgado H, Foyer CH, Scott IM (1998) Parallel changes in H2O2 and catalase during thermotolerance induced by salicylic acid or heat acclimation in mustard seedlings. Plant Physiol 116:1351–1357
de Pinto MC, Tommasi F, De Gara L (2000) Enzymes of the ascorbate biosynthesis and ascorbate-glutathione cycle in cultured cells of tobacco bright yellow 2. Plant Physiol Biochem 38:541–550
De Simone V, Soccio M, Borrelli GM, Pastore D, Trono D (2014) Stay-green trait-antioxidant status interrelationship in durum wheat (Triticum durum) flag leaf during post-flowering. J Plant Res 127:159–171
Farooq M, Bramley H, Palta JA, Siddique KH (2011) Heat stress in wheat during reproductive and grain-filling phases. Critical Rev Plant Sci 30:491–507
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930
Gusta LV, Benning NT, Wu G, Luo X, Liu X, Gusta ML, McHughen A (2009) Superoxide dismutase: an all-purpose gene for agri-biotechnology. Mol Breed 24:103–115
He Y, Huang B (2010) Differential responses to heat stress in activities and isozymes of four antioxidant enzymes for two cultivars of Kentucky bluegrass contrasting in heat tolerance. J American Soc Horti Sci 135:116–124
He Y, Liu Y, Cao W, Huai M, Xu B, Huang B (2005) Effects of salicylic acid on heat tolerance associated with antioxidant metabolism in Kentucky bluegrass. Crop Sci 45:988–995
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: 1 kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Hodges DM, Forney CF (2000) The effects of ethylene, depressed oxygen and elevated carbon dioxide on antioxidant profiles of senescing spinach leaves. J Exp Bot 51:645–655
Im YJ, Ji M, Lee A, Killens R, Grunden AM, Boss WF (2009) Expression of Pyrococcus furiosus superoxide reductase in Arabidopsis enhances heat tolerance. Plant Physiol 151:893–904
Ioannidi E, Kalamaki MS, Engineer C, Peteraki I, Alexandrou D, Mellidou I, Giovannonni J, Kanellis AK (2009) Expression profiling of ascorbic acid-related genes during tomato fruit development and ripening and in response to stress conditions. J Exp Bot 60:663–678
IPCC (2014) Climate change 2014: summary of policymakers. Fifth assessment report (AR5), Washington DC: Intergovernmental panel on climate change
Jiang Y, Huang B (2001) Effects of calcium on antioxidant activities and water relations associated with heat tolerance in two cool‐season grasses. J Exp Bot 52:341–349
Khanna-Chopra R (2012) Leaf senescence and abiotic stresses share reactive oxygen species-mediated chloroplast degradation. Protoplasma 249:469–481
Khanna-Chopra R, Selote DS (2007) Acclimation to drought stress generates oxidative stress tolerance in drought-resistant than -susceptible wheat cultivar under field conditions. Environ Exp Bot 60:276–283
Khanna-Chopra R, Sinha SK (1998) Prospects of success of biotechnological approaches for improving tolerance to drought stress in crop plants. Curr Sci 74:25–34
Kocsy G, Szalai G, Vágújfalvi A, Stéhli L, Orosz G, Galiba G (2004) Genetic study of glutathione accumulation during cold hardening in wheat. Planta 210:295–301
Loggini B, Scartazza A, Brugnoli E, Navari-Izzo F (1999) Antioxidant defense system, pigment composition and photosynthetic efficiency in two wheat varieties subjected to drought. Plant Physiol 119:1091–1099
Long SP, Ort DR (2010) More than taking the heat: crops and global change. Curr Opin Plant Biol 13:1–8
Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin-phenol reagent. J Biol Chem 193:265–275
Mahla R, Madan S, Rana MK, Munjal R (2011) Antioxidant defense system in wheat (Triticum aestivum L.) seedlings under heat stress and revival conditions. J Agri Sci Tech 5:27–34
Miller G, Suzuki N, Rizhsky L, Hegie A, Koussevitzky S, Mittler R (2007) Double mutants deficient in cytosolic and thylakoid ascorbate peroxidase reveal a complex mode of interaction between reactive oxygen species, plant development, and response to abiotic stresses. Plant Physiol 144:1777–1785
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410
Mittler R, Zilinskas BA (1993) Detection of ascorbate peroxidase activity in native gels by inhibition of the ascorbate dependent reduction of nitroblue tetrazolium. Anal Biochem 212:540–546
Mǿller IM, Jensen PE, Hansson A (2007) Oxidative modifications to cellular components in plants. Annu Rev Plant Biol 58:459–481
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Naudts K, Van den Berge J, Farfan E, Rose P, AbdElgawad H, Ceulemans R, Nijs I (2014) Future climate alleviates stress impact on grassland productivity through altered antioxidant capacity. Environ Exp Bot 99:150–158
Navari-Izzo F, Quartacci MF, Pinzino C, Vecchia FD, Sgherri CLM (1998) Thylakoid bound and stromal anti-oxidative enzymes in wheat treated with excess copper. Physiol Plant 104:630–638
Panchuk II, Volkov RA, Schöffl F (2002) Heat stress-and heat shock transcription factor-dependent expression and activity of ascorbate peroxidase in Arabidopsis. Plant Physiol 129:838–853
Reynolds MP, Balota M, Delgado MIB, Amani I, Fischer RA (1994) Physiological and morphological traits associated with spring wheat yield under hot, irrigated conditions. Func Plant Biol 21:717–730
Sairam RK, Srivastava GC, Saxena DC (2000) Increased antioxidant activity under elevated temperatures: a mechanism of heat stress tolerance in wheat genotypes. Biolol Plant 43:245–251
Sairam RK, Rao KV, Srivastava GC (2002) Differential response of wheat genotypes to long term salinity stress in relation to oxidative stress, antioxidant activity and osmolyte concentration. Plant Sci 163:1037–1046
Schaedle M, Bassham A (1977) Chloroplast glutathione reductase. Plant Physiol 53:1011–1012
Seevers PM, Daly JM, Catedral FF (1971) The role of peroxidase isozymes in resistance to wheat stem rust disease. Plant Physiol 48:353–360
Semwal VK, Singh B, Khanna-Chopra R (2014) Delayed expression of SAGs correlates with longevity in CMS wheat plants compared to its fertile plants. Physiol Mol Biol Plants 20:191–199
Shi WM, Muramoto Y, Ueda A, Takabe T (2001) Cloning of peroxisomal ascorbate peroxidase gene from barley and enhanced thermotolerance by overexpressing in Arabidopsis thaliana. Gene 273:23–27
Song XS, Hu WH, Mao WH, Ogweno JO, Zhou YH, Yu JQ (2005) Response of ascorbate peroxidase isoenzymes and ascorbate regeneration system to abiotic stresses in Cucumis sativus L. Plant Physiol Biochem 43:1082–1088
Srivalli S, Khanna-Chopra R (2009) Delayed wheat flag leaf senescence due to removal of spikelets is associated with increased activities of leaf antioxidant enzymes, reduced glutathione/oxidized glutathione ratio and oxidative damage to mitochondrial proteins. Plant Physiol Biochem 47:663–670
Suzuki N, Mittler R (2006) Reactive oxygen species and temperature stresses: a delicate balance between signaling and destruction. Physiol Plant 126:45–51
Suzuki N, Miller G, Sejima H, Herper J, Mittler R (2012) Enhanced seed production under prolonged heat stress conditions in Arabidopsis thaliana plants deficient in cytosolic ascorbate peroxidase 2. J Exp Bot 64:253–263
Szalai G, Tibor Kell T, Galiba G, Kocsy G (2009) Glutathione as an antioxidant and regulatory molecule in plants under abiotic stress conditions. J Plant Growth Reg 28:66–80
Takahashi S, Murata N (2008) How do environmental stresses accelerate photoinhibition? Trends Plant Sci 13:178–182
Tian FX, Gong JF, Wang GP, Wang GK, Fan ZY, Wang W (2012) Improved drought resistance in a wheat stay-green mutant tasg1 under field conditions. Biol Plant 56:509–515
Vacca RA, de Pinto MC, Valenti D, Passarella S, Marra E, De Gara L (2004) Production of reactive oxygen species, alteration of cytosolic ascorbate peroxidase and impairment of mitochondrial metabolism are early events in heat shock induced programmed cell death in tobacoo Bright-Yellow 2 cells. Plant Physiol 134:1100–1112
Veljovic-Jovanovic S, Noctor G, Foyer CH (2002) Are leaf hydrogen peroxide concentrations commonly overestimated? the potential influence of artefactual interference by tissue phenolics and ascorbate. Plant Physiol Biochem 40:501–507
Wang SY, Jiao HJ, Faust M (1991) Changes in ascorbate, glutathione and related enzyme activities during thidiazuron-induced bud break of apple. Physiol Plant 82:231–236
Woodbury W, Spencer AK, Stahmann MA (1971) An improved procedure using ferricyanide for detecting catalase isozymes. Anal Biochem 44:301–305
Yamasaki H, Takahashi S, Heshiki R (1999) The tropical fig Ficus microcarpaL. f. cv. golden leaves lacks heat-stable dehydroascorbate reductase activity. Plant Cell Physiol 40:640–646
Zentgraf U, Zimmermann P, Smykowski A (2012) Role of intracellular hydrogen peroxide as signalling molecule for plant senescence. In: Nagata T (eds) Senescence. Rejeka, Croatia. ISBN: 978-953-51-0144-4, In Tech, Available from: http://www.intechopen.com/books/senescence/role-of-intracellular-hydrogen-peroxide-as-signelling-molecule-for-plant-senescence, 7 Apr 2012
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Bhumi Nath Tripathi
Rights and permissions
About this article
Cite this article
Khanna-Chopra, R., Chauhan, S. Wheat cultivars differing in heat tolerance show a differential response to oxidative stress during monocarpic senescence under high temperature stress. Protoplasma 252, 1241–1251 (2015). https://doi.org/10.1007/s00709-015-0755-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-015-0755-z