Abstract
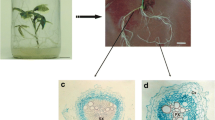
In this work, we studied the impact of the successive steps of the droplet-vitrification protocol technique employed for cryopreservation of Rubia akane hairy roots on the features of cortical, pericycle and endoderm cells of apical and central root segments, using histology techniques and combining qualitative and quantitative observations. In apical segments, plasmolysis (22–71 %, depending on cell type) was observed only after the loading treatment and did not increase after the following steps of the protocol. By contrast, in central segments, plasmolysis (39–45 %) was already observed after the sucrose pretreatment; it increased to 54–68 %, depending on cell type, after the loading treatment, but no further changes were noted after treatment with the vitrification solution. After liquid nitrogen exposure and unloading treatment, deplasmolysis was more rapid in apical segments, with cortical and pericycle cells having retrieved their original features. In central segments, only cortical cells had retrieved their original features and endoderm and pericycle cells were still highly plasmolysed. Nuclei were more strongly impacted by the cryopreservation protocol in central segments, where they displayed a highly condensed nucleoplasm from the loading treatment onwards and had not retrieved their original aspect after the unloading treatment. By contrast, nuclei had a much less condensed nucleoplasm in cells of apical segments, and they had retrieved their original aspect after the unloading treatment.




Similar content being viewed by others
References
Bagniol S, Engelmann F, Michaux-Ferrière N (1992) Histo-cytological study of apices from in vitro plantlets of date palm (Phoenix dactylifera L.) during a cryopreservation process. CryoLetters 13:405–412
Barraco G, Sylvestre I, Collin M, Escoute J, Lartaud M, Verdeil JL, Engelmann F (2013) Histological study of yam shoot tips during their cryopreservation using the encapsulation-dehydration technique. Protoplasma. doi:10.1007/s00709-013-0536-5
Benson EE, Hamill JD (1991) Cryopreservation and post freeze molecular and biosynthetic stability in transformed roots of Beta vulgaris and Nicotiana rustica. Plant Cell Tiss Org Cult 24:163–172
Buffard-Morel J, Verdeil JL, Pannetier C (1992) Embryogenèse somatique du cocotier (Cocos nucifera L.) à partir d'explants foliaires: étude histologique. Can J Bot 70:735–741
Engelmann F, Dussert S (2012) Cryopreservation. In: Normah MN, Chin HF, Reed BM (eds) Conservation of tropical plant species. Springer, Berlin, pp 107–120
Fisher DB (1968) Protein sustaining of ribboned epon sections for light microscopy. Histochemie 16:92–96
Fahy GM, MacFarlane DR, Angell CA, Meryman HT (1984) Vitrification as an approach to cryopreservation. Cryobiology 21:407–426
Gallard A (2008) Etude de la cryoconservation d'apex en vue d'une conservation à long terme de collections de ressources génétiques végétales: compréhension des phénomènes mis en jeu et évaluation de la qualité du matériel régénéré sur le modèle Pelargonium. PhD Dissertation, Ecole doctorale d'Angers, AGROCAMPUS-OUEST, Centre d'Angers, France
Georgiev ML, Pavlov AI, Bley T (2007) Hairy root type plant in vitro systems as sources of bioactive substances. Appl Microbiol Biotech 74:1175–1185
Gilbert KG, Cooke GT (2001) Dyes from plants: past usage, present understanding and potential. Plant Growth Reg 34:57–69
Hirata K, Mukai M, Goda S, Ishio-Kinugasa M, Yoshida K, Sakai A, Miyamoto K (2002) Cryopreservation of hairy root cultures of Vinca minor (L.) by encapsulation-dehydration. Biotech Lett 24:371–376
Hu ZB, Du M (2006) Hairy root and its application in plant genetic engineering. J Integr Plant Biol 48:21–127
Jung DW, Sung CK, Touno K, Yoshmatsu K, Shimomura K (2001) Cryopreservation of Hyoscyamus niger adventitious roots by vitrification. J Plant Physiol 158:801–805
Kim JB, Kim HH, Baek HJ, Cho EG, Kim YH, Engelmann F (2005) Changes in sucrose and glycerol content in garlic shoot tips during freezing using PVS3 solution. CryoLetters 26:103–112
Kim HH, Lee YG, Shin DJ, Kim T, Cho EG, Engelmann F (2009a) Development of alternative plant vitrification solutions in droplet-vitrification procedures. CryoLetters 30:320–334
Kim HH, Lee YG, Ko HC, Park SU, Gwag JG, Cho EG, Engelmann F (2009b) Development of alternative loading solutions in droplet-vitrification procedures. CryoLetters 30:291–299
Kim HH, Popova EV, Yi JY, Cho GT, Park SU, Lee SC, Engelmann F (2010) Cryopreservation of hairy roots of Rubia akane (Nakai) using a droplet-vitrification procedure. CryoLetters 31:473–484
Kim HH, Popova EV, Yi JY, Shin DJ, Baek HJ, Park SU, Engelmann F (2012) Development of a droplet-vitrification protocol for cryopreservation of Rubia akane (Nakai) hairy roots using a systematic approach. CryoLetters 33:506–517
Lambert E, Goossens A, Panis B, Van Labeke MC, Geelen D (2009) Cryopreservation of hairy root cultures of Maesa lanceolata and Medicago truncatula. Plant Cell Tiss Org Cult 96:289–296
Mankessi F, Saya A, Montes F, Lartaud M, Verdeil JL, Monteuuis O (2011) Histocytological characteristics of Eucalyptus urophylla x Eucalyptus grandis shoot apical meristems of different physiological ages. Trees: Struct Funct 25:415–424
Mankessi F, Saya A, Boudon F, Guedon Y, Montes F, Lartaud M, Verdeil JL, Monteuuis O (2010) Phase change-related variations of dome shape in Eucalyptus urophylla x Eucalyptus grandis shoot apical meristems. Trees: Struct Funct 24:743–752
Mari S, Engelmann F, Chabrillange N, Huet C, Michaux-Ferrière N (1995) Histo-cytological study of apices coffee (Coffea racemosa and C. sessiliflora) in vitro plantlets during their cryopreservation using the encapsulation-dehydration technique. CryoLetters 16:289–298
Matsumoto T, Sakai A, Yamada K (1994) Cryopreservation of in vitro grown apical meristems of wasabi (Wasabia japonica) by vitrification and subsequent high plant regeneration. Plant Cell Rep 13:442–446
McMullen RL, Bauza E, Gondran C, Oberto G, Domloge N, Dal Farra C, Moore DJ (2010) Image analysis to quantify histological and immunofluorescent staining of ex vivo skin and skin cell cultures. Intl J Cosmetic Sci 32:143–154
Murashige T, Skoog F (1962) A revised medium for a rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:437–497
Nishizawa S, Sakai A, Amano AY, Matsuzawa T (1993) Cryopreservation of asparagus (Asparagus officinalis L.) embryogenic suspension cells and subsequent plant regeneration by vitrification. Plant Sci 91:67–73
Panis B, Piette B, Swennen R (2005) Droplet-vitrification of apical meristems: a cryopreservation protocol applicable to all Musaceae. Plant Sci 168:45–55
Sakai A, Engelmann F (2007) Vitrification and encapsulation-vitrification and droplet-vitrification: a review. CryoLetters 28:151–172
Sakai A, Hirai D, Niino T (2008) Development of PVS-based vitrification and encapsulation-vitrification protocols. In: Reed BM (ed) Plant cryopreservation: a practical guide. Springer, Berlin, pp 33–57
Sakai A, Kobayashi S, Oiyama IE (1990) Cryopreservation of nucellar cells of navel orange (Citrus sinensis Osb. var. brasiliensis Tanaka) by vitrification. Plant Cell Rep 9:30–33
Teoh KH, Weathers PJ, Cheetham RD, Walcerz DB (1996) Cryopreservation of transformed (hairy) roots of Artemisia annua. Cryobiology 33:106–117
Volk GM, Caspersen AM (2007) Plasmolysis and recovery of different cell types in cryoprotected shoot tips of Mentha x piperita. Protoplasma 231:215–226
Xue SH, Luo XJ, Wu ZH, Zhang HL, Wang XY (2008) Cold storage and cryopreservation of hairy root cultures of medicinal plant Eruca sativa Mill., Astragalus membranaceus and Gentiana macrophylla Pall. Plant Cell Tiss Org Cult 92:251–260
Yap LV, Normah MN, Clyde MM, Chin HF (2011) Cryopreservation of Garcinia cowa shoot tips by vitrification: the effects of sucrose preculture and loading treatment on ultrastructural changes in meristematic cells. CryoLetters 32:188–196
Yi JY, Sylvestre I, Collin M, Salma M, Lee SY, Kim HH, Park HJ, Engelmann F (2012) Improved cryopreservation using droplet-vitrification method and histological changes associated with cryopreservation of madder (Rubia akane Nakai). Kor J Hort Sci Technol 30:79–84
Zeng JW, Yi GJ, Zhang QM (2005) Cell ultrastructure of papaya shoot-tip during cryopreservation. Acta Hort Sin 32:15–19
Acknowledgments
This work has received partial financial support from ARCAD, a flagship programme of Agropolis Fondation (Montpellier, France) (I. Engelmann-Sylvestre), and from the PHC 2010–11 STAR project no. 23200UJ. Montpellier Rio Imaging (MRI) is gratefully acknowledged for providing access to its facilities.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Peter Nick
Rights and permissions
About this article
Cite this article
Salma, M., Engelmann-Sylvestre, I., Collin, M. et al. Effect of the successive steps of a cryopreservation protocol on the structural integrity of Rubia akane Nakai hairy roots. Protoplasma 251, 649–659 (2014). https://doi.org/10.1007/s00709-013-0565-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-013-0565-0