Abstract
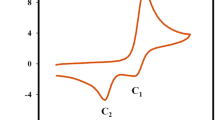
The electrochemical oxidation of 2,5-diethoxy-4-morpholinoaniline has been studied in various pHs using cyclic voltammetry and controlled potential coulometry. The results indicate that the electrochemically generated p-quinonediimine participates in different types of reactions (hydrolysis, hydroxylation, and trimerization). Instability of the produced p-quinonediimine depends on its structure, higher nucleophilicity of 2,5-diethoxy-4-morpholinoaniline, and pH of solution. In lower pH range, rate of hydrolysis is faster than hydroxylation and dimerization. In intermediate pHs rate of the dimerization is faster than hydrolysis and hydroxylation, and in the strongly alkaline solution, rate of the hydroxylation is faster than hydrolysis and dimerization. The effect of the charge of reaction site (C1) and N1=C1 bond order (Wiberg bond indices) on the hydrolysis rate were studied. Calculations were performed using density functional theory B3LYP level of theory and 6−311+G(p,d) basis set.
Graphical abstract











Similar content being viewed by others
References
Costentin C (2008) Chem Rev 108:2145
Houmam A (2008) Chem Rev 108:2180
Sun JJ, Wu Y, Song JF (2014) Electrochim Acta 115:386
Duan X, Tian L, Liu W, Chang L (2013) Electrochim Acta 94:192
Rafiee M, Nematollahi D (2007) Electroanalysis 19:1382
Saraswat A, Sharma LK, Singh S, Singh RKP (2013) Synth Met 167:31
Nematollahi D, Forooghi Z (2003) Electroanalysis 15:1639
Bard AJ, Faulker LR (2001) Electrochemical Methods, 2nd edn. Wiley, New York
Rafiee M, Nematollahi D (2008) Electrochim Acta 53:2751
Fotouhi L, Tammari E, Asadi S, Heravi MM, Nematollahi D (2009) Int J Chem Kinet 41:426
Esmaili R, Nematollahi D (2012) J Electrochem Soc 159:H792
Nematollahi D, Bamzadeh M, Shayani-Jam H (2010) Chem Pharm Bull 58:23
Greef R, Peat R, Peter LM, Pletcher D, Robinson J (1990) Instrumental methods in electrochemistry. Ellis Horwood Limited, New York
Saveant JM (2006) Elements of molecular and biomolecular electrochemistry. John Wiley & Sons, Jersey
Esmaili R, Varmaghani, Nematollahi D (2012) J Electrochem Soc 159:H680
Nematollahi D, Maleki A (2009) Electrochem Commun 11:488
Wang G, Fu X, Huang J, Wu L, Deng J (2011) J Electroanal Chem 661:351
Kadar M, Nagy Z, Karancsi T, Farsang G (2001) Electrochim Acta 46:3405
Miras MC, Silber JJ, Serno L (1986) J Electroanal Chem 2019:367
Dvorak V, Nemec I, Zyka J (1967) Microchem J 12:324
Salavagione HJ, Arias J, Garces P, Morallon E, Barbero C, Vazquez JL (2004) J Electroanal Chem 565:375
Steckan E, Baizer MM, Lund H (eds) (1991) Organic electrochemistry, an introduction and a guide. Marcel Dekker, New York, p 15
Esmaili R, Nematollahi D (2011) Electrochim Acta 56:3899
Beiginejad H, Nematollahi D, Varmaghani F (2013) J Electrochem Soc 160:H41
Nematollahi D, Shayani-Jam H, Alimoradi M, Niroomand S (2009) Electrochim Acta 54:7407
Beiginejad H, Nematollahi D (2013) Electrochim Acta 114:242
Beiginejad H, Nematollahi D (2014) J Org Chem 79:6326
Beiginejad H, Nematollahi D, Varmaghani F, Bayat M (2013) J Electrochem Soc 160:H469
Kirdant AS, Magar BK, Chondhekar TK (2012) J Chem Biol Phys Sci 2:147
Beginejad H, Nematollahi D, Varmaghani F, Shayani-Jam H (2013) Monatsh Chem 144:1481
Beiginejad H, Nematollahi D, Varmaghani F, Bayat M (2013) J Electrochem Soc 160:G3001
Varmaghani F, Nematollahi D, Mallakpour S, Esmaili R (2012) Green Chem 14:963
Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski J, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson BG, Chen W, Wong MW, Gonzalez C, Pople JA (2003) Gaussian 03, revision B. 04. Gaussian, Inc., Pittsburgh
Becke AD (1993) J Chem Phys 98:5648
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785
HyperChem (1997) Release 5.02. Hypercube Inc., Gainesville
Glendening ED, Badenhoop JK, Reed AE, Carpenter JE, Bohmann JA, Morales CM, Weinhold F (2001) NBO 5.0. Theoretical Chemistry Institute, University of Wisconsin, Madison
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beiginejad, H., Nematollahi, D. Mechanistic study of electrochemical oxidation of 2,5-diethoxy-4-morpholinoaniline in aqueous solutions: hydrolysis, trimerization, and hydroxylation processes. Monatsh Chem 146, 1495–1502 (2015). https://doi.org/10.1007/s00706-015-1409-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-015-1409-8