Abstract
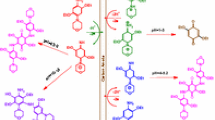
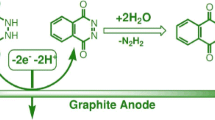
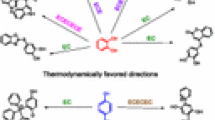
The electrochemical oxidation of 2,5-diethoxy-4-morpholinoaniline has been studied in the presence of thiobarbituric acid by the use of experimental and calculation methods. Cyclic voltammetry, controlled-potential coulometry and density functional theory (DFT) were used in this study. By the use of the cyclic voltammetry, the best condition of synthesis was obtained, and controlled-potential coulometry was performed under this condition. The results indicated that the produced p-benzoquinonediimine is unstable, and it was hydrolyzed in the experimental condition. It was found that the produced p-quinoneimine from oxidation/hydrolysis of 2,5-diethoxy-4-morpholinoaniline reacts with the thiobarbituric acid as a nucleophile and via an ECCCEC mechanism converts to the product. Also, computational calculations were used to analyze oxidation peaks in voltammetry. DFT, BP86 level of theory and 6-31G(p,d) basis set, was used to obtain the theoretical results.












Similar content being viewed by others
References
U.J. Pandit, G.A. Naikoo, M.U.D. Sheikh, G.A. Khan, K.K. Raj, S.N. Limaye, J. Pharm. Anal. 7, 258 (2017)
J. Zima, I. Svancara, J. Barek, J. Barek, K. Vytras, Crit. Rev. Anal. Chem. 39, 204 (2009)
N. Erk, Anal. Bioanal. Chem. 378, 1351 (2004)
K.M. Naik, S.T. Nandibewoor, J. Ind. Eng. Chem. 19, 1933 (2013)
S.W. da Silva, E.M.O. Navarro, M.A.S. Rodrigues, A.M. Bernardes, V. Pérez-Herranz, J. Electroanal. Chem. 832, 112 (2019)
J.H.Q. Lee, Y.R. Koh, R.D. Webster, J. Electroanal. Chem. 799, 92 (2017)
J.G. Bell, J. Wang, Electrochim. Acta 222, 678 (2016)
M.A.M. Cartaxo, C.M. Borges, M.I.S. Pereira, M.H. Mendonça, Electrochim. Acta 176, 1010 (2015)
A.M. Oliveira Brett, J.A.P. Piedade, S.H.P. Serrano, Electroanalysis 12, 969 (2000)
W.B.S. Machini, A.M. Oliveira-Brett, Electroanalysis 29, 1490 (2017)
K. Buchholz, R.H. Schirmer, J.K. Eubel, M.B. Akoachere, T. Dandekar, K. Becker, S. Gromer, Agents Chemother. 52, 183 (2008)
R.H. Schirmer, B. Coulibaly, A. Stich, M. Scheiwein, H. Merkle, J. Eubel, K. Becker, H. Becher, O. Müller, T. Zich, W. Schiek, B. Kouyaté, Redox Rep. 8, 272 (2003)
S. Koutros, C.F. Lynch, X. Ma, W.J. Lee, J.A. Hoppin, C.H. Christensen, G. Andreotti, L.B. Freeman, J.A. Rusiecki, L. Hou, D.P. Sandler, M.C.R. Alavanja, Int. J. Cancer 124, 1206 (2009)
D. Nematollahi, R. Esmaili, Tetrahedron Lett. 51, 4862 (2010)
R. Esmaili, D. Nematollahi, Electrochim. Acta 56, 3899 (2011)
H. Beiginejad, J. Electroanal. Chem. 825, 77 (2018)
A. Amani, S. Khazalpour, D. Nematollahi, J. Electroanal. Chem. 670, 36 (2012)
E. Salahifar, D. Nematollahi, M. Bayat, A. Mahyari, H. Amiri Rudbari, Org. Lett. 17, 4666 (2015)
S. Khazalpour, D. Nematollahi, Green Chem. 17, 3508 (2015)
D. Nematollahi, S. Momeni, S. Khazalpour, J. Electrochem. Soc. 161, H75 (2014)
H. Beiginejad, D. Nematollahi, Electrochim. Acta 114, 242 (2013)
H. Beiginejad, D. Nematollahi, J. Org. Chem. 79, 6326 (2014)
H. Beiginejad, D. Nematollahi, Monatsh. Chem. 146, 1495 (2015)
H. Beiginejad, A. Amani, D. Nematollahi, S. Khazalpour, Electrochim. Acta 154, 235 (2015)
P.M.W. Gill, B. Johnson, W. Chen, M.W. Wong, C. Gonzalez, J.A. Pople, Gaussian03, Version D. 01, Gaussian Inc., Pittsburgh, PA, 2005
HyperChem, Released 5.02 (Hypercube Inc., Gainesville, 1997)
V. Barone, M. Cossi, J. Phys. Chem. A 102, 1995 (1998)
H. Beiginejad, D. Nematollahi, M. Bayat, F. Varmaghani, A. Nazaripour, J. Electrochem. Soc. 160, H693 (2013)
H. Beiginejad, D. Nematollahi, S. Khazalpour, J. Electrochem. Soc. 163, H234 (2016)
H. Beiginejad, D. Nematollahi, S. Khazalpour, J. Iran. Chem. Soc. 14, 873 (2017)
H. Beiginejad, D. Nematollahi, S. Khazalpour, J. Electrochem. Soc. 164, H946 (2017)
Kosuke Izutsu, Electrochemistry in Nonaqueous Solutions, 2nd edn. (Wiley, Weinheim, 2002)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beiginejad, H., Nematollahi, D. Electrochemical oxidation of 2,5-diethoxy 4-morpholinoaniline in the presence of thiobarbituric acid: present an ECCCEC Mechanism. J IRAN CHEM SOC 16, 2177–2185 (2019). https://doi.org/10.1007/s13738-019-01687-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-019-01687-2