Abstract
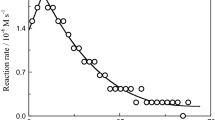
The interaction between chromium(III) and picolinic acid in weak acid aqueous solution was studied, resulting in the formation of a complex upon substitution of water molecules in the chromium(III) coordination sphere. Experimental results show that the reaction takes place in multiple steps. The first step is the formation of an ion pair, the second step (two consecutive steps) is the slow one corresponding to substitution of the first water molecule from the chromium aqueous complex coordination sphere by a picolinic acid molecule via oxygen atom of the carboxylic acid group and substitution of the second water molecule via nitrogen of the pyridine ring forming an 1:1 complex. Both consecutive steps were independent of chromium concentration. The rate constants of the 1st and 2nd consecutive steps were increased by increasing picolinic acid concentration. The corresponding activation parameters are ∆H *1obs = 28.4 ± 4 kJ mol−1, ∆S *1obs = −202 ± 26 J K−1 mol−1, ∆H *2obs = 39.6 ± 5 kJ mol−1, and ∆S *2obs = −175 ± 19 J K−1 mol−1. The third step is fast, corresponding to formation of the final complex [Cr(pic)3]. The logarithms of the formation constants of 1:1 and 1:3 complexes were found to be 1.724 and 4.274, respectively.
Graphical Abstract









Similar content being viewed by others
References
Idriss KA, Saleh MS, Sedaira H, Seleim MM, Hashem EY (1991) Monatsh Chem 122:507
Pope CG, Matijecic E, Pate RC (1981) J Colloid Interface Sci 80:74
Lannon M, Lappin AG, Segal MG (1984) Inorg Chem 3:4167
Anderson RA (1993) Essential and toxic trace elements in human health and disease. Wiley-Liss, New York, p 221
Anderson RA (1995) J Adv Med 8:37
Evans GW (1989) Int J Biosci Med Res 11:163
Press RI, Geller J, Evans GW (1990) West J Med 152:41
Abdullah MA, Barrett J, Obrien P (1984) J Chem Soc Dalton Trans 1647
Piispanen J, Lajumen LHJ (1996) Acta Chem Scand 50:1074
Thomas V, Tampouris K, Petrou AL (2008) Bioinorg Chem Appl Article ID 624583
Weyh JA, Hamm RE (1969) Inorg Chem 8:2298
Lubes V (2005) J Solut Chem 34:899
Bhattacharya M, De GS (1981) Indian J Chem 20A:780
Baes CF, Mensner RE (1976) The hydrolysis of cations. Wiley, New York
Cotton FA, Wilkson G, Murillo CA, Bochmann M (1999) Advanced inorganic chemistry, 6th edn. Wiley, New York
Khan IA, Kabir-ud-Din J (1981) J Inorg Nucl Chem 43:1082
Khan IA, Kabir-ud-Din J (1984) Indian J Chem 23A:98
Khan IA, Shahid M, Kabir-ud-Din J (1983) Indian J Chem 23A:382
Khan IA, Kabir-ud-Din J (1986) Transition Met Chem 11:391
Secco F, Venturini M (1975) Inorg Chem 14:1978
Perlumutter-Hayman B, Secco F, Tapnhi E, Venturini M (1980) J Chem Soc Dalton Trans 1124
Campisi A, Tregloan PA (1985) Inorg Chim Acta 100:251
Pelizzett E, Mentasti E, Giraudi G (1975) Inorg Chim Acta 15:L1
Mentasti E, Secco F, Venturini M (1982) Inorg Chem 21:602
Funahashi S, Ishihara K, Tanaka M (1983) Inorg Chem 22:2072
Kimura M, Shirai J (1978) J Inorg Nucl Chem 40:1085
Kirshnamoorthy CR, Harris GM (1980) J Coord Chem 10:55
Kabir-ud-Din, Shahid M, Khan IA (1990) Z Phys Chem 271:101
Hamm RE, Jhanson RL, Pertein RH, Davis RE (1958) J Am Chem Soc 804:469
Basolo F, Pearson RG (1958) Mechanism of inorganic reaction, 2nd edn. Wiley, New York, Chapter 6
Shahid M, Khan IA, Kabir-ud-Din (1990) J Chem Soc Dalton Trans 3007
Guindy NM, Abou-Gamra ZM, Abdel-Messih MF (2000) Monatsh Chem 131:857
Guindy NM, Abou-Gamra ZM, Abdel-Messih MF (1999) J Chim Phys 96:851
Espenson JH (1971) Inorg Chem 8:1554
Tyagi SC, Khan AA (1978) J Inorg Nucl Chem 40:1899
Swaddle TW (1974) Coord Chem Rev 14:217
Swaddle TW, Xu FC, Krourse HIR (1985) Inorg Chem Rev 24:267
Lincoln SF, Merbach AE (1995) Adv Inorg Chem 42:1
Plane RA, Taube H (1952) J Phys Chem 56:33
McBane GC (1998) J Chem Educ 75:919
Broadhusst CL, Schmidt WF, Reeves JB III, Polansky MM, Gautschi K, Anderson RA (1997) J Inorg Biochem 66:119
Kita E, Lączna M (2001) Transit Met Chem 26:510
Emorson K, Graven WM (1959) Inorg Nucl 11:309
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdel-Messih, M.F., Abou-Gamra, Z.M. Kinetics and mechanism of the reaction between chromium(III) and picolinic acid in weak acidic aqueous solution. Monatsh Chem 143, 211–216 (2012). https://doi.org/10.1007/s00706-011-0598-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-011-0598-z