Abstract
The first-generation, live attenuated rotavirus (RV) vaccines, such as RotaTeq and Rotarix, were successful in reducing the number of RV-induced acute gastroenteritis (AGE) and child deaths globally. However, the low efficacy of these first-generation oral vaccines, coupled with safety concerns, required development of improved RV vaccines. The highly conserved structural protein VP6 is highly immunogenic, and it can generate self-assembled nano-sized structures, including tubes and spheres (virus-like particles; VLPs). Amongst the RV proteins, only VP6 shows these features. Interestingly, VP6-assembled structures, in addition to being highly immunogenic, have several other useful characteristics that could allow them to be used as adjuvants, immunological carriers, and drug-delivery vehicles as well as acting a scaffold for production of valuable nano-biomaterials. This review provides an overview of the self-assembled nano-sized structures of VP6-tubes/VLPs and their various functions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rotavirus (RV) virions are triple-layered particles with icosahedral symmetry, enclosing 11 segments of double-stranded (ds) RNA, the viral RNA-dependent RNA polymerase (VP1), and the capping enzyme (VP3) [14, 39, 45]. RVs were first identified in animals [1, 75], and later in humans [12, 13, 48, 63]. They are prominent causative agents of acute gastroenteritis (AGE) in children less than 5 years of age worldwide [93, 94, 112] and were responsible for around 500,000 deaths each year, prior to the beginning of global vaccination programs [112]. However, after the introduction of RV vaccines, the number of RV deaths decreased to 215,000 in 2013 [113] and then to 128,500 in 2016 [115], globally. The vaccines that have been introduced include live attenuated oral vaccines, which are used at both the global (RotaTeq and Rotarix) [7, 8, 19, 20, 106, 119, 120] and national (RotaVac, Rotavin-M1, ROTASIIL, and Lanzhou lamb RV vaccines) [7,8,9,10, 19, 20, 59] levels. Despite the success of live attenuated oral vaccines in reducing the number of RV deaths worldwide, several concerns, such as efficacy, safety, and cost, have impeded their development, and novel alternative approaches to producing RV vaccines are being considered.
Among the RV proteins, the highly conserved structural protein VP6 was the first RV antigen (Ag) to be used for classification of RV strains. Accordingly, 10 groups (A–J) and two new tentative groups, K (for RVC-like group) and L (for RVH-like group) [6, 58, 62, 82, 87], have been identified based on serological cross‐reaction to the VP6 protein (group Ag). In addition, further VP6 classification into four subgroups (SGI, II, I + II, and non‐I, non‐II) based on reactivity with one, both, or neither of the monoclonal antibodies (mAbs) 255/60 and 631/9 has been proposed [52, 73]. However, other classification systems based on the genotypes of the outer capsid proteins VP7 and VP4, the glycoprotein 36G, and the protease-sensitive protein 51P have also been described [58, 69, 79, 81]. Furthermore, a whole-genome‐based RV classification system has also been introduced that defines the genotypes of all 11 genomic RNA segments [79, 80].
Besides being a highly immunogenic protein containing dominant antigenic epitopes, VP6 is also the most abundant viral protein and is capable of self-assembling into complex structures when expressed in isolation under defined conditions. These structures include trimeric, spherical, and tubular forms and sheet structures. Structural proteins from several viruses such as hepatitis B virus (HBV), human papilloma virus (HPV), human immunodeficiency virus (HIV), adeno-associated virus (AAV), hepatitis C virus (HCV), norovirus (NoV), human influenza virus, respiratory syncytial virus (RSV), coronaviruses, and bacteriophages have also been shown to be capable of producing immunogenic VLPs that can be used for vaccine development [88, 100]. However, VP6 is one of only a limited number of structured viral protein assemblies that are able to be used as carrier proteins with strong adjuvant activity. Moreover, VP6 is one of the very few immune-carrier/adjuvant systems that are capable of producing tubular forms and sheet structures. One well-known example of a viral carrier protein is the highly immunogenic hepatitis B core antigen (HBcAg), which is used as a carrier for heterologous Ags/epitopes to enhance antibody production and T-cell responses directed to heterologous antigens [105]. However, despite the powerful adjuvant activity of HBcAg VLPs, they do not make tubular forms or sheet structures like VP6.
The first biochemical analyses of the native oligomeric structure of VP6 were performed using infected cells or virus particles, selectively removing VP6 from double-layered particles (DLPs; VP2/6) [11, 51]. In parallel, several other researchers attempting heterologous expression of RV VP6 also reported the production of this protein in its native oligomeric state [2, 17, 37, 38, 67, 72, 89, 90, 126, 127] and suggested the use of nano-tubular (tube)/nano-spherical (VLP) structures of VP6 as an immunological carrier for heterologous and homologous Ags [49, 102, 111] as well as a drug delivery system [126]. Of note, these structures were shown to react with VP6-specific mAbs, indicating that their native immunoreactive determinants were conserved [44]. It has also been shown that self-assembly of VP6 into nano-scale structures can create multifunctional scaffolds for construction of organized nanoparticle-based biomaterials with novel functions and characteristics [99]. Therefore, based on its strong immunogenicity, conserved characteristics, and ability to self-assemble, VP6 is being considered as both an antigen and a platform to develop alternative non-living RV vaccine candidates, as well as an organized nanoparticle-based structure for biomedical applications.
We have previously reviewed several promising VP6-based vaccine platforms (VP6 DNA vaccine, VP6 recombinant protein vaccine, and VP6-VLPs vaccine) [3, 60] that were able to induce heterologous cross-protective immunity to RV in animal models. In the present article, we provide an updated overview of potential applications of VP6 tubes/VLPs as an adjuvant, immunological carrier, and drug delivery tool and their use in the design of a new generation of vaccines and as a scaffold for generation of nano-biomaterials.
Vp6 expression and formation of structured VP6 tubes/VLPs
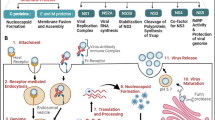
Various expression systems (Fig. 1A), from the prokaryotic E. coli [2, 72, 126] to eukaryotes such as yeast (including the baker's yeast Saccharomyces cerevisiae and the methylotrophic yeasts Pichia pastoris and Hansenula polymorpha), baculovirus-silkworm systems [17], insect cell/baculovirus (IC-BV) systems [38, 67], mammalian cells [38], herpes simplex virus 1 (HSV-1)-based vectors [90], and plant cells [37, 89, 127] have been used to produce multimeric VP6 structures that morphologically resemble VP2- and VP6-containing VLPs, as well as tubes.
The RV protein VP6 is capable of self-assembling into nano-sized spherical and tubular forms when expressed in isolation under defined conditions. (A) Different expression systems from prokaryotic E. coli [2, 72, 126] to eukaryotic systems such as yeast [17], insect cell/baculovirus systems (IC-BV) [38, 67], mammalian cells [38], HSV-1 [90], and plant cells [37, 89, 127] have been used successfully to produce assembled structures of VP6 tubes/VLPs. (B) Applications of VP6 tubes/VLPs, as vaccines (immunogenicity and protection) [15, 44, 54, 55, 64,65,66, 70, 95, 108], adjuvants [15, 16, 55, 76,77,78, 108,109,110], immunological carriers [49, 53, 92, 96, 102, 111], drug delivery tools [5, 91, 122, 126], and nano-biomaterials [5, 23, 24, 41, 42, 50, 91, 122, 126]
The assembly of VP6 into different structural forms depends on the pH, ionic strength, and concentrations of the divalent cations Ca+2 and Mg+2 [68, 101]. Spherical multimeric structures are formed in the pH range of 3.5-5.5, whereas large and small tubular multimeric structures are formed in the pH range of 5.5-7 and above 7, respectively [68, 86]. In contrast, structured protein assemblies of VP6 are disassembled at Ca+2 concentrations above 10 mM [68, 86] and at increased oxidant concentrations during the stages of the particle formation [25]. The formation of VP6 nanotubes in vitro was first reported in 1987 [101], but recently, a simple and efficient ultrafiltration method to purify VP6 tubes/VLPs in vitro was developed [67]. More recently, an easy and reproducible method to produce VP6 nanotubes from VP6 monomers was introduced that not only offers a novel approach to producing VP6 tubes in vitro but also makes it possible to modulate the length of the tubes [104]. Progress in the production/purification and development of various VP6 tubes/VLPs has provided the opportunity to investigate their potential application as immunogens, adjuvants, delivery tools, and nano-biomaterials (Table 1).
VP6 immunogenicity
Various RV VP6 Ag-encoding vaccine modalities, including DNA vaccines [3, 26,27,28, 36, 56, 57, 60, 74, 121, 123, 125, 128], subunit vaccines (harboring recombinant VP6 protein) [2, 3, 29,30,31,32,33,34,35, 40, 46, 60, 66, 83,84,85, 107, 116, 117, 121, 124, 127], and self‐assembled structures [3, 15, 16, 18, 44, 60, 64, 76,77,78, 95, 108, 110, 121] have been reported to induce an immune response and/or protection in animal models. The results of these animal immunization studies have indicated that, even in the absence of neutralizing Abs, immune responses to VP6 alone might be protective. It is known that neutralizing epitopes are only present on the VP4 and VP7 proteins of RV. Therefore, the observed protection of animals immunized with VP6 preparations lacking VP4 and VP7 proteins indicates that neutralizing Abs (directed against VP4 and VP7) might not be needed for protection against RV infection [3, 60]. Similarly, there have been several reports showing that non-neutralizing Abs are capable of clearing RV infection, suggesting the occurrence of intracellular inhibition of RV infection by IgA [4, 21, 47] and IgG [22] following transcytosis. Interestingly, among the various VP6 preparations, self-assembled VP6 structures such as VP6 tubes/VLPs have been reported to be the strongest immunogens, not only inducing a stronger immune response and a higher level of immunogenicity and protection [95] but also exhibiting adjuvant and immunological carrier properties [15].
VP6 tube/VLP immunogenicity and protective measures
Due to their superior immunogenicity compared to other VP6 preparations, VP6 tubes/VLPs have been selected for use in VP6-based RV vaccine candidates. Despite the challenges to efficient VLP generation on a large scale in insect cells, the establishment of IC-BV expression systems has been given high priority for production of VP6 tubes/VLPs. Indeed, in the first studies in 1987 [44], a recombinant baculovirus encoding the VP6 gene of strain SA11 was used to express VP6 proteins capable of assembling into tubular structures while maintaining their native antigenic determinants and oligomeric structure [44], and several studies demonstrated the immunogenicity of these insect-cell-derived VP6 tubes/VLPs. In this context, immunization of BALB/c mice with a combination of norovirus GII-4 VLPs and RV VP6 tubes resulted in a robust systemic cross-reactive and cross-blocking antibody response to both norovirus and RV [15], indicating sufficient immunogenicity of both Ags in the combined preparation without any dominance of one over the other. In line with this finding, a trivalent combination vaccine containing norovirus GII-4 and GI-3 VLPs and RV VP6 tubes was also formulated by the same group of the researchers [108]. Immunization of mice with this trivalent vaccine demonstrated the ability of VP6 tubes to induce a robust, long-lasting, high-avidity IgG responses against various RV strains [108]. In still another study by the same group, the immunogenicity of the assembled VP6 tubes and double-layered (dl) 2/6-VLPs produced by coexpression of VP2 and VP6 was investigated [64]. The results indicated that this vaccine formulation induced a balanced Th1/Th2-type immune response with high levels of cross-reactive serum IgG against different RV strains, as well as mucosal IgG and IgA Abs, and cellular immune responses with high levels of IFN-γ production [64]. Of particular note, the results of these and further studies indicated that mucosal VP6-specific Abs against VP6 tubes, which are found in the gut of immunized animals, might act as a first line of defense to inhibit infection by different RV strains in vitro and in vivo [64, 65, 108]. Similarly, other immunization studies in which VP6 assemblies were administered via the intramuscular or intranasal route in BALB/c mice indicated that serum VP6-specific IgA titers correlated with at least 65% protection against RV infection regardless of the delivery route [66]. These results suggested a significant role of mucosal immunity and VP6-specific IgA in inhibition of RV replication and heterotypic protection against RV infection. It should be noted, however, that immunization of BALB/c mice with VP6 tubes via the intradermal or intranasal route is also capable of generating diverse CD4+ T cell subsets and induction of the antiviral cytokine IFN-γ, interleukin-4 (IL-4), and the pro-inflammatory cytokine IL-17 [54]. The induction of such diverse cellular immune responses might imply that immunization with VP6 tubes provides protection against RV infection by other mechanisms besides intracellular inhibition by VP6-specific IgA and IgG.
In an attempt to assess the immunogenicity of the various assemblies of RV VP6 in vaccine formulations, the immune responses elicited by VP6 tubes, dl2/6-VLPs, and trimers were compared [95], and the results indicated that immunization of mice with one dose of VP6 tubes induced the highest IgG titers and provided a level of protection against RV infection similar to that of two doses of either dl2/6-VLPs or trimers, suggesting the superiority of RV VP6 tubes for vaccine formulations [95].
In addition to the baculovirus expression system, E. coli has also been used to produce various RV VP6 assemblies [18, 70,71,72]. The morphology of the E. coli-derived VP6 tubes/VLPs and trimers was verified using transmission electron microscopy (TEM) and atomic force microscopy (AFM) [18, 61, 70,71,72, 118]. Interestingly, the efficacy of the E. coli-derived VP6-VLPs or trimers against RV infection was significantly lower than that of E. coli-derived DLP2/6 [70]. In addition to baculovirus and E. coli expression systems, mammalian cells [38] and HSV-1-based vectors [90] as well as plant cells [37, 89, 127] have also been shown to have the capacity to produce self-assembled RV VP6 structures to induce protective immune responses against virus infection [78, 90].
Overall, among the different RV VP6 preparations, VP6 tubes/VLPs might have the highest potential as a non-living RV vaccine platform due to their strong immunogenicity and maintenance of conserved epitopes.
The adjuvant role of RV VP6 tubes/VLPs
Several studies have demonstrated the ability of RV VP6 tubes/VLPs to enhance immune responses against co‐administered homologous/heterologous antigens (Fig. 1B). The priming effect of immunization with the RV VP6 protein to enhance the generation of neutralizing Abs against heterotypic RV VP4 or VP7 proteins has been reported [43]. Several years later, in the early 2010s, the adjuvant effect of VP6 tubes/VLPs on a co-administered heterologous antigen (norovirus GI-3 protein), enhancing the induction of GI-3-specific cross-reactive Abs in the immunized mice, was shown [15]. In still another recent study, the adjuvant effect of VP6 tubes in mice immunized with suboptimal doses of norovirus GII.4 or GI.3 VLPs, either alone or in combination with VP6 tubes, was investigated [16]. The results of that study indicated that suboptimal doses of the norovirus VLPs alone did not induce substantial anti-norovirus Abs, but the combined vaccine induced considerable titers of cross-reactive Abs against both norovirus genotypes, indicating the adjuvant effect of RV VP6 on norovirus GII.4 or GI.3 VLPs [16]. It was later shown that the adjuvant effect of VP6 tubes on co-administered norovirus VLPs (as a mixture), compared to the separate administration of two antigens, is strictly dependent on co-localization of the VP6 with norovirus VLPs [77]. This finding was further supported by showing enhanced uptake of norovirus VLPs into antigen-presenting cells (APCs) [103] when mixed with VP6 tubes [76]. This process might induce the production of several cytokines/chemokines (tumor necrosis factor [TNF]-α, IL-6, IL-1, and granulocyte macrophage colony-stimulating factor [GMCSF]), indicating the potential adjuvant effect of VP6 on norovirus-specific T-cell immunity [76, 77]. The potential roles of the administered dose and morphological features of the VP6 structural assemblies on their adjuvant activity were also addressed in studies on immunization with norovirus VLP [77]. In this context, it was shown that despite higher induction of the cytokine IL-4 by co-administration of VP6 tubes with norovirus VLPs than with VP6 VLPs, both of these oligomeric VP6 assemblies exerted a similar dose-dependent adjuvant effect on norovirus-specific Ab responses [77]. More-recent studies have indicated that the particulate nature and size of the co-administered Ag might also affect the adjuvant effect of VP6 VLPs [55]. In this context, co-administration of VP6 VLPs with either a 20-nm norovirus P particle or a 23-mer extracellular domain of matrix protein M2 (M2e, a monomeric peptide, ~3 kDa) of human influenza A virus showed an adjuvant effect of VP6 VLPs only, enhancing Ab and T-cell immune responses against the preceding Ag (norovirus P particle), and not M2e [55]. VP6 tubes/VLPs have also been shown to enhance the uptake and presentation of co-administered norovirus GII.4 VLPs by dendritic cells [109, 110]. VP6 tube/VLP structures, and even their aggregates lacking high structural order, were efficiently internalized by bone-marrow-derived dendritic cells (BMDCs). Interestingly, compared to VP6 tube/VLP structures, an increased level of internalization was observed by the aggregates. Although the reasons behind these observations still remain to be explained, similar levels of IFN-γ production by splenocytes of VP6-immunized mice were detected regardless of the structural assembly [109]. Finally, it is worth mentioning that similar adjuvant effects were observed when plant-derived VP6 tubes were co-administered with plant-derived norovirus VLPs, indicating the independence of this adjuvant characteristic on the source of the cell/host used for vaccine production [78].
Overall, VP6 tubes/VLPs not only induce higher levels of immunogenicity compared to trimers or aggregates of VP6 but also have great potential to act as an adjuvant and to enhance prominent Ab and T-cell immune responses against foreign antigens.
VP6 tube/VLPs as immunological carriers
The strong immunogenicity and inherent immunostimulatory and immunomodulatory characteristics of VP6 tubes/VLPs make this structured protein assembly an important candidate vaccine-delivery platform for foreign/heterologous Ag presentation. The first reports on applications of RV VP6 spherical VLPs as antigen carriers were in the early 1990s, when heterologous peptides/proteins were efficiently coupled to these carriers and succssesfully used as strong immunogens to induce high titers of specific Abs against the coupled peptides/proteins without the use of adjuvants (Fig. 1B) [49, 102]. Since then, there have been many reports on the successful application of RV VP6 structured protein assemblies for chimeric Ag delivery, including insertion of (i) a 14-amino-acid peptide derived from the simian paramyxovirus 5 (V5 epitope) by genetic fusion [96], (ii) three epitopes of poliovirus type 1 (PV1) [92], (iii) three epitopes derived from the VP4 of RV [114], (iv) two peptides (23 and 140 amino acids) derived from the M2 and hemagglutinin (HA) genes of influenza A virus [53], and, more recently, (v) insertion of the receptor-binding domain (RBD) of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spike protein (S) to the surface loops or the N-terminus of VP6 without affecting protein conformation and immunogenicity [111]. The results of the immunization studies using these VP6-based chimeric Ag delivery systems in animal models have demonstrated the potency of VP6-based structured protein assemblies (VP6 tubes/VLPs) for induction of high titers of Abs and strong T-cell immune responses against both VP6 and the inserted Ag, suggesting the conformational conservation of both Ags in the chimera.
VP6 tubes/VLPs as drug-delivery tools
Besides serving as immunological carriers, RV VP6 structured protein assemblies in the form of nanotubes/VLPs are capable of encapsulating therapeutic reagents for the purpose of drug delivery (Fig. 1B) [122, 126]. Indeed, owing to their biocompatibility, biodegradability, and encapsulation characteristics, such viral nanoparticles are suggested to act as carriers of targeted therapeutics and anticancer vehicles [122]. In this context, it has been reported that self-assembled RV VP6 particles fused to the small ubiquitin modifier (SUMO) domain maintain their nanotube/VLP structure in the gastrointestinal (GI) tract when administered orally and represent a suitable biodegradable carrier [5, 91, 122, 126]. These results suggest the potential application of self-assembled RV VP6 particles for drug delivery to the GI tract.
VP6 tubes/VLPs as nano-biomaterials
Biological molecules with the ability to self-assemble are highly ordered, symmetrical, homogeneous structures with various sizes and shapes [41]. Among the different biomolecules (nucleic acids, proteins, peptides, and viral particles), viral protein assemblies, which are highly ordered complex structures with multifunctional binding properties and devoid of genetic material, would be ideal as a new platform for the synthesis of natural nano-materials [41, 42]. One protein with these features is RV VP6, which is ideal for obtaining tubes/VLPs that can form multifunctional scaffolds for construction of organized nano-biomaterials (Fig. 1B) [23, 97, 99]. It has also been shown that RV VP6 in the context of trimer and nanotube assembly can act as a scaffold to allow charge transfer processes and to create bio-electrochemical interfaces [50].
The selection of VP6 assemblies as scaffolds for the synthesis of the metallic nanoparticles, functionalized in situ with metals such as silver (Ag), gold (Au), platinum (Pt), and palladium (Pd) was first reported in 2009 [99]. Although functionalization with metals usually results to the formation of nanotubes/particles with the metal attached to the external surface [97,98,99], reduction of the metal in situ to produce nanorods and nanowires inside the nanotubes has also been reported [23]. Finally, it is worth mentioning that molecular docking simulations have shown that exposed residues of RV VP6 are predicted to be able to bind to Pd (II) ions and to produce nucleation sites for the growth, stabilization, and control of Pd particles [24].
Overall, the accumulated data indicate that RV VP6 tubes/VLPs can be used as scaffolds to generate nanotubes with metal on either the external or internal surface. These properties might find important applications in nano-biotechnology.
Conclusions
The highly conserved and abundant structural protein VP6 of RV is an immunogen that is capable of self-assembling into nano-sized spherical and tubular forms when expressed in isolation under defined conditions. Different expression systems from prokaryotic E. coli to eukaryotic systems such as yeast, insect cell/baculovirus systems, mammalian cells, and plant cells have been used successfully to produce assembled VP6 structures such as VP6 tubes/VLPs. These structures have been shown to be stronger immunogens than VP6 monomers for protecting vaccinated animals from RV infection and thus might be appropriate alternatives for RV vaccine formulations. VP6 tubes/VLPs also show adjuvant activity when co-administered with other Ags such as norovirus VLPs. This adjuvant activity, which involves enhanced uptake of the Ag into APCs, has been shown to be strictly dependent on co-localization of VP6 with norovirus VLPs. The applicability of VP6-tubes/VLPs as immunological carriers has been shown by fusing them to heterologous Ags from several viruses, such as polio virus, influenza virus, and SARS-CoV-2, and the successful delivery of the chimeric Ags. VP6 tubes/VLPs are biocompatible and biodegradable nanocompounds that are capable of encapsulating therapeutic reagents for treatment of diseases such as cancer. Finally, VP6 tubes/VLPs can form multifunctional scaffolds that can be used to create bio-electrochemical interfaces and for synthesis of organized nano-biomaterials and metallic nanoparticles. These properties of RV VP6 tubes/VLPs make them potentially suitable for various practical applications.
References
Adams W, Kraft L (1963) Epizootic diarrhea of infant mice: identification of the etiologic agent. Science 141:359–360
Afchangi A, Arashkia A, Shahosseini Z, Jalilvand S, Marashi SM, Roohvand F, Mohajel N, Shoja Z (2018) Immunization of mice by rotavirus NSP4-VP6 fusion protein elicited stronger responses compared to VP6 alone. Viral Immunol 31:233–241
Afchangi A, Jalilvand S, Mohajel N, Marashi SM, Shoja Z (2019) Rotavirus VP6 as a potential vaccine candidate. Rev Med Virol 29:e2027
Aiyegbo MS, Sapparapu G, Spiller BW, Eli IM, Williams DR, Kim R, Lee DE, Liu T, Li S, Woods VL Jr (2013) Human rotavirus VP6-specific antibodies mediate intracellular neutralization by binding to a quaternary structure in the transcriptional pore. PLoS ONE 8:e61101
Amalfitano A, Martini C, Nocca G, Papi M, De Spirito M, Sanguinetti M, Vitali A, Bugli F, Arcovito A (2019) A protein chimera self-assembling unit for drug delivery. Biotechnol Prog 35:e2769
Banyai K, Kemenesi G, Budinski I, Foldes F, Zana B, Marton S, Varga-Kugler R, Oldal M, Kurucz K, Jakab F (2017) Candidate new rotavirus species in Schreiber’s bats, Serbia. Infect Genet Evolut 48:19–26
Banyai K, Estes MK, Martella V, Parashar UD (2018) Viral gastroenteritis. Lancet 392:175–186
Bergman H, Henschke N, Hungerford D, Pitan F, Ndwandwe D, Cunliffe N, Soares-Weiser K (2021) Vaccines for preventing rotavirus diarrhoea: vaccines in use. Cochrane Database Syst Rev 11:CD008521.
Bhandari N, Rongsen-Chandola T, Bavdekar A, John J, Antony K, Taneja S, Goyal N, Kawade A, Kang G, Rathore SS, Juvekar S, Muliyil J, Arya A, Shaikh H, Abraham V, Vrati S, Proschan M, Kohberger R, Thiry G, Glass R, Greenberg HB, Curlin G, Mohan K, Harshavardhan GV, Prasad S, Rao TS, Boslego J, Bhan MK, India Rotavirus Vaccine G (2014) Efficacy of a monovalent human-bovine (116E) rotavirus vaccine in Indian infants: a randomised, double-blind, placebo-controlled trial. Lancet 383:2136–2143
Bhandari N, Rongsen-Chandola T, Bavdekar A, John J, Antony K, Taneja S, Goyal N, Kawade A, Kang G, Rathore SS, Juvekar S, Muliyil J, Arya A, Shaikh H, Abraham V, Vrati S, Proschan M, Kohberger R, Thiry G, Glass R, Greenberg HB, Curlin G, Mohan K, Harshavardhan GV, Prasad S, Rao TS, Boslego J, Bhan MK, India Rotavirus Vaccine G (2014) Efficacy of a monovalent human-bovine (116E) rotavirus vaccine in Indian children in the second year of life. Vaccine 32(Suppl 1):A110-116
Bican P, Cohen J, Charpilienne A, Scherrer R (1982) Purification and characterization of bovine rotavirus cores. J Virol 43:1113–1117
Bishop RF, Davidson GP, Holmes IH, Ruck BJ (1973) Virus particles in epithelial cells of duodenal mucosa from children with acute non-bacterial gastroenteritis. Lancet 2:1281–1283
Bishop RF, Davidson GP, Holmes IH, Ruck BJ (1974) Detection of a new virus by electron microscopy of faecal extracts from children with acute gastroenteritis. Lancet 1:149–151
Bishop RF (1996) Natural history of human rotavirus infection. Arch Virol Suppl 12:119–128
Blazevic V, Lappalainen S, Nurminen K, Huhti L, Vesikari T (2011) Norovirus VLPs and rotavirus VP6 protein as combined vaccine for childhood gastroenteritis. Vaccine 29:8126–8133
Blazevic V, Malm M, Arinobu D, Lappalainen S, Vesikari T (2016) Rotavirus capsid VP6 protein acts as an adjuvant in vivo for norovirus virus-like particles in a combination vaccine. Hum Vaccin Immunother 12:740–748
Bredell H, Smith JJ, Prins WA, Görgens JF, van Zyl WH (2016) Expression of rotavirus VP6 protein: a comparison amongst Escherichia coli, Pichia pastoris and Hansenula polymorpha. FEMS Yeast Res 16:fow001
Bugli F, Caprettini V, Cacaci M, Martini C, Paroni Sterbini F, Torelli R, Della Longa S, Papi M, Palmieri V, Giardina B, Posteraro B, Sanguinetti M, Arcovito A (2014) Synthesis and characterization of different immunogenic viral nanoconstructs from rotavirus VP6 inner capsid protein. Int J Nanomed 9:2727–2739
Burke RM, Tate JE, Kirkwood CD, Steele AD, Parashar UD (2019) Current and new rotavirus vaccines. Curr Opin Infect Dis 32:435
Burnett E, Parashar UD, Tate JE (2020) Real-world effectiveness of rotavirus vaccines, 2006–19: a literature review and meta-analysis. Lancet Glob Health 8:e1195–e1202
Burns JW, Siadat-Pajouh M, Krishnaney AA, Greenberg HB (1996) Protective effect of rotavirus VP6-specific IgA monoclonal antibodies that lack neutralizing activity. Science 272:104–107
Caddy SL, Vaysburd M, Wing M, Foss S, Andersen JT, O ‘Connell K, Mayes K, Higginson K, Iturriza-Gómara M, Desselberger U, Leo CJ (2020) Intracellular neutralisation of rotavirus by VP6-specific IgG. PLoS Pathog 16:e1008732
Carreño-Fuentes L, Ascencio JA, Medina A, Aguila S, Palomares LA, Ramírez OT (2013) Strategies for specifically directing metal functionalization of protein nanotubes: constructing protein coated silver nanowires. Nanotechnology 24:235602
Carreño-Fuentes L, Bahena D, Palomares LA, Ramírez OT, José-Yacamán M, Plascencia-Villa G (2016) Molecular docking and aberration-corrected STEM of palladium nanoparticles on viral templates. Metals 6:200
Castro-Acosta RM, Rodríguez-Limas WA, Valderrama B, Ramírez OT, Palomares LA (2014) Effect of metal catalyzed oxidation in recombinant viral protein assemblies. Microb Cell Fact 13:1–12
Chen SC, Fynan EF, Robinson HL, Lu S, Greenberg HB, Santoro JC, Herrmann JE (1997) Protective immunity induced by rotavirus DNA vaccines. Vaccine 15:899–902
Chen SC, Jones DH, Fynan EF, Farrar GH, Clegg JC, Greenberg HB, Herrmann JE (1998) Protective immunity induced by oral immunization with a rotavirus DNA vaccine encapsulated in microparticles. J Virol 72:5757–5761
Choi AH, Knowlton DR, McNeal MM, Ward RL (1997) Particle bombardment-mediated DNA vaccination with rotavirus VP6 induces high levels of serum rotavirus IgG but fails to protect mice against challenge. Virology 232:129–138
Choi AH, Basu M, McNeal MM, Clements JD, Ward RL (1999) Antibody-independent protection against rotavirus infection of mice stimulated by intranasal immunization with chimeric VP4 or VP6 protein. J Virol 73:7574–7581
Choi AH, Basu M, McNeal MM, Flint J, VanCott JL, Clements JD, Ward RL (2000) Functional mapping of protective domains and epitopes in the rotavirus VP6 protein. J Virol 74:11574–11580
Choi AH, McNeal MM, Basu M, Flint JA, Stone SC, Clements JD, Bean JA, Poe SA, VanCott JL, Ward RL (2002) Intranasal or oral immunization of inbred and outbred mice with murine or human rotavirus VP6 proteins protects against viral shedding after challenge with murine rotaviruses. Vaccine 20:3310–3321
Choi AH, McNeal MM, Flint JA, Basu M, Lycke NY, Clements JD, Bean JA, Davis HL, McCluskie MJ, VanCott JL, Ward RL (2002) The level of protection against rotavirus shedding in mice following immunization with a chimeric VP6 protein is dependent on the route and the coadministered adjuvant. Vaccine 20:1733–1740
Choi AH, McNeal MM, Basu M, Bean JA, VanCott JL, Clements JD, Ward RL (2003) Functional mapping of protective epitopes within the rotavirus VP6 protein in mice belonging to different haplotypes. Vaccine 21:761–767
Choi AH, Basu M, McNeal MM, Bean JA, Clements JD, Ward RL (2004) Intranasal administration of an Escherichia coli-expressed codon-optimized rotavirus VP6 protein induces protection in mice. Protein Expr Purif 38:205–216
Choi AH, Smiley K, Basu M (2005) Induction of immune responses and partial protection in mice after skin immunization with rotavirus VP6 protein and the adjuvant LT(R192G). Vaccine 23:2290–2293
Choi AH, Smiley K, Basu M, McNeal MM, Shao M, Bean JA, Clements JD, Stout RR, Ward RL (2007) Protection of mice against rotavirus challenge following intradermal DNA immunization by Biojector needle-free injection. Vaccine 25:3215–3218
Chung IS, Kim CH, Kim KI, Hong SH, Park JH, Kim JK, Kim WY (2000) Production of recombinant rotavirus VP6 from a suspension culture of transgenic tomato (Lycopersicon esculentum Mill.) cells. Biotechnol Lett 22:251–255
da Silva Junior HC, de Mendonça MCL, de Souza Pereira MC, da Rocha NA, de Azevedo MLB, Leite JPG, de Moraes MTB (2012) Comparison of two eukaryotic systems for the expression of VP6 protein of rotavirus species A: transient gene expression in HEK293-T cells and insect cell-baculovirus system. Biotechnol Lett 34:1623–1627
Desselberger U (2014) Rotaviruses. Virus Res 190:75–96
Dong JL, Liang BG, Jin YS, Zhang WJ, Wang T (2005) Oral immunization with pBsVP6-transgenic alfalfa protects mice against rotavirus infection. Virology 339:153–163
Douglas T, Young M (2006) Viruses: making friends with old foes. Science 312:873–875
Dujardin E, Peet C, Stubbs G, Culver JN, Mann S (2003) Organization of metallic nanoparticles using tobacco mosaic virus templates. Nano Lett 3:413–417
Esquivel FR, Lopez S, Guitierrez XL, Arias C (2000) The internal rotavirus protein VP6 primes for an enhanced neutralizing antibody response. Adv Virol 145:813–825
Estes MK, Crawford SE, Penaranda ME, Petrie BL, Burns JW, Chan WK, Ericson B, Smith GE, Summers MD (1987) Synthesis and immunogenicity of the rotavirus major capsid antigen using a baculovirus expression system. J Virol 61:1488–1494
Estes MK, Cohen J (1989) Rotavirus gene structure and function. Microbiol Rev 53:410–449
Feng H, Li X, Song W, Duan M, Chen H, Wang T, Dong J (2017) Oral administration of a seed-based bivalent rotavirus vaccine containing VP6 and NSP4 induces specific immune responses in mice. Front Plant Sci 8:910
Feng N, Lawton JA, Gilbert J, Kuklin N, Vo P, Prasad BV, Greenberg HB (2002) Inhibition of rotavirus replication by a non-neutralizing, rotavirus VP6-specific IgA mAb. J Clin Investig 109:1203–1213
Flewett TH, Bryden AS, Davies H (1973) Letter: virus particles in gastroenteritis. Lancet 2:1497
Frenchick PJ, Sabara MI, Ready KF, Babiuk LA (1992) Biochemical and immunological characterization of a novel peptide carrier system using rotavirus VP6 particles. Vaccine 10:783–791
Garcia-Garcia W, Vidal-Limon A, Arrocha-Arcos A, Palomares L, Ramirez O, Miranda-Hernández M (2019) Rotavirus VP6 protein as a bio-electrochemical scaffold: molecular dynamics and experimental electrochemistry. Bioelectrochemistry 127:180–186
Gorziglia M, Larrea C, Liprandi F, Esparza J (1985) Biochemical evidence for the oligomeric (possibly trimeric) structure of the major inner capsid polypeptide (45K) of rotaviruses. J Gen Virol 66:1889–1900
Greenberg H, McAuliffe V, Valdesuso J, Wyatt R, Flores J, Kalica A, Hoshino Y, Singh N (1983) Serological analysis of the subgroup protein of rotavirus, using monoclonal antibodies. Infect Immun 39:91–99
Gröhn S, Heinimäki S, Tamminen K, Blazevic V (2021) Expression of influenza A virus-derived peptides on a rotavirus VP6-based delivery platform. Adv Virol 166:213–217
Heinimäki S, Malm M, Vesikari T, Blazevic V (2018) Intradermal and intranasal immunizations with oligomeric middle layer rotavirus VP6 induce Th1, Th2 and Th17 T cell subsets and CD4+ T lymphocytes with cytotoxic potential. Antivir Res 157:1–8
Heinimäki S, Tamminen K, Hytönen VP, Malm M, Blazevic V (2020) Rotavirus inner capsid VP6 acts as an adjuvant in formulations with particulate antigens only. Vaccines 8:365
Herrmann JE, Chen SC, Fynan EF, Santoro JC, Greenberg HB, Robinson HL (1996) DNA vaccines against rotavirus infections. Arch Virol Suppl 12:207–215
Herrmann JE, Chen SC, Fynan EF, Santoro JC, Greenberg HB, Wang S, Robinson HL (1996) Protection against rotavirus infections by DNA vaccination. J Infect Dis 174(Suppl 1):S93-97
http://rega.kuleuven.be/cev/viralmetagenomics/virus-classification/rcwg. Accessed 25 Mar 2021
Isanaka S, Guindo O, Langendorf C, Matar Seck A, Plikaytis BD, Sayinzoga-Makombe N, McNeal MM, Meyer N, Adehossi E, Djibo A, Jochum B, Grais RF (2017) Efficacy of a low-cost, heat-stable oral rotavirus vaccine in Niger. N Engl J Med 376:1121–1130
Jalilvand S, Marashi SM, Shoja Z (2015) Rotavirus VP6 preparations as a non-replicating vaccine candidates. Vaccine 33:3281–3287
Jiménez-Zaragoza M, Yubero MP, Martín-Forero E, Castón JR, Reguera D, Luque D, de Pablo PJ, Rodríguez JM (2018) Biophysical properties of single rotavirus particles account for the functions of protein shells in a multilayered virus. Elife 7:e37295
Johne R, Tausch SH, Grützke J, Falkenhagen A, Patzina-Mehling C, Beer M, Höper D, Ulrich RG (2019) Distantly related rotaviruses in common shrews, Germany, 2004–2014. Emerg Infect Dis 25:2310
Kapikian AZ, Kim HW, Wyatt RG, Rodriguez WJ, Ross S, Cline WL, Parrott RH, Chanock RM (1974) Reoviruslike agent in stools: association with infantile diarrhea and development of serologic tests. Science 185:1049–1053
Lappalainen S, Tamminen K, Vesikari T, Blazevic V (2013) Comparative immunogenicity in mice of rotavirus VP6 tubular structures and virus-like particles. Hum Vaccin Immunother 9:1991–2001
Lappalainen S, Pastor AR, Tamminen K, Lopez-Guerrero V, Esquivel-Guadarrama F, Palomares LA, Vesikari T, Blazevic V (2014) Immune responses elicited against rotavirus middle layer protein VP6 inhibit viral replication in vitro and in vivo. Hum Vaccin Immunother 10:2039–2047
Lappalainen S, Pastor AR, Malm M, Lopez-Guerrero V, Esquivel-Guadarrama F, Palomares LA, Vesikari T, Blazevic V (2015) Protection against live rotavirus challenge in mice induced by parenteral and mucosal delivery of VP6 subunit rotavirus vaccine. Adv Virol 160:2075–2078
Lappalainen S, Vesikari T, Blazevic V (2016) Simple and efficient ultrafiltration method for purification of rotavirus VP6 oligomeric proteins. Adv Virol 161:3219–3223
Lepault J, Petitpas I, Erk I, Navaza J, Bigot D, Dona M, Vachette P, Cohen J, Rey FA (2001) Structural polymorphism of the major capsid protein of rotavirus. EMBO J 20:1498–1507
Li K, Lin XD, Huang KY, Zhang B, Shi M, Guo WP, Wang MR, Wang W, Xing JG, Li MH, Hong WS, Holmes EC, Zhang YZ (2016) Identification of novel and diverse rotaviruses in rodents and insectivores, and evidence of cross-species transmission into humans. Virology 494:168–177
Li T, Lin H, Zhang Y, Li M, Wang D, Che Y, Zhu Y, Li S, Zhang J, Ge S, Zhao Q, Xia N (2014) Improved characteristics and protective efficacy in an animal model of E. coli-derived recombinant double-layered rotavirus virus-like particles. Vaccine 32:1921–1931
Li Z, Cui K, Huang K, Liu F, Shi D, Liu Q (2019) Self-assembling rotavirus VP6 nanoparticle vaccines expressed in Escherichia coli elicit systemic and mucosal responses in mice. Protein Pept Lett 26:904–909
Li Z, Cui K, Wang H, Liu F, Huang K, Duan Z, Wang F, Shi D, Liu Q (2019) A milk-based self-assemble rotavirus VP6–ferritin nanoparticle vaccine elicited protection against the viral infection. J Nanobiotechnol 17:1–13
Lin Y-P, Kao C-L, Chang S-Y, Taniguchi K, Hung P-Y, Lin H-C, Huang L-M, Huang H-H, Yang J-Y, Lee C-N (2008) Determination of human rotavirus VP6 genogroups I and II by reverse transcription-PCR. J Clin Microbiol 46:3330–3337
Lopez-Guerrero DV, Arias N, Gutierrez-Xicotencatl L, Chihu-Amparan L, Gonzalez A, Pedroza-Saavedra A, Rosas-Salgado G, Villegas-Garcia JC, Badillo-Godinez O, Fernandez G, Lopez S, Esquivel-Guadarrama F (2018) Enhancement of VP6 immunogenicity and protective efficacy against rotavirus by VP2 in a genetic immunization. Vaccine 36:3072–3078
Malherbe H, Harwin R, Ulrich M (1963) The cytopathic effects of vervet monkey viruses. S Afr Med J 37:407–411
Malm M, Tamminen K, Lappalainen S, Vesikari T, Blazevic V (2016) Rotavirus recombinant VP6 nanotubes act as an immunomodulator and delivery vehicle for norovirus virus-like particles. J Immunol Res 2016:9171632
Malm M, Heinimaki S, Vesikari T, Blazevic V (2017) Rotavirus capsid VP6 tubular and spherical nanostructures act as local adjuvants when co-delivered with norovirus VLPs. Clin Exp Immunol 189:331–341
Malm M, Diessner A, Tamminen K, Liebscher M, Vesikari T, Blazevic V (2019) Rotavirus VP6 as an adjuvant for bivalent norovirus vaccine produced in Nicotiana benthamiana. Pharmaceutics 11:229
Matthijnssens J, Ciarlet M, Heiman E, Arijs I, Delbeke T, McDonald SM, Palombo EA, Iturriza-Gomara M, Maes P, Patton JT, Rahman M, Van Ranst M (2008) Full genome-based classification of rotaviruses reveals a common origin between human Wa-Like and porcine rotavirus strains and human DS-1-like and bovine rotavirus strains. J Virol 82:3204–3219
Matthijnssens J, Ciarlet M, Rahman M, Attoui H, Banyai K, Estes MK, Gentsch JR, Iturriza-Gomara M, Kirkwood CD, Martella V, Mertens PP, Nakagomi O, Patton JT, Ruggeri FM, Saif LJ, Santos N, Steyer A, Taniguchi K, Desselberger U, Van Ranst M (2008) Recommendations for the classification of group A rotaviruses using all 11 genomic RNA segments. Adv Virol 153:1621–1629
Matthijnssens J, Ciarlet M, McDonald SM, Attoui H, Banyai K, Brister JR, Buesa J, Esona MD, Estes MK, Gentsch JR, Iturriza-Gomara M, Johne R, Kirkwood CD, Martella V, Mertens PP, Nakagomi O, Parreno V, Rahman M, Ruggeri FM, Saif LJ, Santos N, Steyer A, Taniguchi K, Patton JT, Desselberger U, Van Ranst M (2011) Uniformity of rotavirus strain nomenclature proposed by the Rotavirus Classification Working Group (RCWG). Adv Virol 156:1397–1413
Matthijnssens J, Otto PH, Ciarlet M, Desselberger U, Van Ranst M, Johne R (2012) VP6-sequence-based cutoff values as a criterion for rotavirus species demarcation. Adv Virol 157:1177–1182
McNeal MM, VanCott JL, Choi AH, Basu M, Flint JA, Stone SC, Clements JD, Ward RL (2002) CD4 T cells are the only lymphocytes needed to protect mice against rotavirus shedding after intranasal immunization with a chimeric VP6 protein and the adjuvant LT(R192G). J Virol 76:560–568
McNeal MM, Stone SC, Basu M, Bean JA, Clements JD, Hendrickson BA, Choi AH, Ward RL (2006) Protection against rotavirus shedding after intranasal immunization of mice with a chimeric VP6 protein does not require intestinal IgA. Virology 346:338–347
McNeal MM, Basu M, Bean JA, Clements JD, Lycke NY, Ramne A, Lowenadler B, Choi AH, Ward RL (2007) Intrarectal immunization of mice with VP6 and either LT(R192G) or CTA1-DD as adjuvant protects against fecal rotavirus shedding after EDIM challenge. Vaccine 25:6224–6231
Mena JA, Ramírez OT, Palomares LA (2006) Intracellular distribution of rotavirus structural proteins and virus-like particles expressed in the insect cell-baculovirus system. J Biotechnol 122:443–452
Mihalov-Kovacs E, Gellert A, Marton S, Farkas SL, Feher E, Oldal M, Jakab F, Martella V, Banyai K (2015) Candidate new rotavirus species in sheltered dogs, Hungary. Emerg Infect Dis 21:660–663
Mohsen MO, Zha L, Cabral-Miranda G, Bachmann MF (2017) Major findings and recent advances in virus-like particle (VLP)-based vaccines. In: Seminars in immunology. Elsevier, pp 123–132
O’Brien GJ, Bryant CJ, Voogd C, Greenberg HB, Gardner RC, Bellamy AR (2000) Rotavirus VP6 expressed by PVX vectors in Nicotiana benthamiana coats PVX rods and also assembles into viruslike particles. Virology 270:444–453
Palacios CA, Claus J, Mattion N (2015) Rotavirus VP6 protein expressed in cell culture by HSV-1-based vectors. Rev Argent Microbiol 47:80–81
Palmieri V, Bugli F, Papi M, Ciasca G, Maulucci G, Galgano S, Arcovito A, Sanguinetti M, De Spirito M (2015) VP6-SUMO self-assembly as nanocarriers for gastrointestinal delivery. J Nanomater 16:1–6
Pan XX, Wang J, Xia WY, Li XF, Yang LJ, Huang C, Chen YD (2016) Immunogenicity and antigenicity of a recombinant chimeric protein containing epitopes of poliovirus type 1. Acta Virol 60:234–241
Parashar UD, Hummelman EG, Bresee JS, Miller MA, Glass RI (2003) Global illness and deaths caused by rotavirus disease in children. Emerg Infect Dis 9:565–572
Parashar UD, Gibson CJ, Bresee JS, Glass RI (2006) Rotavirus and severe childhood diarrhea. Emerg Infect Dis 12:304–306
Pastor AR, Rodriguez-Limas WA, Contreras MA, Esquivel E, Esquivel-Guadarrama F, Ramirez OT, Palomares LA (2014) The assembly conformation of rotavirus VP6 determines its protective efficacy against rotavirus challenge in mice. Vaccine 32:2874–2877
Peralta A, Molinari P, Taboga O (2009) Chimeric recombinant rotavirus-like particles as a vehicle for the display of heterologous epitopes. Virol J 6:192
Plascencia-Villa G, Mena JA, Castro-Acosta RM, Fabián JC, Ramírez OT, Palomares LA (2011) Strategies for the purification and characterization of protein scaffolds for the production of hybrid nanobiomaterials. J Chromatogr B 879:1105–1111
Plascencia-Villa G, Medina A, Palomares LA, Ramirez OT, Ascencio JA (2013) Structural characterization of rotavirus-directed synthesis and assembly of metallic nanoparticle arrays. J Nanosci Nanotechnol 13:5572–5579
Plascencia-Villa G, Saniger JM, Ascencio JA, Palomares LA, Ramírez OT (2009) Use of recombinant rotavirus VP6 nanotubes as a multifunctional template for the synthesis of nanobiomaterials functionalized with metals. Biotechnol Bioeng 104:871–881
Plummer EM, Manchester M (2011) Viral nanoparticles and virus-like particles: platforms for contemporary vaccine design. Wiley Interdiscip Rev Nanomed Nanobiotechnol 3:174–196
Ready K, Sabara M (1987) In vitro assembly of bovine rotavirus nucleocapsid protein. Virology 157:189–198
Redmond MJ, Ohmann HB, Hughes HP, Sabara M, Frenchick PJ, Poku SK, Ijaz MK, Parker MD, Laarveld B, Babiuk LA (1991) Rotavirus particles function as immunological carriers for the delivery of peptides from infectious agents and endogenous proteins. Mol Immunol 28:269–278
Rodriguez M, Wood C, Sanchez-Lopez R, Castro-Acosta RM, Ramirez OT, Palomares LA (2014) Understanding internalization of rotavirus VP6 nanotubes by cells: towards a recombinant vaccine. Adv Virol 159:1005–1015
Rodríguez M, Castro-Acosta RM, Ruiz-Morales ER, Villanueva-Flores F, Ramírez OT, Palomares LA (2021) A novel method for the in vitro assembly of virus-like particles and multimeric proteins. Biotechnol Lett 43:1155–1161
Roose K, Baets SD, Schepens B, Saelens X (2013) Hepatitis B core–based virus–like particles to present heterologous epitopes. Expert Rev Vaccines 12:183–198
Ruiz-Palacios GM, Perez-Schael I, Velazquez FR, Abate H, Breuer T, Clemens SC, Cheuvart B, Espinoza F, Gillard P, Innis BL, Cervantes Y, Linhares AC, Lopez P, Macias-Parra M, Ortega-Barria E, Richardson V, Rivera-Medina DM, Rivera L, Salinas B, Pavia-Ruz N, Salmeron J, Ruttimann R, Tinoco JC, Rubio P, Nunez E, Guerrero ML, Yarzabal JP, Damaso S, Tornieporth N, Saez-Llorens X, Vergara RF, Vesikari T, Bouckenooghe A, Clemens R, De Vos B, O’Ryan M, Human Rotavirus Vaccine Study G (2006) Safety and efficacy of an attenuated vaccine against severe rotavirus gastroenteritis. N Engl J Med 354:11–22
Smiley KL, McNeal MM, Basu M, Choi AH, Clements JD, Ward RL (2007) Association of gamma interferon and interleukin-17 production in intestinal CD4+ T cells with protection against rotavirus shedding in mice intranasally immunized with VP6 and the adjuvant LT(R192G). J Virol 81:3740–3748
Tamminen K, Lappalainen S, Huhti L, Vesikari T, Blazevic V (2013) Trivalent combination vaccine induces broad heterologous immune responses to norovirus and rotavirus in mice. PLoS ONE 8:e70409
Tamminen K, Heinimäki S, Gröhn S, Blazevic V (2020) Internalization and antigen presentation by mouse dendritic cells of rotavirus VP6 preparations differing in nanostructure. Mol Immunol 123:26–31
Tamminen K, Heinimäki S, Vesikari T, Blazevic V (2020) Rotavirus VP6 adjuvant effect on norovirus GII. 4 virus-like particle uptake and presentation by bone marrow-derived dendritic cells in vitro and in vivo. J Immunol Res 2020:3194704
Tamminen K, Heinimäki S, Gröhn S, Blazevic V (2021) Fusion protein of rotavirus VP6 and SARS-CoV-2 receptor binding domain induces T cell responses. Vaccines 9:733
Tate JE, Burton AH, Boschi-Pinto C, Steele AD, Duque J, Parashar UD, W-cGRS N (2012) 2008 estimate of worldwide rotavirus-associated mortality in children younger than 5 years before the introduction of universal rotavirus vaccination programmes: a systematic review and meta-analysis. Lancet Infect Dis 12:136–141
Tate JE, Burton AH, Boschi-Pinto C, Parashar UD, World Health Organization-Coordinated Global Rotavirus Surveillance N (2016) Global, regional, and national estimates of rotavirus mortality in children <5 years of age, 2000–2013. Clin Infect Dis 62(Suppl 2):S96–S105
Teng Y, Zhao B, Pan X, Wen Y, Chen Y (2014) A new rotavirus VP6-based foreign epitope presenting vector and immunoreactivity of VP4 epitope chimeric proteins. Viral Immunol 27:96–104
Troeger C, Khalil IA, Rao PC, Cao S, Blacker BF, Ahmed T, Armah G, Bines JE, Brewer TG, Colombara DV, Kang G, Kirkpatrick BD, Kirkwood CD, Mwenda JM, Parashar UD, Petri WA Jr, Riddle MS, Steele AD, Thompson RL, Walson JL, Sanders JW, Mokdad AH, Murray CJL, Hay SI, Reiner RC Jr (2018) Rotavirus vaccination and the global burden of rotavirus diarrhea among children younger than 5 years. JAMA Pediatr 172:958–965
Vega CG, Bok M, Vlasova AN, Chattha KS, Fernandez FM, Wigdorovitz A, Parreno VG, Saif LJ (2012) IgY antibodies protect against human Rotavirus induced diarrhea in the neonatal gnotobiotic piglet disease model. PLoS ONE 7:e42788
Vega CG, Bok M, Vlasova AN, Chattha KS, Gómez-Sebastián S, Nuñez C, Alvarado C, Lasa R, Escribano JM, Garaicoechea LL (2013) Recombinant monovalent llama-derived antibody fragments (VHH) to rotavirus VP6 protect neonatal gnotobiotic piglets against human rotavirus-induced diarrhea. PLoS Pathog 9:e1003334
Venkataraman S, Allison DP, Qi H, Morrell-Falvey JL, Kallewaard N, Crowe JE Jr, Doktycz MJ (2006) Automated image analysis of atomic force microscopy images of rotavirus particles. Ultramicroscopy 106:829–837
Vesikari T, Clark HF, Offit PA, Dallas MJ, DiStefano DJ, Goveia MG, Ward RL, Schodel F, Karvonen A, Drummond JE, DiNubile MJ, Heaton PM (2006) Effects of the potency and composition of the multivalent human-bovine (WC3) reassortant rotavirus vaccine on efficacy, safety and immunogenicity in healthy infants. Vaccine 24:4821–4829
Vesikari T, Matson DO, Dennehy P, Van Damme P, Santosham M, Rodriguez Z, Dallas MJ, Heyse JF, Goveia MG, Black SB, Shinefield HR, Christie CD, Ylitalo S, Itzler RF, Coia ML, Onorato MT, Adeyi BA, Marshall GS, Gothefors L, Campens D, Karvonen A, Watt JP, O’Brien KL, DiNubile MJ, Clark HF, Boslego JW, Offit PA, Heaton PM, Rotavirus E, Safety Trial Study T (2006) Safety and efficacy of a pentavalent human-bovine (WC3) reassortant rotavirus vaccine. N Engl J Med 354:23–33
Ward RL, McNeal MM (2010) VP6: a candidate rotavirus vaccine. J Infect Dis 202:S101–S107
Wu Y, Li J, Shin H-J (2021) Self-assembled viral nanoparticles as targeted anticancer vehicles. Biotechnol Bioprocess Eng 26:25–38
Yang K, Wang S, Chang KO, Lu S, Saif LJ, Greenberg HB, Brinker JP, Herrmann JE (2001) Immune responses and protection obtained with rotavirus VP6 DNA vaccines given by intramuscular injection. Vaccine 19:3285–3291
Yu J, Langridge W (2003) Expression of rotavirus capsid protein VP6 in transgenic potato and its oral immunogenicity in mice. Transgenic Res 12:163–169
Yuan L, Azevedo MS, Gonzalez AM, Jeong KI, Van Nguyen T, Lewis P, Iosef C, Herrmann JE, Saif LJ (2005) Mucosal and systemic antibody responses and protection induced by a prime/boost rotavirus-DNA vaccine in a gnotobiotic pig model. Vaccine 23:3925–3936
Zhao Q, Chen W, Chen Y, Zhang L, Zhang J, Zhang Z (2011) Self-assembled virus-like particles from rotavirus structural protein VP6 for targeted drug delivery. Bioconjug Chem 22:346–352
Zhou B, Zhang Y, Wang X, Dong J, Wang B, Han C, Yu J, Li D (2010) Oral administration of plant-based rotavirus VP6 induces antigen-specific IgAs, IgGs and passive protection in mice. Vaccine 28:6021–6027
Zhou H, Guo L, Wang M, Qu J, Zhao Z, Wang J, Hung T (2011) Prime immunization with rotavirus VLP 2/6 followed by boosting with an adenovirus expressing VP6 induces protective immunization against rotavirus in mice. Virol J 8:3
Acknowledgements
This study was funded and supported by Pasteur Institute of Iran through grant no. 1097.
Author information
Authors and Affiliations
Contributions
All authors contributed to the writing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicting financial or other interests.
Additional information
Handling Editor: Tim Skern.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shoja, Z., Jalilvand, S., Latifi, T. et al. Rotavirus VP6: involvement in immunogenicity, adjuvant activity, and use as a vector for heterologous peptides, drug delivery, and production of nano-biomaterials. Arch Virol 167, 1013–1023 (2022). https://doi.org/10.1007/s00705-022-05407-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-022-05407-9