Abstract
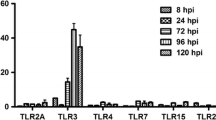
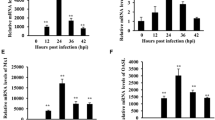
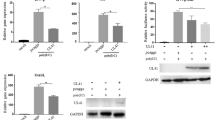
Newcastle disease virus (NDV) is an avian paramyxovirus that can selectively replicate in and destroy human tumor cells. In this report, we demonstrate that NDV infection in HeLa cells leads to the activation of the pattern recognition Toll-like receptor 3 (TLR3). Overexpression of TLR3 enhanced the activity of the IFN-β promoter and the transcription factor NF-kappa B (NF-κB), thereby decreasing viral protein synthesis and the virus titer. In addition, the reduction of endogenous TLR3 by small interfering RNA (siRNA) increased NDV replication. Similar anti-NDV effects were observed in DF-1 chicken fibroblast cells with the exogenous expression of chicken TLR3 (cTLR3). Immunofluorescence staining of HeLa cells indicated that the dsRNA generated during NDV replication colocalized with TLR3 in punctate subcellular structures. Altogether, our results strongly suggest that TLR3 actively participates in the recognition of the innate pro-inflammatory response after NDV infection and leads to the consequent antiviral cytokine/interferon secretion.







Similar content being viewed by others
References
Alexander D (1997) Newcastle disease and other avian Paramyxoviridae infections. In: Caineck BW (ed) Diseases of poultry. Iowa State University Press, Ames, pp 541–569
Alexopoulou L, Holt AC, Medzhitov R, Flavell RA (2001) Recognition of double-stranded RNA and activation of NF-kappaB by Toll-like receptor 3. Nature 413:732–738
Boukhvalova MS, Sotomayor TB, Point RC, Pletneva LM, Prince GA, Blanco JC (2010) Activation of interferon response through toll-like receptor 3 impacts viral pathogenesis and pulmonary toll-like receptor expression during respiratory syncytial virus and influenza infections in the cotton rat Sigmodon hispidus model. J Interferon Cytokine Res: Off J Int Soc Interferon Cytokine Res 30:229–242
Doyle SE, O’Connell R, Vaidya SA, Chow EK, Yee K, Cheng G (2003) Toll-like receptor 3 mediates a more potent antiviral response than Toll-like receptor. J Immunol (Baltimore, Md: 1950) 170:3565–3571
Fournier P, Wilden H, Schirrmacher V (2012) Importance of retinoic acid-inducible gene I and of receptor for type I interferon for cellular resistance to infection by Newcastle disease virus. Int J Oncol 40:287–298
Garcia-Sastre A, Biron CA (2006) Type 1 interferons and the virus-host relationship: a lesson in detente. Science (New York, NY) 312:879–882
Goodbourn S, Didcock L, Randall RE (2000) Interferons: cell signalling, immune modulation, antiviral response and virus countermeasures. J Gen Virol 81:2341–2364
Hewson CA, Jardine A, Edwards MR, Laza-Stanca V, Johnston SL (2005) Toll-like receptor 3 is induced by and mediates antiviral activity against rhinovirus infection of human bronchial epithelial cells. J Virol 79:12273–12279
Isogawa M, Robek MD, Furuichi Y, Chisari FV (2005) Toll-like receptor signaling inhibits hepatitis B virus replication in vivo. J Virol 79:7269–7272
Iwasaki A, Medzhitov R (2004) Toll-like receptor control of the adaptive immune responses. Nat Immunol 5:987–995
Jiang Z, Zamanian-Daryoush M, Nie H, Silva AM, Williams BR, Li X (2003) Poly(I-C)-induced Toll-like receptor 3 (TLR3)-mediated activation of NFkappa B and MAP kinase is through an interleukin-1 receptor-associated kinase (IRAK)-independent pathway employing the signaling components TLR3-TRAF6-TAK1-TAB2-PKR. J Biol Chem 278:16713–16719
Kato H, Sato S, Yoneyama M, Yamamoto M, Uematsu S, Matsui K, Tsujimura T, Takeda K, Fujita T, Takeuchi O, Akira S (2005) Cell type-specific involvement of RIG-I in antiviral response. Immunity 23:19–28
Kato H, Takeuchi O, Sato S, Yoneyama M, Yamamoto M, Matsui K, Uematsu S, Jung A, Kawai T, Ishii KJ, Yamaguchi O, Otsu K, Tsujimura T, Koh CS, Reis e Sousa C, Matsuura Y, Fujita T, Akira S (2006) Differential roles of MDA5 and RIG-I helicases in the recognition of RNA viruses. Nature 441:101–105
Lamb RA, Griffin DE (2007) Paramyxoviridae: the viruses and their replication. In: Knipe DM (ed) Field virology, 5th edn. Lippincott Williams & Wilkins, Philadelphia, pp 1449–1496
Lau YF, Tang LH, Ooi EE, Subbarao K (2010) Activation of the innate immune system provides broad-spectrum protection against influenza A viruses with pandemic potential in mice. Virology 406:80–87
Le Goffic R, Pothlichet J, Vitour D, Fujita T, Meurs E, Chignard M, Si-Tahar M (2007) Cutting Edge: Influenza A virus activates TLR3-dependent inflammatory and RIG-I-dependent antiviral responses in human lung epithelial cells. J Immunol 178:3368–3372
Li Q, Verma IM (2002) NF-kappaB regulation in the immune system. Nat Rev Immunol 2:725–734
Liang Z, Wu S, Li Y, He L, Wu M, Jiang L, Feng L, Zhang P, Huang X (2011) Activation of Toll-like receptor 3 impairs the dengue virus serotype 2 replication through induction of IFN-beta in cultured hepatoma cells. PLoS One 6:e23346
Manuse MJ, Parks GD (2010) TLR3-dependent upregulation of RIG-I leads to enhanced cytokine production from cells infected with the parainfluenza virus SV5. Virology 397:231–241
Matsumoto M, Seya T (2008) TLR3: interferon induction by double-stranded RNA including poly(I:C). Adv Drug Deliv Rev 60:805–812
Menager P, Roux P, Megret F, Bourgeois JP, Le Sourd AM, Danckaert A, Lafage M, Prehaud C, Lafon M (2009) Toll-like receptor 3 (TLR3) plays a major role in the formation of rabies virus Negri Bodies. PLoS Pathog 5:e1000315
Naka K, Dansako H, Kobayashi N, Ikeda M, Kato N (2006) Hepatitis C virus NS5B delays cell cycle progression by inducing interferon-beta via Toll-like receptor 3 signaling pathway without replicating viral genomes. Virology 346:348–362
Nelson CB, Pomeroy BS, Schrall K, Park WE, Lindeman RJ (1952) An outbreak of conjunctivitis due to Newcastle disease virus (NDV) occurring in poultry workers. Am J Public Health Nation’s Health 42:672–678
O’Neill LA, Golenbock D, Bowie AG (2013) The history of Toll-like receptors—redefining innate immunity. Nat Rev Immunol 13:453–460
Oshiumi H, Matsumoto M, Funami K, Akazawa T, Seya T (2003) TICAM-1, an adaptor molecule that participates in Toll-like receptor 3-mediated interferon-beta induction. Nat Immunol 4:161–167
Pichlmair A, Schulz O, Tan CP, Näslund TI, Liljeström P, Weber F, Reis e Sousa C (2006) RIG-I-mediated antiviral responses to single-stranded RNA bearing 5’-phosphates. Science 314:997–1001
Pothlichet J, Meunier I, Davis BK, Ting JP, Skamene E, von Messling V, Vidal SM (2013) Type I IFN triggers RIG-I/TLR3/NLRP3-dependent inflammasome activation in influenza A virus infected cells. PLoS Pathog 9:e1003256
Romer-Oberdorfer A, Werner O, Veits J, Mebatsion T, Mettenleiter TC (2003) Contribution of the length of the HN protein and the sequence of the F protein cleavage site to Newcastle disease virus pathogenicity. J Gen Virol 84:3121–3129
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 3:1101–1108
Seth RB, Sun L, Chen ZJ (2006) Antiviral innate immunity pathways. Cell Res 16:141–147
Sun Y, Ding N, Ding SS, Yu S, Meng C, Chen H, Qiu X, Zhang S, Yu Y, Zhan Y, Ding C (2013) Goose RIG-I functions in innate immunity against Newcastle disease virus infections. Mol Immunol 53:321–327
Sun Y, Yu S, Ding N, Meng C, Meng S, Zhang S, Zhan Y, Qiu X, Tan L, Chen H, Song C, Ding C (2014) Autophagy benefits the replication of newcastle disease virus in chicken cells and tissues. J Virol 88:525–537
Takeda K, Kaisho T, Akira S (2003) Toll-like receptors. Ann Rev Immunol 21:335–376
Takeuchi O, Akira S (2010) Pattern recognition receptors and inflammation. Cell 140:805–820
Taniguchi T, Takaoka A (2002) The interferon-alpha/beta system in antiviral responses: a multimodal machinery of gene regulation by the IRF family of transcription factors. Curr Opin Immunol 14:111–116
Trapp S, Derby NR, Singer R, Shaw A, Williams VG, Turville SG, Bess JW Jr, Lifson JD, Robbiani M (2009) Double-stranded RNA analog poly(I:C) inhibits human immunodeficiency virus amplification in dendritic cells via type I interferon-mediated activation of APOBEC3G. J Virol 83:884–895
Tsai YT, Chang SY, Lee CN, Kao CL (2009) Human TLR3 recognizes dengue virus and modulates viral replication in vitro. Cell Microbiol 11:604–615
Wang T, Town T, Alexopoulou L, Anderson JF, Fikrig E, Flavell RA (2004) Toll-like receptor 3 mediates West Nile virus entry into the brain causing lethal encephalitis. Nat Med 10:1366–1373
Wilson JR, de Sessions PF, Leon MA, Scholle F (2008) West Nile virus nonstructural protein 1 inhibits TLR3 signal transduction. J Virol 82:8262–8271
Yamamoto M, Sato S, Hemmi H, Hoshino K, Kaisho T, Sanjo H, Takeuchi O, Sugiyama M, Okabe M, Takeda K, Akira S (2003) Role of adaptor TRIF in the MyD88-independent toll-like receptor signaling pathway. Science (New York, NY) 301:640–643
Yoneyama M, Kikuchi M, Natsukawa T, Shinobu N, Imaizumi T, Miyagishi M, Taira K, Akira S, Fujita T (2004) The RNA helicase RIG-I has an essential function in double-stranded RNA-induced innate antiviral responses. Nat Immunol 5:730–737
Zamarin D, Palese P (2012) Oncolytic Newcastle disease virus for cancer therapy: old challenges and new directions. Future Microbiol 7:347–367
Zhou Y, Wang X, Liu M, Hu Q, Song L, Ye L, Zhou D, Ho W (2010) A critical function of toll-like receptor-3 in the induction of anti-human immunodeficiency virus activities in macrophages. Immunology 131:40–49
Acknowledgments
We are grateful to Professor Takashi Fujita (Kyoto University, Japan) for providing the p125-luc plasmid. This work was financially supported by the Chinese National High-Tech R&D Program (2011AA10A209), the Priority Academic Program Development of Jiangsu Higher Education Institutions, the China Agriculture Research System (CARS-41-K08), and the Chinese Special Fund for Agro-Scientific Research in the Public Interest (201003012 and 201303033).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Cheng, J., Sun, Y., Zhang, X. et al. Toll-like receptor 3 inhibits Newcastle disease virus replication through activation of pro-inflammatory cytokines and the type-1 interferon pathway. Arch Virol 159, 2937–2948 (2014). https://doi.org/10.1007/s00705-014-2148-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-014-2148-6