Abstract
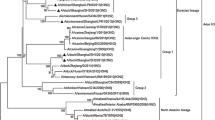
In 2007, a total of eight H9N2 influenza viruses were isolated from the water and fowl feces in Dongting Lake wetland, China. The genomes of the eight viruses were sequenced, and all eight gene segments were subjected to phylogenetic analysis. The results showed that all the isolates belonged to the same genotype, in which the HA, NA and NS gene segments were Chicken/Beijing/94-like; the PB2, PB1, PA and NP gene segments were Chicken/Shanghai/F/98-like; and the M gene was Quail/Hong Kong/G1/97-like. Animal experiments showed low pathogenicity of the selected viruses for chickens, although some chickens died after inoculation. The viruses showed no overt clinical signs in mice, but they could replicate in murine lungs prior to adaptation.



Similar content being viewed by others
References
Aamir UB, Wernery U, Ilyushina N, Webster RG (2007) Characterization of avian H9N2 influenza viruses from United Arab Emirates 2000 to 2003. Virology 361:45–55
Alexander DJ (2007) An overview of the epidemiology of avian influenza. Vaccine 25:5637–5644
Cameron KR, Gregory V, Banks J, Brown IH, Alexander DJ et al (2000) H9N2 subtype influenza A viruses in poultry in Pakistan are closely related to the H9N2 viruses responsible for human infection in Hong Kong. Virology 278:36–41
Guan Y, Shortridge KF, Krauss S, Chin PS, Dyrting KC et al (2000) H9N2 influenza viruses possessing H5N1-like internal genomes continue to circulate in poultry in southeastern China. J Virol 74:9372–9380
Homme PJ, Easterday BC (1970) Avian influenza virus infections. I. Characteristics of influenza A-turkey-Wisconsin-1966 virus. Avian Dis 14:66–74
Kim JA, Cho SH, Kim HS, Seo SH (2006) H9N2 influenza viruses isolated from poultry in Korean live bird markets continuously evolve and cause the severe clinical signs in layers. Vet Microbiol 118:169–176
Nili H, Asasi K (2002) Natural cases and an experimental study of H9N2 avian influenza in commercial broiler chickens of Iran. Avian Pathol 31:247–252
Peiris M, Yuen KY, Leung CW, Chan KH, Ip PL et al (1999) Human infection with influenza H9N2. Lancet 354:916–917
Guan Y, Shortridge KF, Krauss S, Webster RG (1999) Molecular characterization of H9N2 influenza viruses: were they the donors of the “internal” genes of H5N1 viruses in Hong Kong? Proc Natl Acad Sci USA 96:9363–9367
Tosh C, Nagarajan S, Behera P, Rajukumar K, Purohit K et al (2008) Genetic analysis of H9N2 avian influenza viruses isolated from India. Arch Virol 153:1433–1439
Xu KM, Smith GJ, Bahl J, Duan L, Tai H et al (2007) The genesis and evolution of H9N2 influenza viruses in poultry from southern China, 2000 to 2005. J Virol 81:10389–10401
Munster VJ, Baas C, Lexmond P, Waldenstrom J, Wallensten A et al (2007) Spatial, temporal, and species variation in prevalence of influenza A viruses in wild migratory birds. PLoS Pathog 3:e61
Webster RG, Bean WJ, Gorman OT, Chambers TM, Kawaoka Y (1992) Evolution and ecology of influenza A viruses. Microbiol Rev 56:152–179
Brown JD, Goekjian G, Poulson R, Valeika S, Stallknecht DE (2009) Avian influenza virus in water: infectivity is dependent on pH, salinity and temperature. Vet Microbiol 136:20–26
Brown JD, Swayne DE, Cooper RJ, Burns RE, Stallknecht DE (2007) Persistence of H5 and H7 avian influenza viruses in water. Avian Dis 51:285–289
Stallknecht DE, Shane SM, Kearney MT, Zwank PJ (1990) Persistence of avian influenza viruses in water. Avian Dis 34:406–411
Ito T, Okazaki K, Kawaoka Y, Takada A, Webster RG et al (1995) Perpetuation of influenza A viruses in Alaskan waterfowl reservoirs. Arch Virol 140:1163–1172
Khalenkov A, Laver WG, Webster RG (2008) Detection and isolation of H5N1 influenza virus from large volumes of natural water. J Virol Methods 149:180–183
Hoffmann E, Stech J, Guan Y, Webster RG, Perez DR (2001) Universal primer set for the full-length amplification of all influenza A viruses. Arch Virol 146:2275–2289
Li OT, Barr I, Leung CY, Chen H, Guan Y et al (2007) Reliable universal RT-PCR assays for studying influenza polymerase subunit gene sequences from all 16 haemagglutinin subtypes. J Virol Methods 142:218–222
Kumar S, Tamura K, Nei M (2004) MEGA3: integrated software for molecular evolutionary genetics analysis and sequence alignment. Brief Bioinform 5:150–163
Reed L, Muench H (1938) A simple method of estimating fifty percent endpoints. Am J Hyg 27:493–497
Callan RJ, Hartmann FA, West SE, Hinshaw VS (1997) Cleavage of influenza A virus H1 hemagglutinin by swine respiratory bacterial proteases. J Virol 71:7579–7585
Guo YJ, Krauss S, Senne DA, Mo IP, Lo KS et al (2000) Characterization of the pathogenicity of members of the newly established H9N2 influenza virus lineages in Asia. Virology 267:279–288
Lin YP, Shaw M, Gregory V, Cameron K, Lim W et al (2000) Avian-to-human transmission of H9N2 subtype influenza A viruses: relationship between H9N2 and H5N1 human isolates. Proc Natl Acad Sci USA 97:9654–9658
Suzuki H, Saito R, Masuda H, Oshitani H, Sato M et al (2003) Emergence of amantadine-resistant influenza A viruses: epidemiological study. J Infect Chemother 9:195–200
Chen H, Deng G, Li Z, Tian G, Li Y et al (2004) The evolution of H5N1 influenza viruses in ducks in southern China. Proc Natl Acad Sci USA 101:10452–10457
Lu JH, Liu XF, Shao WX, Liu YL, Wei DP et al (2005) Phylogenetic analysis of eight genes of H9N2 subtype influenza virus: a mainland China strain possessing early isolates’ genes that have been circulating. Virus Genes 31:163–169
Lang AS, Kelly A, Runstadler JA (2008) Prevalence and diversity of avian influenza viruses in environmental reservoirs. J Gen Virol 89:509–519
Vong S, Ly S, Mardy S, Holl D, Buchy P (2008) Environmental contamination during influenza A virus (H5N1) outbreaks, Cambodia, 2006. Emerg Infect Dis 14:1303–1305
Li KS, Xu KM, Peiris JS, Poon LL, Yu KZ et al (2003) Characterization of H9 subtype influenza viruses from the ducks of southern China: a candidate for the next influenza pandemic in humans? J Virol 77:6988–6994
Xu XJ, Xu GY, Zhou HB, Yu ZJ, Zhang AD et al (2008) Evolutionary characterization of influenza virus A/duck/Hubei/W1/2004 (H9N2) isolated from central China. Virus Genes 36:79–83
Buckler-White AJ, Naeve CW, Murphy BR (1986) Characterization of a gene coding for M proteins which is involved in host range restriction of an avian influenza A virus in monkeys. J Virol 57:697–700
Ward AC (1995) Specific changes in the M1 protein during adaptation of influenza virus to mouse. Arch Virol 140:383–389
Choi YK, Ozaki H, Webby RJ, Webster RG, Peiris JS et al (2004) Continuing evolution of H9N2 influenza viruses in Southeastern China. J Virol 78:8609–8614
Wu R, Sui ZW, Zhang HB, Chen QJ, Liang WW et al (2008) Characterization of a pathogenic H9N2 influenza A virus isolated from central China in 2007. Arch Virol 153:1549–1555
Acknowledgments
This study was supported by the following research funds: European Union Project (SSPE-CT-2006-44405); National 973 Project (2010CB530301); Chinese Academy of Sciences (KSCX1-YW-R-14); National Key Technology R&D Program of China (2006BAD06A03).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, H., Xu, B., Chen, Q. et al. Characterization of H9N2 influenza viruses isolated from Dongting Lake wetland in 2007. Arch Virol 156, 95–105 (2011). https://doi.org/10.1007/s00705-010-0836-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-010-0836-4