Abstract
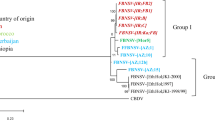
Fiji leaf gall is an important disease of sugarcane in Australia and other Asia-Pacific countries. The causative agent is the reovirus Fiji disease virus (FDV). Previous reports indicate that there is variation in pathology between virus isolates. To investigate the amount of genetic variation found in FDV, 25 field isolates from Australia, Papua New Guinea and Malaysia were analysed by partial sequencing of genome segments S3 and S9. There was up to 15% divergence in the nucleotide sequence among the 25 isolates. A similar amount of divergence and pattern of relationships was found for each of the two genomic segments for most of the field isolates, although reassortment of genome segments seems likely for at least one of the Papua New Guinean isolates. The finding of a high level of variation in FDV isolated in different regions has implications for quarantine and disease management.




Similar content being viewed by others
References
Croft BJ, Magarey R, Whittle P (2000) Disease management. In: Hogarth DM, Allsopp PG (eds) Manual of canegrowing. Bureau of Sugar Experiment Stations, Brisbane, pp 263–289
Daniels J, Ayub Husain A, Hutchinson PB, Wismer CA (1968) An insectary method for testing sugarcane varieties for resistance to Fiji disease. Proc Int Soc Sugar Cane Technol 13:1100–1106
Daniels J, Hussain AA, Hutchinson PB (1971) The control of sugarcane diseases in Fiji. Proc Int Soc Sugar Cane Technol 14:1007–1014
Egan BT, Ryan CC, Francki RIB (1989) Fiji disease. In: Ricaud C, Egan BT, Gillaspie AGJ, Hughes CG (eds) Diseases of sugarcane: major diseases. Elsevier, Amsterdam, pp 263–288
Fennah RG (1979) New species and new records of Perkinsiella (Hemiptera: Delphacidae) from Papua New Guinea. Bull Entomol Res 69:507–517
Francki RIB, Ryan CC, Hatta T, Rohozinski J, Grivell CJ (1986) Serological detection of Fiji disease virus antigens in the planthopper Perkinsiella saccharicida and its inefficient ability to transmit the virus. Plant Pathol 35:324–328
Gray MS, Banerjee N (1999) Mechanisms of arthropod transmission of plant and animal viruses. Microbiol Mol Biol Rev 63:128–148
Harding RM, Burns P, Geijskes RJ, McQualter RM, Dale JL, Smith GR (2006) Molecular analysis of Fiji disease virus segments 2, 4 and 7 completes the genome sequence. Virus Genes 32:43–47
Hayes AG (1974) Fiji disease of sugar cane: evidence for different strains of the causal virus at Condong, N.S.W. Proc Queensl Soc Sugar Cane Technol 41:105–110
Hussain AA, Brown AHD, Hutchinson PB, Wismer CA (1965) The testing of sugarcane varieties for resistance to Fiji disease Fiji. In: Congress of the international society of sugar cane technologists, San Juan, Puerto Rico. Elsevier, Amsterdam, pp 1154–1164
Idris AM, Brown JK (2004) Cotton leaf crumple virus is a distinct western hemisphere begomovirus species with complex evolutionary relationships indicative of recombination and reassortment. Phytopathology 94:1068–1074
James AP, Fowler AR, Hekmeijer S, Smith GR, Whittle PJL (1999) Indexing quarantined sugarcane for Fiji disease virus using RT-PCR. In: Asia-Pacific plant pathology for the new millennium, Canberra, p 51
McQualter R, Smith GR, Dale JL, Harding RM (2003) Molecular analysis of Fiji disease Fijivirus genome segments 1 and 3. Virus Genes 26:283–289
Mertens P (2004) The dsRNA viruses. Virus Res 101:3–13
Milne RC, del Vas M, Harding RM, Marzachi R, Mertens PPC (2005) Fijivirus. In: Fauquet CM, Mayo MA, Maniloff J, Desselberger U, Ball LA (eds) Virus taxonomy—eighth report of the international committee on taxonomy of viruses. Elsevier, San Diego, pp 534–542
Miranda GJ, Azzam O, Shirako Y (2000) Comparison of nucleotide sequences between northern and southern Philippine isolates of rice grassy stunt virus indicates occurrence of natural genetic reassortment. Virology 266:26–32
Mungomery RW, Bell AF (1933) Fiji disease of sugar-cane and its transmission Division of Pathology Bulletin. Bureau of Sugar Experiment Stations, Division of Pathology Bulletin
Ocfemia GO (1934) An insect vector of the Fiji disease of sugarcane. Am J Bot 21:113–120
Omura T, Yan J, Zhong B, Wada M, Zhu Y, Tomaru M, Maruyama W, Kikuchi A, Watanabe Y, Kimura I, Hibino H (1998) The P2 protein of rice dwarf phytoreovirus is required for adsorption of the virus to cells of the insect vector. J Virol 72:9370–9373
Omura T, Yan J (1999) Role of outer capsid proteins in transmission of Phytoreovirus by insect vectors. Adv Virus Res 54:15–43
Power AG (2000) Insect transmission of plant viruses: a constraint on virus variability. Curr Opin Plant Biol 3:336–340
Reddy DVR, Boccarda G, Outridge R, Teakle DS, Black LM (1975) Electrophoretic separation of dsRNA genome segments from Fiji disease and maize rough dwarf viruses. Virology 63:287–291
Ridley AW, Dhileepan K, Johnson KN, Allsopp PG, Nutt KA, Walter GH, Croft BJ (2006) Is the distribution of Fiji leaf gall in Australian sugarcane explained by the variation in the vector Perkinsiella saccharicida? Australas Plant Pathol 35:103–112
Smith GR, Van de Velde R, Dale JL (1992) PCR amplification of a specific double-standed RNA region of Fiji disease virus from diseased sugarcane. J Virol Meth 39:237–246
Smith GR, Van de Velde R (1994) Detection of sugarcane mosaic virus and Fiji disease virus in diseased sugarcane using the polymerase chain reaction. Plant Dis 78:557–561
Smith GR, Candy JM (2004) Improving Fiji diseases resistance screening trials in sugarcane by considering virus transmission class and possible origin of Fiji disease virus. Aust J Agric Res 55:665–672
Soo HM, Handley JA, Maugeri MM, Burns P, Smith GR, Dale JL, Harding RM (1998) Molecular characterization of Fiji disease fijivirus genome segment 9. J Genral Virol 79:3155–3161
Toohey CL, Nielsen PJ (1972) Fiji disease at Bundaberg. Proc Aust Soc Sugar Cane Technol 39:191–196
Uyeda I, Milne RG (1995) Introduction: genomic organization, diversity and evolution of plant reoviruses. Semin Virol 6:85–88
White WH, Reagan TE, Sosa O Jr (1995) The sugarcane delphacid (Homoptera: Delphacidae) extends its North American range into Louisiana. Fla Entomol 78:617–619
Whittle P (2000) Quarantine. In: Hogarth DM, Allsopp PG (eds) Manual of cane growing. Bureau of Sugar Experiment Stations, pp 339–352
Zhang H, Chen J, Adams M (2001) Molecular characterisation of segments 1 to 6 of Rice black-streaked dwarf virus from China provides the complete genome. Arch Virol 146:2331–2339
Zhang H, Chen J, Lei J, Adams M (2001) Sequence analysis shows that a dwarfing disease on rice, maize and wheat in China is caused by Rice black-streaked dwarf virus (RBSDV). Eur J Plant Pathol 107:563–567
Acknowledgments
We thank Anthony James (BSES) for co-ordination of samples and FDV expertise. For collection of samples we thank Robert Magarey, PNG; Tan Tack Nee, Malaysia; Robert Aitken, Harwood; Anthony Cattle, Newrybar; Victor Schwenke, Rocky Point. The collection of samples from Papua New Guinea was funded by Australian Centre for International Agricultural Research.
Author information
Authors and Affiliations
Corresponding author
Additional information
The sequence data presented in this paper are available in the GenBank database under the accession numbers EU434958–EU435006.
Rights and permissions
About this article
Cite this article
Jiang, J., Ridley, A.W., Tang, H. et al. Genetic variability of genome segments 3 and 9 of Fiji disease virus field isolates. Arch Virol 153, 839–848 (2008). https://doi.org/10.1007/s00705-008-0058-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-008-0058-1