Abstract
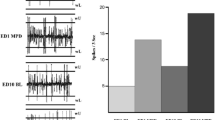
The objective of this study is to gain insight into the behavioral and neuronal changes induced by acute and chronic methylphenidate (MPD) administration. Specifically, there is limited knowledge of the effects of MPD on the locus coeruleus (LC), the main site of norepinephrine synthesis in the brain. In this study, LC neuronal firing rate was recorded simultaneously with locomotor activity in freely moving adolescent rats. Adolescent rats were chosen to mimic the age group in humans most affected by MPD exposure. Following acute dose of 0.6, 2.5 or 10 mg/kg MPD, all rats showed an increase in locomotor activity. However, in response to chronic MPD doses, individual rats showed either a further increase or decrease in their locomotor activity as compared to the effect initiated by the acute dose—expressing either behavioral sensitization or tolerance, respectively. The LC neuronal recordings from animals expressing behavioral sensitization showed that the majority of units responded to chronic MPD exposure by further increasing firing rates as compared to the initial response to the acute MPD exposure. For the LC neuronal units recorded from animals expressing behavioral tolerance, however, the majority of the units responded to chronic exposure by attenuating or no significant effect on their firing rate as compared to the acute MPD exposure. This observation indicates a correlation between the LC neuronal responses and behavioral activity to chronic MPD exposure. The study shows that LC participates in the effect of MPD and the behavioral expression of sensitization and tolerance to chronic exposure of MPD.





Similar content being viewed by others
References
Algahim MF, Yang PB, Wilcox VT, Burau KD, Swann AC, Dafny N (2009) Prolonged methylphenidate treatment alters the behavioral diurnal activity pattern of adult male Sprague-Dawley rats. Pharmacol Biochem Behav 92(1):93–99
Amaral DG, Sinnamon HM (1977) The locus coeruleus: neurobiology of a central noradrenergic nucleus. Prog Neurobiol 9(3):147–196
Askenasy EP, Taber KH, Yang PB, Dafny N (2007) Methylphenidate (Ritalin): behavioral studies in the rat. Int J Neurosci 117(6):757–794
Barron E, Yang PB, Swann AC, Dafny N (2009) Adolescent and adult male spontaneous hyperactive rats (SHR) respond differently to acute and chronic methylphenidate (Ritalin). Int J Neurosci 119(1):40–58
Biederman J, Spencer T (1999) Attention-deficit/hyperactivity disorder (ADHD) as a noradrenergic disorder. Biol Psychiatry 46(9):1234–1242
Britton GB (2012) Cognitive and emotional behavioural changes associated with methylphenidate treatment: a review of preclinical studies. Int J Neuropsychopharmacol 15(1):41–53
Claussen CM, Chong SL, Dafny N (2014) Nucleus accumbens neuronal activity correlates to the animal’s behavioral response to acute and chronic methylphenidate. Physiol Behav 22(129):85–94
Dafny N (1980) Neurophysiological evidence for tolerance and dependence on opiates: simultaneous multiunit recordings from septum, thalamus, and caudate nucleus. J Neurosci Res 5(4):339–349
Deupree JD, Reed AL, Bylund DB (2007) Differential effects of the tricyclic antidepressant desipramine on the density of adrenergic receptors in juvenile and adult rats. J Pharmacol Exp Ther 321(2):770–776
Drouin C, Page M, Waterhouse B (2006) Methylphenidate enhances noradrenergic transmission and suppresses mid- and long-latency sensory responses in the primary somatosensory cortex of awake rats. J Neurophysiol 96(2):622–632
Dulcan M (1997) Practice parameters for the assessment and treatment of children, adolescents, and adults with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry 36(10 Suppl):85S–121S
Frolov A, Reyes-Vasquez C, Dafny N (2015) Behavioral and neuronal recording of the nucleus accumbens in adolescent rats following acute and repetitive exposure to methylphenidate. J Neurophysiol 113(1):369–379
Gaytan O, Yang P, Swann A, Dafny N (2000) Diurnal differences in sensitization to methylphenidate. Brain Res 864(1):24–39
Godfrey J (2009) Safety of therapeutic methylphenidate in adults: a systematic review of the evidence. J Psychopharmacol 23(2):194–205
Greely H, Sahakian B, Harris J, Kessler RC, Gazzaniga M, Campbell P, Farah MJ (2008) Towards responsible use of cognitive-enhancing drugs by the healthy. Nature 456(7223):702–705
Igata S, Hayashi T, Itoh M, Akasu T, Takano M, Ishimatsu M (2014) Persistent α1-adrenergic receptor function in the nucleus locus coeruleus causes hyperexcitability in AD/HD model rats. J Neurophysiol 111(4):777–786
Jones Z, Dafny N (2014) Acute and chronic dose-response effect of methylphenidate on ventral tegmental area neurons correlated with animal behavior. J Neural Transm (Vienna) 121(3):327–345
Kalivas PW, Duffy P, DuMars LA, Skinner C (1988) Behavioral and neurochemical effects of acute and daily cocaine administration in rats. J Pharmacol Exp Ther 245(2):485–492
Kidani Y, Ishimatsu M, Akasu T (2010) Methylphenidate enhances inhibitory synaptic transmission by increasing the content of norepinephrine in the locus coeruleus of juvenile rats. Kurume Med J 57(1–2):29–38
Kuczenski R, Segal DS (2001) Locomotor effects of acute and repeated threshold doses of amphetamine and methylphenidate: relative roles of dopamine and norepinephrine. J Pharmacol Exp Ther 296(3):876–883
Kuczenski R, Segal DS (2002) Exposure of adolescent rats to oral methylphenidate: preferential effects on extracellular norepinephrine and absence of sensitization and cross-sensitization to methamphetamine. J Neurosci 22(16):7264–7271
Kushikata T, Yoshida H, Kudo M, Kudo T, Kudo T, Hirota K (2011) Role of coerulean noradrenergic neurones in general anaesthesia in rats. Br J Anaesth 107(6):924–929
Lacroix D, Ferron A (1988) Electrophysiological effects of methylphenidate on the coeruleo-cortical noradrenergic system in the rat. Eur J Pharmacol 149(3):277–285
Leblanc-Duchin D, Taukulis HK (2004) Behavioral reactivity to a noradrenergic challenge after chronic oral methylphenidate (ritalin) in rats. Pharmacol Biochem Behav 79(4):641–649
Ma CL, Arnsten AF, Li BM (2005) Locomotor hyperactivity induced by blockade of prefrontal cortical alpha2-adrenoceptors in monkeys. Biol Psychiatry 57(2):192–195
Moore RY, Bloom FE (1979) Central catecholamine neuron systems: anatomy and physiology of the norepinephrine and epinephrine systems. Annu Rev Neurosci 2:113–168
Morton WA, Stockton GG (2000) Methylphenidate Abuse and Psychiatric Side Effects. Prim Care Companion J Clin Psychiatry 2(5):159–164
Murrin LC, Sanders JD, Bylund DB (2007) Comparison of the maturation of the adrenergic and serotonergic neurotransmitter systems in the brain: implications for differential drug effects on juveniles and adults. Biochem Pharmacol 73(8):1225–1236
Nemoda Z, Angyal N, Tarnok Z, Gadoros J, Sasvari-Szekely M (2009) Carboxylesterase 1 gene polymorphism and methylphenidate response in ADHD. Neuropharmacology 57(7–8):731–733
Olpe HR, Berecek KH, Jones RS, Steinmann MW, Sonnenburg C, Hofbauer KG (1985) Reduced activity of locus coeruleus neurons in hypertensive rats. J Hypertens 3(4):S93–S95
Pliszka SR, McCracken JT, Maas JW (1996) Catecholamines in attention-deficit hyperactivity disorder: current perspectives. J Am Acad Child Adolesc Psychiatry 35(3):264–272
Podet A, Lee MJ, Swann AC, Dafny N (2010) Nucleus accumbens lesions modulate the effects of methylphenidate. Brain Res Bull 82(5–6):293–301
Rebec GV, Segal DS (1978) Dose-dependent biphasic alterations in the spontaneous activity of neurons in the rat neostriatum produced by d-amphetamine and methylphenidate. Brain Res 150(2):353–366
Robinson TE (1984) Behavioral sensitization: characterization of enduring changes in rotational behavior produced by intermittent injections of amphetamine in male and female rats. Psychopharmacology 84(4):466–475
Robinson TE, Berridge KC (1993) The neural basis of drug craving: an incentive-sensitization theory of addiction. Brain Res Brain Res Rev 18(3):247–291
Sanchez-Mora C, Ribases M, Mulas F, Soutullo C, Sans A, Pamias M, Casas M, Ramos-Quiroga JA (2012) Genetic bases of attention deficit hyperactivity disorder. Rev Neurol. 55(10):609–618
Sherwood N, Timiras PS (1970) A stereotaxic atlas of the developing rat brain. University of California Press, Berkeley
Stix G (2009) Turbocharging the brain. Sci Am 301(4):46–49 (52–55)
Tang B, Dafny N (2012) Methylphenidate modulates the locus coeruleus neuronal activity in freely behaving rat. Eur J Pharmacol 695(1–3):48–56
Tang B, Dafny N (2013) Behavioral and dorsal raphe neuronal activity following acute and chronic methylphenidate in freely behaving rats. Brain Res Bull 98:53–63
Volkow ND, Swanson JM (2008) The action of enhancers can lead to addiction. Nature 451(7178):520
Volkow ND, Ding YS, Fowler JS, Wang GJ, Logan J, Gatley SJ, Schlyer DJ, Pappas N (1995) A new PET ligand for the dopamine transporter: studies in the human brain. J Nucl Med 36(12):2162–2168
Volkow ND, Wang GJ, Fowler JS, Ding YS (2005) Imaging the effects of methylphenidate on brain dopamine: new model on its therapeutic actions for attention-deficit/hyperactivity disorder. Biol Psychiatry 57(11):1410–1415 (Epub 2005 Jan 12)
Yang PB, Amini B, Swann AC, Dafny N (2003) Strain differences in the behavioral responses of male rats to chronically administered methylphenidate. Brain Res 971(2):139–152
Yang PB, Swann AC, Dafny N (2006) Sensory-evoked potentials recordings from the ventral tegmental area, nucleus accumbens, prefrontal cortex, and caudate nucleus and locomotor activity are modulated in dose-response characteristics by methylphenidate. Brain Res 16(1073–1074):164–174
Yang PB, Swann AC, Dafny N (2007) Methylphenidate treated at the test cage–dose-dependent sensitization or tolerance depend on the behavioral assay used. Crit Rev Neurobiol 19(1):59–77
Acknowledgements
This study was funded by NIH DA ROI 027222 Grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declared that they have no potential conflict of interest.
Rights and permissions
About this article
Cite this article
Kharas, N., Reyes-Vazquez, C. & Dafny, N. Locus coeruleus neuronal activity correlates with behavioral response to acute and chronic doses of methylphenidate (Ritalin) in adolescent rats. J Neural Transm 124, 1239–1250 (2017). https://doi.org/10.1007/s00702-017-1760-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-017-1760-5